Abstract
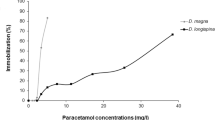
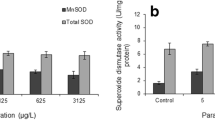
The modern usage of pharmaceutical drugs has led to a progressive increase in their presence and environment concentrations, particularly in the aquatic compartment which is the most common final dumping location for this specific class of chemicals. These substances, due to their chemical and biological properties, can exert mostly uncharacterized toxic effects to non-target aquatic species, given the diverse pathways they activate, and the large number of putative targets in the wild. Among drugs in the environment, paracetamol assumes a leading role, considering its widespread therapeutic use and consequently, environmental presence. The present study aimed to assess the acute and chronic effects of paracetamol, in ecologically relevant levels, in the freshwater cladoceran Daphnia magna, namely focusing on biochemical and reproductive parameters. Considering the pro-oxidant effects of paracetamol, already described for a large set of aquatic organisms, specific enzymes involved in the anti-oxidant and metabolic responses were quantified, namely catalase (CAT) and glutathione S-transferases (GSTs) activities. Cholinesterases (ChEs) activity was quantified to evaluate the capacity of paracetamol to induce neurotoxicity, an indirect outcome of oxidative effects by paracetamol, that may affect feeding behavior and reproductive outcomes of this crustacean. Paracetamol in the tested levels showed no effect on reproductive traits of D. magna. Results obtained for organisms acutely exposed included significant increases in the activities of both GSTs and CAT, demonstrating a short-term pro-oxidative effect by paracetamol. On the contrary, ChEs activity was significantly decreased in organisms exposed to this drug, showing a possible interference with neurotransmission. On the contrary, no noteworthy effects were reported for organisms chronically exposed to ecologically realistic concentrations, evidencing the transient nature of the obtained biological response. These results demonstrate the responsiveness of D. magna to paracetamol, especially for high levels of exposure that, despite not being environmentally relevant, are able to trigger significant antioxidant responses. No population effects were likely to be caused by realistic levels of paracetamol, and the absence of biochemical changes after chronic exposure suggests that this specific organism may not be deleteriously affected by low levels of paracetamol, under real scenarios of contamination.



Similar content being viewed by others
References
Aebi H (1984) Catalase in vitro. Methods Enzymol 6:105–121
Afiyanti M, Chen HJ (2014) Catalase activity is modulated by calcium and calmodulin in detached mature leaves of sweet potato. J Plant Physiol 171(2):35–47. https://doi.org/10.1016/j.jplph.2013.10.003
Allameh A, Alikhani N (2002) Acetaminophen – glutathione conjugate formation in a coupled cytochrome P-450-glutathione S-transferase assay system mediated by subcellular preparations from adult and weanling rat tissues. Toxicol in Vitro 16:637–641
ASTM (1988) Standard practice for conducting acute toxicity tests with fishes, macroinvertebrates and amphibians. Report E 729–80. American Society for Testing and Materials, Philadelphia
Bebianno MJ, Mello ACP, Serrano MAS, Flores-Nunes F, Mattos JJ, Zacchi FL, Piazza CE, Siebert MN, Piazza RS, Gomes CHAM, Melo CMR, Bainy ACD (2017) Transcriptional and cellular effects of paracetamol in the oyster Crassostrea gigas. Ecotoxicol Environ Saf 144(April):258–267. https://doi.org/10.1016/j.ecoenv.2017.06.034
Ben-Shachar R, Chen Y, Luo S, Hartman C, Reed M, Nijhout HF (2012) The biochemistry of acetaminophen hepatotoxicity and rescue: a mathematical model. Theor Biol Med Model 9(1):55. https://doi.org/10.1186/1742-4682-9-55
Bradford M (1976) A rapid and sensitive method for the quantification of microgram quantities of protein utilizing the principle of protein dye binding. Anal Biochem 72:248–254
Brandão FP, Pereira JL, Gonçalves F, Nunes B (2014) The impact of paracetamol on selected biomarkers of the mollusc species Corbicula fluminea. Environ Toxicol 29(1):74–83. https://doi.org/10.1002/tox.20774
Daughton CG (2004) Non-regulated water contaminants: emerging research. Environ Impact Assess Rev 24:711–732. https://doi.org/10.1016/j.eiar.2004.06.003
De Liguoro M, Fioretto B, Poltronieri C, Gallina G (2009) The toxicity of sulfamethazine to Daphnia magna and its additivity to other veterinary sulfonamides and trimethoprim. Chemosphere 75(11):1519–1524. https://doi.org/10.1016/j.chemosphere.2009.02.002
de Oliveira LLD, Antunes SC, Gonçalves F, Rocha O, Nunes B (2015) Evaluation of ecotoxicological effects of drugs on Daphnia magna using different enzymatic biomarkers. Ecotoxicol Environ Saf 119:123–131. https://doi.org/10.1016/j.ecoenv.2015.04.028
Delwing-de Lima D, Wollinger LF, Casagrande ACM, Delwing F, da Cruz JGP, Wyse ATS, Delwing-Dal Magro D (2010) Guanidino compounds inhibit acetylcholinesterase and butyrylcholinesterase activities: effect neuroprotector of vitamins E plus C. Int J Dev Neurosci 28(6):465–473. https://doi.org/10.1016/j.ijdevneu.2010.06.008
Deziel N (2014) Pharmaceuticals in wastewater treatment plant effluent waters. Scholarly Horizons: University of Minnesota, Morris Undergraduate Journal 1(2):12
Diamantino TC, Almeida E, Soares AMVM, Guilhermino L (2003) Characterization of cholinesterases from Daphnia magna Straus and their inhibition by zinc. Bull Environ Contam Toxicol 71(2):219–225. https://doi.org/10.1007/s00128-003-0153-7
Du J, Mei CF, Ying GG, Xu MY (2016) Toxicity thresholds for diclofenac, acetaminophen and ibuprofen in the water flea Daphnia magna. Bull Environ Contam Toxicol 97(1):84–90. https://doi.org/10.1007/s00128-016-1806-7
Ellman GL, Courtney KD, Andres V, Featherstone RM (1961) A new and rapid colorimetric determination of acetylcholinesterase activity. Biochem Pharmacol 7(2):88–95. https://doi.org/10.1016/0006-2952(61)90145-9
Fent K, Weston AA, Caminada D (2006) Ecotoxicology of human pharmaceuticals. Aquat Toxicol 76:122–159, 2006
Gómez MJ, Petrović M, Fernández-Alba AR, Barceló D (2006) Determination of pharmaceuticals of various therapeutic classes by solid-phase extraction and liquid chromatography-tandem mass spectrometry analysis in hospital effluent wastewaters. J Chromatogr A 1114(2):224–233. https://doi.org/10.1016/j.chroma.2006.02.038
Gracia-Lor E, Sancho JV, Serrano R, Hernández F (2012) Occurrence and removal of pharmaceuticals in wastewater treatment plants at the Spanish Mediterranean area of Valencia. Chemosphere 87(5):453–462. https://doi.org/10.1016/j.chemosphere.2011.12.025
Graham GG, Robins S-A, Bryant KJ, Scott KF (2001) Inhibition of prostaglandin synthesis in intact cells by paracetamol (acetaminophen). Inflammopharmacology 9(1/2):131–142
Groning J, Held C, Garten C, Claubnitzer U, Kaschabek SR, Scholmann M (2007) Transformation of diclofenac by the indigenous microflora of river sediments and identification of a major intermediate. Chemosphere 69:509–516
Habig WH, Pabst MJ, Jakoby WB (1974) Glutathione S-transferases—the first enzymatic step in mercapturic acid formation. J Biol Chem 249:7130–7139
Hadi M, Dragovic S, Van Swelm R, Herpers B, Van De Water B, Russel FGM, Groothuis GMM (2013) AMAP, the alleged non-toxic isomer of acetaminophen, is toxic in rat and human liver. Arch Toxicol 87(1):155–165. https://doi.org/10.1007/s00204-012-0924-1
Harshman LG, Zera AJ (2007) The cost of reproduction: the devil in the details. Trends Ecol Evol 22(2):80–86. https://doi.org/10.1016/j.tree.2006.10.008
Henderson CJ, Wolf CR, Kitteringham N, Powell H, Otto D, Park BK (2000) Increased resistance to acetaminophen hepatotoxicity in mice lacking glutathione S-transferase Pi. Proc Natl Acad Sci U S A 97(23):12741–12745. https://doi.org/10.1073/pnas.220176997
Hinson JA, Roberts DW, James LP (2010) Mechanisms of acetaminophen-induced liver necrosis. Handb Exp Pharmacol 196:369–405. https://doi.org/10.1007/978-3-642-00663-0_12
Howard PH, Muir DCG (2013) Identifying new persistent and bioaccumulative organics among chemicals in commerce. III: byproducts, impurities, and transformation products. Environ Sci Technol 47(10):5259–5266. https://doi.org/10.1021/es4004075
Imhof HK, Rusek J, Thiel M, Wolinska J, Laforsch C (2017) Do microplastic particles affect Daphnia magna at the morphological, life history and molecular level? PLoS One 12(11):1–20. https://doi.org/10.1371/journal.pone.0187590
Józwiak-Bebenista M, Nowak JZ (2014) Paracetamol: mechanism of action, applications and safety concern. Acta Pol Pharm Drug Res 71(1):11–23
Kavitha P, Ramesh R, Bupesh G, Stalin A, Subramanian P (2011) Hepatoprotective activity of Tribulus terrestris extract against acetaminophen-induced toxicity in a freshwater fish (Oreochromis mossambicus). In Vitro Cell Dev Biol Anim 47(10):698–706. https://doi.org/10.1007/s11626-011-9457-9
Kim P, Park Y, Ji K, Seo J, Lee S, Choi K, Kho Y, Park J, Choi K (2012) Effect of chronic exposure to acetaminophen and lincomycin on Japanese medaka (Oryzias latipes) and freshwater cladocerans Daphnia magna and Moina macrocopa, and potential mechanisms of endocrine disruption. Chemosphere 89(1):10–18. https://doi.org/10.1016/j.chemosphere.2012.04.006
Kolpin DW, Furlong ET, Meyer MT, Thurman EM, Zaugg SD, Barber LB, Buxton HT (2002) Pharmaceuticals, hormones and other organic wastewater contaminants in US streams, 1999-2000: a national reconnaissance. Environ Sci Technol 36:1202–1211
Kristensen DM, Lesné L, Le Fol V, Desdoits-Lethimonier C, Dejucq-Rainsford N, Leffers H, Jégou B (2012) Paracetamol (acetaminophen), aspirin (acetylsalicylic acid) and indomethacin are anti-androgenic in the rat foetal testis. Int J Androl 35(3):377–384
Küster A, Adler N (2014) Pharmaceuticals in the environment: scientific evidence of risks and its regulation. Philos Trans R Soc Lond Ser B Biol Sci 369(1656):20130587. https://doi.org/10.1098/rstb.2013.0587
Lapworth DJ, Baran N, Stuart ME, Ward RS (2012) Emerging organic contaminants in groundwater: a review of sources, fate and occurrence. Environ Pollut 163:287–303. https://doi.org/10.1016/j.envpol.2011.12.034
Larson AM, Polson J, Fontana RJ, Davern TJ, Lalani E, Hynan LS, Reisch JS, Schiødt FV, Ostapowicz G, Shakil AO, Lee WM (2005) Acetaminophen-induced acute liver failure: results of a United States multicenter, prospective study. Hepatology 42:13641372–13641372. https://doi.org/10.1002/hep.20948
Lee WM (2008) Acetaminophen-related acute liver failure in the United States. Hepatol Res 38(SUPPL. 1):3–8. https://doi.org/10.1111/j.1872-034X.2008.00419.x
Matus GN, Pereira BVR, Silva-Zacarin ECM, Costa MJ, Cordeiro Alves dos Santos A, Nunes B (2018) Behavior and histopathology as biomarkers for evaluation of the effects of paracetamol and propranolol in the neotropical fish species Phalloceros harpagos. Environ Sci Pollut Res 25(28):28601–28618. https://doi.org/10.1007/s11356-018-2839-8
McJunkin B, Barwick KW, Little WC, Winfield JB (1976) Fatal massive hepatic necrosis following acetaminophen overdose. J Am Med Assoc 236(16):1–2
Mohapatra S, Huang CH, Mukherji S, Padhye LP (2016) Occurrence and fate of pharmaceuticals in WWTPs in India and comparison with a similar study in the United States. Chemosphere 159:526–535. https://doi.org/10.1016/j.chemosphere.2016.06.047
Nikolaou A, Meric S, Fatta D (2007) Occurrence patterns of pharmaceuticals in water and wastewater environments. Anal Bioanal Chem 387(4):1225–1234. https://doi.org/10.1007/s00216-006-1035-8
Nunes B, Antunes SC, Santos J, Martins L, Castro BB (2014) Toxic potential of paracetamol to freshwater organisms: a headache to environmental regulators? Ecotoxicol Environ Saf 107:178–185. https://doi.org/10.1016/j.ecoenv.2014.05.027
Nunes B, Verde MF, Soares AMVM (2015) Biochemical effects of the pharmaceutical drug paracetamol on Anguilla anguilla. Environ Sci Pollut Res 22(15):11574–11584. https://doi.org/10.1007/s11356-015-4329-6
Nunes B, Nunes J, Soares AMVM, Figueira E, Freitas R (2017) Toxicological effects of paracetamol on the clam Ruditapes philippinarum: exposure vs recovery. Aquat Toxicol 192:198–206. https://doi.org/10.1016/j.aquatox.2017.09.015
OECD (2004) Test No. 202: Daphnia sp. Acute Immobilisation Test, OECD guidelines for the testing of chemicals, Section 2. OECD Publishing, Paris. https://doi.org/10.1787/9789264069947-en
OECD (2012) Test No. 211: Daphnia magna reproduction test, OECD guidelines for the testing of chemicals, section 2. OECD Publishing, Paris. https://doi.org/10.1787/9789264185203-en
Parolini M, Binelli A, Cogni D, Provini A (2010) Multi-biomarker approach for the evaluation of the cyto-genotoxicity of paracetamol on the zebra mussel (Dreissena polymorpha). Chemosphere 79(5):489–498. https://doi.org/10.1016/j.chemosphere.2010.02.053
Pereira BVR, Matus GN, Costa MJ, Santos ACAD, Silva-Zacarin ECM, do Carmo JB, Nunes B (2018) Assessment of biochemical alterations in the neotropical fish species Phalloceros harpagos after acute and chronic exposure to the drugs paracetamol and propranolol. Environ Sci Pollut Res 25(15):14899–14910. https://doi.org/10.1007/s11356-018-1699-6
Ramos AS, Correia AT, Antunes SC, Gonçalves F, Nunes B (2014) Effect of acetaminophen exposure in Oncorhynchus mykiss gills and liver: detoxification mechanisms, oxidative defence system and peroxidative damage. Environ Toxicol Pharmacol 37(3):1221–1228. https://doi.org/10.1016/j.etap.2014.04.005
Raucy JL, Lasker JM, Lieber CS, Black M (1989) Acetaminophen activation by human liver cytochromes P450IIE1 and P450IA2. Arch Biochem Biophys 271(1):270–283. https://doi.org/10.1016/0003-9861(89)90278-6
Roberts PH, Thomas KV (2006) The occurrence of selected pharmaceuticals in wastewater effluent and surface waters of the lower Tyne catchment. Sci Total Environ 356(1–3):143–153. https://doi.org/10.1016/j.scitotenv.2005.04.031
Saliba SW, Fortwangler E, Ditrich J, Fiebich BL, Marcotegui AR, Perazzo JC, Munoz E, Pinheiro de Oliveira AC (2017) AM404, paracetamol metabolite, prevents prostaglandin synthesis in activated microglia by inhibiting COX activity. J Neuroinflammation 14(1):246
Sims IR, Watson S, Holmes D (1993) Towards a standard Daphnia juvenile production test. Environ Toxicol Chem 12:20532058. https://doi.org/10.1897/15528618(1993)12[2053:TASDJP]2.0.CO;2.
Sui Q, Cao X, Lu S, Zhao W, Qiu Z, Yu G (2015) Occurrence, sources and fate of pharmaceuticals and personal care products in the groundwater: a review. Emerg Contam 1(1):14–24. https://doi.org/10.1016/j.emcon.2015.07.001
Toumi H, Boumaiza M, Millet M, Radetski CM, Camara BI, Felten V, Ferard JF (2015) Investigation of differences in sensitivity between 3 strains of Daphnia magna (crustacean Cladocera) exposed to malathion (organophosphorous pesticide). J Environ Sci Health B 50(1):34–44. https://doi.org/10.1080/03601234.2015.965617
Trautwein C, Kümmerer K, Metzger JW (2008) Aerobic biodegradability of the calcium channel antagonist verapamil and identification of a microbial dead-end transformation product studied by LC-MS/MS. Chemosphere 72(3):442–450. https://doi.org/10.1016/j.chemosphere.2008.02.022
Veiga-Gómez M, Nebot C, Franco CM, Miranda JM, Vázquez B, Cepeda A (2017) Identification and quantification of 12 pharmaceuticals in water collected from milking parlors: food safety implications. J Dairy Sci 100(5):3373–3383. https://doi.org/10.3168/jds.2016-12227
Verlicchi P, Al Aukidy M, Zambello E (2015) What have we learned from worldwide experiences on the management and treatment of hospital effluent? - an overview and a discussion on perspectives. Sci Total Environ 514:467–491. https://doi.org/10.1016/j.scitotenv.2015.02.020
Weiner L, Kreimer D, Roth E, Silman I (1994) Oxidative stress transforms acetylcholinesterase to a molten-globule-like state. Biochem Biophys Res Commun 198(3):915–922
Wu HY, Xu HX, Hong YB, Zhang JF, Wu JC (2011) The use of biomarkers in the antioxidant responses of Daphnia magna to the acute and chronic exposure to no. 20 diesel oil and 2,4-dichlorophenol. Chem Speciat Bioavailab 23(2):80–87. https://doi.org/10.3184/095422911X13027015402949
Xu JJ, Hendriks BS, Zhao J, de Graaf D (2008) Multiple effects of acetaminophen and p38 inhibitors: towards pathway toxicology. FEBS Lett 582(8):1276–1282. https://doi.org/10.1016/j.febslet.2008.01.063
Yalkowsky SH, He Y, Jain P (2010) Handbook of aqueous solubility data, second edn. CRC Press, Boca Raton, p 492
Zupanc M, Kosjek T, Petkovšek M, Dular M, Kompare B, Širok B, Heath E (2013) Removal of pharmaceuticals from wastewater by biological processes, hydrodynamic cavitation and UV treatment. Ultrason Sonochem 20(4):1104–1112. https://doi.org/10.1016/j.ultsonch.2012.12.003
Acknowledgments
Bruno Nunes was hired through the Investigator FCT program (IF/01744/2013).
Funding
This work was financially supported by CESAM (UID/AMB/50017), and by FCT/MEC through national funds, and was co-funded by the FEDER, within the PT2020 Partnership Agreement and Compete 2020.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Cinta Porte
Rights and permissions
About this article
Cite this article
Daniel, D., Dionísio, R., de Alkimin, G.D. et al. Acute and chronic effects of paracetamol exposure on Daphnia magna: how oxidative effects may modulate responses at distinct levels of organization in a model species. Environ Sci Pollut Res 26, 3320–3329 (2019). https://doi.org/10.1007/s11356-018-3788-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-3788-y