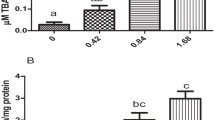
Abstract
The increase in the area treated with the insecticide fipronil has caused concern for aquatic organisms such as fish. Here, we assessed the effect of fipronil on carp indexes of non-specific immunity, oxidative stress, autophagy, and apoptosis following exposure to 0.074 mg/L and 0.185 mg/L of fipronil in the aqueous environment for 1 day, 3 days, 5 days, and 7 days. It was found that glutathione (GSH), malonaldehyde (MDA), and superoxide dismutase (SOD) in gills were significantly reduced (P < 0.05). The increase in exposure time increases the impact on GSH, SOD, and MDA parameters in the liver and intestine. Liver acid phosphatase (ACP), alkaline phosphatase (AKP), and lysozyme (LZM) activity levels increased significantly in the treatment group on the first day after exposure, except for the 0.074 mg/L group of ACP (P < 0.05). The mRNA expression levels of autophagy-related genes ATG12, ATG5, ATG16L, LC3-II, and BECN1 were generally elevated in the liver and intestine during the initial exposure period (P < 0.05), while mTOR was significantly reduced on the first and third days after treatment (P < 0.05). From the results of Western blotting (WB), we can see that the amount of LC3-II was significantly higher than that of LC3-I at 1, 3, and 5 days of exposure (P < 0.05). Furthermore, the apoptosis-related gene Bcl-2 reached its peak in the liver, intestine, and gill on the first day, and caspase3 was significantly downregulated throughout the exposure period (P < 0.05). The results showed that fipronil was potentially harmful to carp and should be used moderately to reduce the damage to aquatic ecosystems. This study complements the mechanism theory of fipronil on fish toxicology and has a certain value for human health risk assessment.









Similar content being viewed by others
Availability of data and materials
I am authorized to confirm that the materials and data in the manuscript are available on behalf of all authors of this article. The work in this article is original and has not been published previously, and the article is not under consideration by any other journal.
References
Aita VM, Liang XH, Murty VV, Pincus LD, Yu WP, Cayanis E, Kalachikov S, Gilliam TC, Levine B (1999) Cloning and genomic organization of beclin 1, a candidate tumor suppressor gene on chromosome 17q21. Genomics 59(1):59–65. https://doi.org/10.1006/geno.1999.5851
Almeida M, Fanini F, Davino S, Aznar A, Koch O, de Moraes Barros S (1997) Pro-and antioxidant parameters in rat liver after short term exposure to hexachlorobenzene. Hum Exp Toxicol 16:257–261
Beketov MA, Matthias L (2008) Potential of 11 pesticides to initiate downstream drift of stream macroinvertebrates. Arch Environ Contam Toxicol 55:247–253
Chaitali B, Goswami R, Verma G, Datta M, Mazumder S (2012) Aeromonas hydrophila induced head kidney macrophage apoptosis in Clarias batrachus involves the activation of calpain and is caspase-3 mediated. Dev Comp Immunol 37:323–333. https://doi.org/10.1016/j.dci.2012.02.005
Chen D, Zhang Z, Yao H, Liang Y, Xing H, Xu S (2015) Effects of atrazine and chlorpyrifos on oxidative stress-induced autophagy in the immune organs of common carp (Cyprinus carpio L.). Fish Shellfish Immunol 44(1):12–20. https://doi.org/10.1016/j.fsi.2015.01.014
Chen J, Liu N, Zhang H, Zhao Y, Cao X (2020) The effects of Aeromonas hydrophila infection on oxidative stress, nonspecific immunity, autophagy, and apoptosis in the common carp. Dev Comp Immunol 105:103587. https://doi.org/10.1016/j.dci.2019.103587
Dong C, Chen L, Feng J, Xu J, Mahboob S, Al-Ghanim K, Li X, Xu P (2016) Genome wide identification, phylogeny, and expression of aquaporin genes in common carp (Cyprinus carpio). PLoS One 11(12):e0166160. https://doi.org/10.1371/journal.pone.0166160
Fazeli F, Ghorbanli M, Niknam V (2007) Effect of drought on biomass, protein content, lipid peroxidation and antioxidant enzymes in two sesame cultivars. Biol Plant 51(1):98–103
Fridovich I (1995) Superoxide radical and superoxide dismutases. Annu Rev Biochem 64:97–112. https://doi.org/10.1146/annurev.bi.64.070195.000525
Geng R, Jia Y, Chi M, Wang Z, Liu H, Wang W (2019) RNase1 alleviates the Aeromonas hydrophila-induced oxidative stress in blunt snout bream. Dev Comp Immunol 91:8–16. https://doi.org/10.1016/j.dci.2018.09.018
Gisbert E, Nolasco H, Solovyev M (2018) Towards the standardization of brush border purification and intestinal alkaline phosphatase quantification in fish with notes on other digestive enzymes. Aquaculture 487:102–108
Grassme H, Jendrossek V, Gulbins E (2001) Molecular mechanisms of bacteria induced apoptosis. Apoptosis 6:441–445. https://doi.org/10.1023/A:1012485506972
Gunasekara AS, Tresca T, Goh KS, Frank S, Tjeerdema R (2007) Environmental fate and toxicology of fipronil. J Pestic Sci 32(3):189–199
Hayasaka D, Korenaga T, Suzuki K, Sã NF, Goka K (2012a) Differences in susceptibility of five cladoceran species to two systemic insecticides, imidacloprid and fipronil. Ecotoxicology 21(2):421–427
Hayasaka D, Korenaga T, Suzuki K, Saito F, Sánchezbayo F, Goka K (2012b) Cumulative ecological impacts of two successive annual treatments of imidacloprid and fipronil on aquatic communities of paddy mesocosms. Ecotoxicol Environ Saf 80(2):355–362
Jin Y, Wu S, Zeng Z, Fu Z (2017) Effects of environmental pollutants on gut microbiota. Environ Pollut 222:1–9
Kabeya Y, Mizushima N, Ueno T, Yamamoto A, Kirisako T, Noda T (2000) LC3, a mammalian homologue of yeast Apg8p, is localized in autophagosome membranes after processing. EMBO J 19:5720–5728
Kamada Y, Sekito T, Ohsumi Y (2004) Autophagy in yeast: a TOR-mediated response to nutrient starvation. Curr Top Microbiol Immunol 279:73–84
Khan S, Jan MH, Kumar D, Telang AG (2015) Fipronil induced spermotoxicity is associated with oxidative stress, DNA damage and apoptosis in male rats. Pestic Biochem Physiol 124:8–14
Kim YC, Guan KL (2015) mTOR: a pharmacologic target for autophagy regulation. J Clin Investig 125(1):25–32. https://doi.org/10.1172/JCI73939
Klionsky DJ (2007) Autophagy: from phenomenology to molecular understanding in less than a decade. Nat Rev Mol Cell Biol 8:931–937
Kong X, Wang S, Jiang H, Nie G, Li X (2012) Responses of acid/alkaline phosphatase, lysozyme, and catalase activities and lipid peroxidation to mercury exposure during the embryonic development of goldfish Carassius auratus. Aquat Toxicol 120:119–125
Kong XH, Qiao D, Zhao XL, Wang L, Zhang HX (2017) The molecular characterizations of Cu/ZnSOD and MnSOD and its responses of mRNA expression and enzyme activity to Aeromonas hydrophila or lipopolysaccharide challenge in Qihe crucian carp Carassius auratus. Fish Shellfish Immunol 67:429–440
Levine B, Daniel JK (2004) Development by self-digestion: molecular mechanisms and biological functions of autophagy. Dev Cell 6(4):463–477. https://doi.org/10.1016/S1534-5807(04)00099-1
Liang XH, Jackson S, Seaman M, Brown K, Kempkes B, Hibshoosh H, Levine B (1999) Induction of autophagy and inhibition of tumorigenesis by beclin 1. [J]. Nature 402:672–676. https://doi.org/10.1038/45257
Liu T, Wang P, Lu Y, Zhou G, Diao J, Zhou Z (2012) Enantioselective bioaccumulation of soil-associated fipronil enantiomers in tubifex tubifex. J Hazard Mater 219-220(6):50–56
Ma J, Zhou C, Li Y, Li X (2014) Biochemical responses to the toxicity of the biocide abamectin on the freshwater snail Physa acuta. Ecotoxicol Environ Saf 101:31–35. https://doi.org/10.1016/j.ecoenv.2013.12.009
Ma J, Yuanyuan L, Wu M, Can Z, Yuqing C, Weiguo L, Xiaoyu (2018) Serum immune responses in common carp (Cyprinus carpio L.) to paraquat exposure: the traditional parameters and circulating microRNAs. Fish Shellfish Immunol 76:133–142. https://doi.org/10.1016/j.fsi.2018.02.046
Mathieu P, Leeanna EH, Arnim P (2018) mTOR pathways in cancer and autophagy. Cancers 10(1):18. https://doi.org/10.3390/cancers10010018
Mcmahen RL, Strynar MJ, Dagnino S, Herr DW, Moser VC, Garantziotis S, Andersen EM, Freeborn DL, Lm MM, Lindstrom AB (2015) Identification of fipronil metabolites by time-of-flight mass spectrometry for application in a human exposure study. Environ Int 78:16–23
Mizushima N (2007) Autophagy: process and function. Genes Dev 21:2861–2873
Mizushima N (2010) The role of the ATG1/ULK1 complex in autophagy regulation. Curr Opin Cell Biol 22(2):132–139. https://doi.org/10.1016/j.ceb.2009.12.004
Ohsumi Y (2001) Molecular dissection of autophagy: two ubiquitin-like systems. Nat Rev Mol Cell Biol 2:211–216
Pisa LW, Amaral-Rogers V, Belzunces LP, Bonmatin JM, Downs CA, Goulson D, Kreutzweiser DP, Krupke C, Liess M, McField M, Morrissey CA, Noome DA, Settele J, Simon-Delso N, Stark JD, Van der Sluijs JP, Van Dyck H, Wiemers M (2014) Effects of neonicotinoids and fipronil on non-target invertebrates. Environ Sci Pollut Res 22(1):68–102
Planchart A, Mattingly CJ, Allen D, Ceger P, Casey W, Hinton D, Kanungo J, Kullman SW, Tal T, Bondesson M, Burgess SM (2016) Advancing toxicology research using in vivo high throughput toxicology with small fish models. ALTEX 33:435–452
Qi XZ, Xue MY, Yang SB, Zha JW, Zha F (2017) Ammonia exposure alters the expression of immune-related and antioxidant enzymes-related genes and the gut microbial community of crucian carp (Carassius auratus). Fish Shellfish Immunol 70:485–492
Qiao Q, Liang H, Zhang X (2013) Effect of cyanobacteria on immune function of crucian carp (Carassius auratus) via chronic exposure in diet. Chemosphere 90:1167–1176
Rahman K (2007) Studies on free radicals, antioxidants, and co-factors. Clin Interv Aging 2:219
Rathmell JC, Thompson CB (2002) Pathways of apoptosis in lymphocyte development, homeostasis and disease. Cell 109:S97–S107. https://doi.org/10.1016/S0092-8674(02)00704-3
Saurabh S, Sahoo PK (2008) Lysozyme: an important defence molecule of fish innate immune system. Aquac Res 39:223–239
Sengupta S, Peterson TR, Sabatini DM (2010) Regulation of the mTOR complex 1 pathway by nutrients, growth factors, and stress. Mol Cell 40:310–322
Simmons SO, Fan CY, Ramabhadran R (2009) Cellular stress response pathway system as a sentinel ensemble in toxicological screening. Toxicol Sci 111:202–225
Slaninova A, Smutna M, Modra H, Svobodova Z (2009) A review: oxidative stress in fish induced by pesticides. Neuroendocrinol Lett 30:2–12
Tanida I, Ueno T, Kominami E (2004) LC3 conjugation system in mammalian autophagy. Int J Biochem Cell Biol 36(12):2503–2518. https://doi.org/10.1016/j.biocel.2004.05.009
Tanida I, Minematsu-Ikeguchi N, Ueno T, Kominami E (2005) Lysosomal turnover, but not a cellular level, of endogenous LC3 is a marker for autophagy. Autophagy 1:89–91
Tekirdag KA, Korkmaz G, Ozturk DG, Agami R, Gozuacik D (2013) MIR181A regulates starvation- and rapamycin-induced autophagy through targeting of ATG5. Autophagy 9:374–385. https://doi.org/10.4161/auto.23117
Tingle CCD, Rother JA, Dewhurst CF, Lauer S, King WJ (2003) Fipronil: environmental fate, ecotoxicology, and human health concerns. Rev Environ Contam Toxicol 176(1):1–66
Wang WN, Zhou J, Wang P, Tian TT, Zheng Y, Liu Y, Ma JW, Wang AL (2009) Oxidative stress, DNA damage and antioxidant enzyme gene expression in the Pacific white shrimp, Litopenaeus vannamei when exposed to acute pH stress. Comp Biochem Physiol C 150:428–435
Wang X, Xing H, Jiang Y, Wu H, Sun G, Xu Q et al (2013) Accumulation, histopathological effects and response of biochemical markers in the spleens and head kidneys of common carp exposed to atrazine and chlorpyrifos. Food Chem Toxicol 62:148–158
Wang X, Martínez MA, Wu Q, Ares I, Martínez--Larranaga MR, Anadón A, Yuan ZH (2016) Fipronil insecticide toxicology: oxidative stress and metabolism. Crit Rev Toxicol 46(10):876–899 1-24
Wang L, Jinnan L, Wang C'a, Zhigang Z, Luo L, Xue D, Xu Q (2019) Effect of N-carbamoylglutamate supplementation on the growth performance, antioxidant status and immune response of mirror carp (Cyprinus carpio) fed an arginine-deficient diet. Fish Shellfish Immunol 84:280–289
Weidinger A, Kozlov AV (2015) Biological activities of reactive oxygen and nitrogen species: oxidative stress versus signal transduction. Biomolecules 5(2):472–484
Wirawan E, VandeWalle L, Kersse K, Cornelis S, Claerhout S, Vanoverberghe I, Roelandt R, De Rycke R, Verspurten J, Declercq W, Agostinis P, Vanden Berghe T, Lippens S, Vandenabeele P (2010) Caspase-mediated cleavage of Beclin-1 inactivates Beclin-1-induced autophagy and enhances apoptosis by promoting the release of proapoptotic factors from mitochondria. Cell Death Dis 1:e18
Wu J, Lu J, Lu H, Lin Y, Wilson PC (2015) Occurrence and ecological risks from fipronil in aquatic environments located within residential landscapes. Sci Total Environ 518-519:139–147
Wu P, Mo W, Wang Y, Wu Y, Zhang Y, Chen Z, Li N (2019) Effluent containing Rubrivivax gelatinosus promoting the yield, digestion system, disease resistance, mTOR and NF-kB signaling pathway, intestinal microbiota and aquaculture water quality of crucian carp. Fish Shellfish Immunol 94:166–174. https://doi.org/10.1016/j.fsi.2019.08.015
Xia ZQ, Wu SJ (2018) Effects of glutathione on the survival, growth performance and non-specific immunity of white shrimps (Litopenaeus vannamei). Fish Shellfish Immunol 73:141–144
Xia X, Wang P, Wan R, Huo W, Chang Z (2018) Toxic effects of cyhalofop-butyl on embryos of the Yellow River carp (Cyprinus carpio var.): alters embryos hatching, development failure, mortality of embryos, and apoptosis. Environ Sci Pollut Res 25:24305–24315. https://doi.org/10.1007/s11356-018-2489-x
Yang Y, Dong F, Liu X, Xu J, Wu X, Liu W, Zheng Y (2018) Crosstalk of oxidative damage, apoptosis, and autophagy under endoplasmic reticulum (ER) stress involved in thifluzamide-induced liver damage in zebrafish (Danio rerio). Environ Pollut 243:1904–1911. https://doi.org/10.1016/j.envpol.2018.09.041
Yorimitsu T, Klionsky DJ (2005) Autophagy: molecular machinery for self-eating. Cell Death Differ 12(Suppl. 2):1542–1552. https://doi.org/10.1038/sj.cdd.4401765
Zhang Z, Qi L, Cai J, Jie Y, Qiang S, Xu S (2017) Chlorpyrifos exposure in common carp (Cyprinus carpio L.) leads to oxidative stress and immune responses. Fish Shellfish Immunol 67:604–611
Zhang Y, Li S, Li J, Han L, He Q, Wang R, Wang X, Liu K (2018) Developmental toxicity induced by PM2.5 through endoplasmic reticulum stress and autophagy pathway in zebrafish embryos. Chemosphere 197:611–621. https://doi.org/10.1016/j.chemosphere.2018.01.092
Funding
This work was supported by the Henan Provincial Key Scientific and Technological Project in China (no. 192102110195, 152102210081), Henan Normal University Ph.D. Startup Fund (no. qd17143).
Author information
Authors and Affiliations
Contributions
Jianjun Chen: data curation, investigation, writing—review and editing; Nana Liu: data curation, formal analysis, investigation; Baohua Li: validation; Huajie Zhang: formal analysis; Yidi Zhao: investigation, methodology; Xianglin Cao: conceptualization, supervision, funding acquisition.
Corresponding author
Ethics declarations
Ethics approval
The fish were handled according to the guidelines in the China Law for Animal Health Protection and Instructions for Granting Permits for Animal Experimentation for Scientific Purposes (Ethics Approval No. SCXK (YU) 2005–0001).
Consent to participate
All authors have agreed to participate in the work of this manuscript.
Consent for publication
All authors have approved the version submitted for publication.
Competing interests
The authors declare no competing interests.
Additional information
Responsible editor: Bruno Nunes
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Chen, J., Liu, N., Li, B. et al. The effects of fipronil exposure on oxidative stress, non-specific immunity, autophagy, and apoptosis in the common carp. Environ Sci Pollut Res 28, 27799–27810 (2021). https://doi.org/10.1007/s11356-021-12573-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-021-12573-x