Abstract
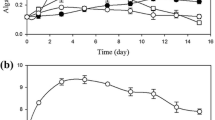
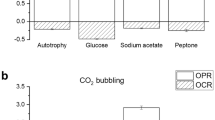
Microalgae-bacteria consortia application to wastewater treatment is considered as a potential and cheap strategy towards a self-sustaining oxygen-carbon dioxide gas exchange. However, microalgae can also carry out mixotrophy, thus reducing the net oxygen production, due to consumption of organic substrates. In this work, respirometric tests were used to quantify the oxygen reduction in the presence of biodegradable COD (chemical oxygen demand), which resulted up to 70%, depending on the biodegradability of the carbon substrate. The implication of mixotrophic metabolism on nutrient removal in urban wastewater was also measured by co-cultivating C. protothecoides with bacteria from activated sludge. To better understand the contribution of different populations, ad hoc experiments under controlled conditions were designed to quantify the nutrient consumption of bacteria and microalgae. Microalgae and bacteria were cultivated together and separately, with and without external bubbling, so to better ascertain the specific role of gas production and nutrient removal. Results showed that microalgae can remove up to 100 and 85% of P and N respectively, but the contribution on COD consumption may affect the net O2 supply to heterotrophic bacteria. However, a mutual COD consumption by microalgae and bacteria was proved by both experimental growth curves and mass balance application, based on stoichiometry experimentally adjusted.




Similar content being viewed by others
References
Abinandan S, Shanthakumar S (2015) Challenges and opportunities in application of microalgae (Chlorophyta) for wastewater treatment: a review. Renew Sust Energ Rev 52:123–132. https://doi.org/10.1016/j.rser.2015.07.086
Alcántara C, Fernández C, García-Encina PA, Muñoz R (2015) Mixotrophic metabolism of Chlorella sorokiniana and algal-bacterial consortia under extended dark-light periods and nutrient starvation. Appl Microbiol Biotechnol 99:2393–2404. https://doi.org/10.1007/s00253-014-6125-5
Barros A, Guerra LT, Simoes M et al (2017) Mass balance analysis of carbon and nitrogen in industrial scale mixotrophic microalgae cultures. Algal Res 21:35–41. https://doi.org/10.1016/j.algal.2016.10.014
de-Bashan LE, Bashan Y (2010) Immobilized microalgae for removing pollutants: review of practical aspects. Bioresour Technol 101:1611–1627. https://doi.org/10.1016/j.biortech.2009.09.043
Bertucco A, Beraldi M, Sforza E (2014) Continuous microalgal cultivation in a laboratory-scale photobioreactor under seasonal day-night irradiation: experiments and simulation. Bioprocess Biosyst Eng 37:1535–1542. https://doi.org/10.1007/s00449-014-1125-5
Boelee NC, Temmink H, Janssen M, Buisman CJN, Wijffels RH (2011) Nitrogen and phosphorus removal from municipal wastewater effluent using microalgal biofilms. Water Res 45:5925–5933. https://doi.org/10.1016/j.watres.2011.08.044
Boelee NC, Temmink H, Janssen M, Buisman CJN, Wijffels RH (2014) Balancing the organic load and light supply in symbiotic microalgal-bacterial biofilm reactors treating synthetic municipal wastewater. Ecol Eng 64:213–221. https://doi.org/10.1016/j.ecoleng.2013.12.035
Cho S, Luong TT, Lee D, Oh YK, Lee T (2011) Reuse of effluent water from a municipal wastewater treatment plant in microalgae cultivation for biofuel production. Bioresour Technol 102:8639–8645. https://doi.org/10.1016/j.biortech.2011.03.037
Chojnacka K, Marquez-Rocha F-J (2004) Kinetic and stoichiometric relationships of the energy and carbon metabolism in the culture of microalgae. Biotechnology 3:21–34
De-Bashan LE, Moreno M, Hernandez JP, Bashan Y (2002) Removal of ammonium and phosphorus ions from synthetic wastewater by the microalgae Chlorella vulgaris coimmobilized in alginate beads with the microalgae growth-promoting bacterium Azospirillum brasilense. Water Res 36:2941–2948. https://doi.org/10.1016/S0043-1354(01)00522-X
Decostere B, Janssens N, Alvarado A, Maere T, Goethals P, van Hulle SWH, Nopens I (2013) A combined respirometer-titrimeter for the determination of microalgae kinetics: experimental data collection and modelling. Chem Eng J 222:85–93. https://doi.org/10.1016/j.cej.2013.01.103
Fukami K, Nishijima T, Ishida Y (1997) Stimulative and inhibitory effects of bacteria on the growth of microalgae. Hydrobiologia 358:185–191. https://doi.org/10.1023/A:1003139402315
Gupta PL, Choi HJ, Pawar RR, Jung SP, Lee SM (2016) Enhanced biomass production through optimization of carbon source and utilization of wastewater as a nutrient source. J Environ Manag 184:585–595. https://doi.org/10.1016/j.jenvman.2016.10.018
Gutzeit G, Lorch D, Weber A et al (2015) Bioflocculent algal-bacterial biomass improves low-cost wastewater treatment. Water Sci Technol 52:9–18
He PJ, Mao B, Lu F et al (2013) The combined effect of bacteria and Chlorella vulgaris on the treatment of municipal wastewaters. Bioresour Technol 146:562–568. https://doi.org/10.1016/j.biortech.2013.07.111
Karya NGAI, van der Steen NP, Lens PNL (2013) Photo-oxygenation to support nitrification in an algal-bacterial consortium treating artificial wastewater. Bioresour Technol 134:244–250. https://doi.org/10.1016/j.biortech.2013.02.005
Kouzuma A, Watanabe K (2015) Exploring the potential of algae/bacteria interactions. Curr Opin Biotechnol 33:125–129. https://doi.org/10.1016/j.copbio.2015.02.007
Le Chevanton M, Garnier M, Bougaran G et al (2013) Screening and selection of growth-promoting bacteria for Dunaliella cultures. Algal Res 2:212–222. https://doi.org/10.1016/j.algal.2013.05.003
Liu L, Zhao Y, Jiang X, Wang X, Liang W (2018) Lipid accumulation of Chlorella pyrenoidosa under mixotrophic cultivation using acetate and ammonium. Bioresour Technol 262:342–346. https://doi.org/10.1016/j.biortech.2018.04.092
Longo S, d’Antoni BM, Bongards M, Chaparro A, Cronrath A, Fatone F, Lema JM, Mauricio-Iglesias M, Soares A, Hospido A (2016) Monitoring and diagnosis of energy consumption in wastewater treatment plants. A state of the art and proposals for improvement. Appl Energy 179:1251–1268. https://doi.org/10.1016/j.apenergy.2016.07.043
Markou G, Vandamme D, Muylaert K (2014) Microalgal and cyanobacterial cultivation: the supply of nutrients. Water Res 65:186–202. https://doi.org/10.1016/j.watres.2014.07.025
Metcalf & Eddy, Tchobanoglous G, Stendel HD, et al (2014) Wastewater engineering: treatment and resource recovery, 5th edn
Nur MMA, Buma AGJ (2018) Opportunities and challenges of microalgal cultivation on wastewater, with special focus on palm oil mill effluent and the production of high value compounds. Waste Biomass Valoriz 1–19. doi:https://doi.org/10.1007/s12649-018-0256-3
OECD (2001) http://www.oecd.org/chemicalsafety/testing/oecdguidelinesforthetestingofchemicals.htm
Olguín EJ (2012) Dual purpose microalgae-bacteria-based systems that treat wastewater and produce biodiesel and chemical products within a biorefinery. Biotechnol Adv 30:1031–1046. https://doi.org/10.1016/j.biotechadv.2012.05.001
Ramos Tercero EA, Sforza E, Morandini M, Bertucco A (2013) Cultivation of Chlorella protothecoides with urban wastewater in continuous photobioreactor: biomass productivity and nutrient removal. Appl Biochem Biotechnol 172:1470–1485. https://doi.org/10.1007/s12010-013-0629-9
Rossi S, Bellucci M, Marazzi F, et al (2018) Activity assessment of microalgal-bacterial consortia based on respirometric tests. Water Sci Technol wst2018078. doi:https://doi.org/10.2166/wst.2018.078
Ruiz-Martinez A, Martin Garcia N, Romero I et al (2012) Microalgae cultivation in wastewater: nutrient removal from anaerobic membrane bioreactor effluent. Bioresour Technol 126C:247–253. https://doi.org/10.1016/j.biortech.2012.09.022
Sforza E, Cipriani R, Morosinotto T, Bertucco A, Giacometti GM (2012) Excess CO2 supply inhibits mixotrophic growth of Chlorella protothecoides and Nannochloropsis salina. Bioresour Technol 104:523–529. https://doi.org/10.1016/j.biortech.2011.10.025
Sforza E, Urbani S, Bertucco A (2015) Evaluation of maintenance energy requirements in the cultivation of Scenedesmus obliquus: effect of light intensity and regime. J Appl Phycol 27:1453–1462. https://doi.org/10.1007/s10811-014-0460-x
Sforza E, Barbera E, Girotto F, Cossu R, Bertucco A (2017) Anaerobic digestion of lipid-extracted microalgae: enhancing nutrient recovery towards a closed loop recycling. Biochem Eng J 121:139–146. https://doi.org/10.1016/j.bej.2017.02.004
Su Y, Mennerich A, Urban B (2012) Synergistic cooperation between wastewater-born algae and activated sludge for wastewater treatment: influence of algae and sludge inoculation ratios. Bioresour Technol 105:67–73. https://doi.org/10.1016/j.biortech.2011.11.113
Subashchandrabose SR, Ramakrishnan B, Megharaj M, Venkateswarlu K, Naidu R (2011) Consortia of cyanobacteria/microalgae and bacteria: biotechnological potential. Biotechnol Adv 29:896–907. https://doi.org/10.1016/j.biotechadv.2011.07.009
Torabizadeh H (2011) All proteins have a basic molecular formula. Int J Chem Mol Nucl Mater Metall Eng 5:501–505
Unnithan VV, Unc A, Smith GB (2014) Mini-review: a priori considerations for bacteria-algae interactions in algal biofuel systems receiving municipal wastewaters. Algal Res 4:35–40. https://doi.org/10.1016/j.algal.2013.11.009
Acknowledgements
The authors would like to thank Montebello Vicentino (VI, Italy) treatment plants for providing the wastewaters and activated sludge inoculum for the experiments. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Gerald Thouand
Electronic supplementary material
ESM 1
(DOCX 145 kb)
Rights and permissions
About this article
Cite this article
Sforza, E., Pastore, M., Spagni, A. et al. Microalgae-bacteria gas exchange in wastewater: how mixotrophy may reduce the oxygen supply for bacteria. Environ Sci Pollut Res 25, 28004–28014 (2018). https://doi.org/10.1007/s11356-018-2834-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-2834-0