Abstract
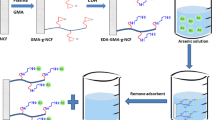
A novel quaternary ammonium polyethylene nonwoven fabric for removing chromium ions from water was prepared via radiation-induced grafting of glycidyl methacrylate and further modification with N,N′-dimethylethylenediamine. The structural and morphological characteristics of the adsorbent were analyzed using Fourier transform infrared spectroscopy (FTIR), thermogravimetry and differential thermogravimetry (TG/DTG), scanning electron microscopy (SEM), and X-ray photoelectron spectroscopy (XPS). The influences of several principal factors, including pH value, initial Cr(VI) concentration, contact time, and coexisting anions (including SO42−, CO32−, NO3−, PO43−, and Cl−), on adsorption performance were investigated via batch tests. The results showed that the optimum removal efficiency was 99.2% at pH 3 and the maximum adsorption quantity for Cr(VI) at 25 °C was 336 mg/g. The adsorption kinetic parameters were better fitted with the pseudo-second-order kinetic model, and the equilibrium data were described very well by the Freundlich isotherm model. Furthermore, the as-synthesized adsorbent exhibited excellent regeneration and recyclability while maintaining high adsorption performance after five adsorption/desorption cycles.











Similar content being viewed by others
References
Agrawal A, Pal C, Sahu KK (2008) Extractive removal of chromium (VI) from industrial waste solution. J Hazard Mater 159:458–464
Aliabadi M, Irani M, Ismaeili J, Piri H, Parnian MJ (2013) Electrospun nanofiber membrane of PEO/chitosan for the adsorption of nickel, cadmium, lead and copper ions from aqueous solution. Chem Eng J 220:237–243
Altın I, Sökmen M (2014) Preparation of TiO 2-polystyrene photocatalyst from waste material and its usability for removal of various pollutants. Appl Catal B 144:694–701. https://doi.org/10.1016/j.apcatb.2013.06.014
Ansari MO, Kumar R, Ansari SA, Ansari SP, Barakat MA, Alshahrie A, Cho MH (2017) Anion selective pTSA doped polyaniline@ graphene oxide-multiwalled carbon nanotube composite for Cr (VI) and Congo red adsorption. J Colloid Interface Sci 496:407–415. https://doi.org/10.1016/j.jcis.2017.02.034
Ashrafi A, Rahbar-Kelishami A, Shayesteh H (2017) Highly efficient simultaneous ultrasonic assisted adsorption of Pb (II) by Fe3O4@ MnO2 core-shell magnetic nanoparticles: synthesis and characterization, kinetic, equilibrium, and thermodynamic studies. J Mol Struct 1147:40–47
Asl SMH, Masomi M, Hosseini M, Javadian H, Ruiz M, Sastre AM (2017) Synthesis of hydrous iron oxide/aluminum hydroxide composite loaded on coal fly ash as an effective mesoporous and low-cost sorbent for Cr (VI) sorption: fuzzy logic modeling. Process Saf Environ Prot 107:153–167
Azizian S (2004) Kinetic models of sorption: a theoretical analysis. J Colloid Interface Sci 276(1):47–52. https://doi.org/10.1016/j.jcis.2004.03.048
Bayat B (2002) Comparative study of adsorption properties of Turkish fly ashes. II. The case of chromium(VI) and cadmium(II). J Hazard Mater 95(3):275–290
Bayramoğlu G, Arıca MY (2005) Ethylenediamine grafted poly (glycidylmethacrylate-co-methylmethacrylate) adsorbent for removal of chromate anions. Sep Purif Technol 45(3):192–199. https://doi.org/10.1016/j.seppur.2005.03.009
Chen JH, Hsu KC, Chang YM (2013) Surface modification of hydrophobic resin with tricaprylmethylammonium chloride for the removal of trace hexavalent chromium. Ind Eng Chem Res 52(33):11685–11694. https://doi.org/10.1021/ie401233r
Chi F, Wang X, Xiong J, Hu S (2013) Polyvinyl alcohol fibers with functional phosphonic acid group: synthesis and adsorption of uranyl (VI) ions in aqueous solutions. J Radioanal Nucl Chem 296(3):1331–1340. https://doi.org/10.1007/s10967-012-2303-z
Flores-Rojas GG, Bucio E (2016) Radiation-grafting of ethylene glycol dimethacrylate (EGDMA) and glycidyl methacrylate (GMA) onto silicone rubber. Radiat Phys Chem 127:21–26. https://doi.org/10.1016/j.radphyschem.2016.05.015
Gao Q, Hua J, Li R, Xing Z, Pang L, Zhang M, Xu L, Wu G (2017) Radiation-induced graft polymerization for the preparation of a highly efficient UHMWPE fibrous adsorbent for Cr (VI) removal. Radiat Phys Chem 130:92–102. https://doi.org/10.1016/j.radphyschem.2016.08.004
Golbaz S, Jafari AJ, Rafiee M, Kalantary RR (2014) Separate and simultaneous removal of phenol, chromium, and cyanide from aqueous solution by coagulation/precipitation: mechanisms and theory. Chem Eng J 253:251–257. https://doi.org/10.1016/j.cej.2014.05.074
Gupta VK, Kumar R, Nayak A, Saleh TA, Barakat MA (2013) Adsorptive removal of dyes from aqueous solution onto carbon nanotubes: a review. Adv Colloid Interf Sci 193:24–34
Hlihor RM, Figueiredo H, Tavares T, Gavrilescu M (2017) Biosorption potential of dead and living Arthrobacter viscosus biomass in the removal of Cr (VI): batch and column studies. Process Saf Environ Prot 108:44–56. https://doi.org/10.1016/j.psep.2016.06.016
Huang SH, Chen DH (2009) Rapid removal of heavy metal cations and anions from aqueous solutions by an amino-functionalized magnetic nano-adsorbent. J Hazard Mater 163(1):174–179. https://doi.org/10.1016/j.jhazmat.2008.06.075
Javadian H (2014) Adsorption performance of suitable nanostructured novel composite adsorbent of poly (N-methylaniline) for removal of heavy metal from aqueous solutions. J Ind Eng Chem 20(6):4344–4352. https://doi.org/10.1016/j.jiec.2014.01.042
Jiang TJ, Yang M, Li SS, Ma MJ, Zhao NJ, Guo Z, Liu JH, Huang XJ (2017) In situ underwater laser-induced breakdown spectroscopy analysis for trace Cr (VI) in aqueous solution supported by electrosorption enrichment and a gas-assisted localized liquid discharge apparatus. Anal Chem 89(10):5557–5564. https://doi.org/10.1021/acs.analchem.7b00629
Jin H, An Q, Zhao Q, Qian J, Zhu M (2010) Pervaporation dehydration of ethanol by using polyelectrolyte complex membranes based on poly (N-ethyl-4-vinylpyridinium bromide) and sodium carboxymethyl cellulose. J Membr Sci 347:183–192
Kavaklı C, Barsbay M, Tilki S, Güven O, Kavaklı PA (2016) Activation of polyethylene/polypropylene nonwoven fabric by radiation-induced grafting for the removal of Cr (VI) from aqueous solutions. Water Air Soil Pollut 227(12):473. https://doi.org/10.1007/s11270-016-3184-5
Ke Z, Huang Q, Zhang H, Yu Z (2011) Reduction and removal of aqueous Cr (VI) by glow discharge plasma at the gas–solution interface. Environ Sci Technol 45(18):7841–7847. https://doi.org/10.1021/es201680m
Kebir M, Chabani M, Nasrallah N, Bensmaili A, Trari M (2011) Coupling adsorption with photocatalysis process for the Cr (VI) removal. Desalination 270:166–173
Kong Z, Wu X, Wei J, Zhang H, Cui L (2016) Preparation and characterization of hydrophilicity fibers based on 2-(dimethyamino) ethyl mathacrylate grafted polypropylene by UV-irradiation for removal of Cr(VI) and As(V). J Polym Res 23(9):199. https://doi.org/10.1007/s10965-016-1079-1
Kumar R, Rashid J, Barakat MA (2014) Synthesis and characterization of a starch–AlOOH–FeS 2 nanocomposite for the adsorption of congo red dye from aqueous solution. RSC Adv 4(72):38334–38340. https://doi.org/10.1039/C4RA05183A
Kuppusamy S, Thavamani P, Megharaj M, Venkateswarlu K, Lee YB, Naidu R (2016) Potential of Melaleuca diosmifolia leaf as a low-cost adsorbent for hexavalent chromium removal from contaminated water bodies. Process Saf Environ Prot 100:173–182. https://doi.org/10.1016/j.psep.2016.01.009
Lee SM, Tiwari D (2016) Synthesis of functionalized biomaterials and its application in the efficient remediation of aquatic environment contaminated with Cr (VI). Chem Eng J 296:35–44. https://doi.org/10.1016/j.cej.2016.03.077
Lei C, Zhu X, Zhu B, Jiang C, Le Y, Yu J (2017) Superb adsorption capacity of hierarchical calcined Ni/Mg/Al layered double hydroxides for Congo red and Cr (VI) ions. J Hazard Mater 321:801–811. https://doi.org/10.1016/j.jhazmat.2016.09.070
Li J, Lin Q, Zhang X (2010) Mechanism of electron transfer in the bioadsorption of hexavalent chromium within Leersia hexandra Swartz granules by X-ray photoelectron spectroscopy. J Hazard Mater 182:598–602
Li Y, Gao B, Wu T, Sun D, Li X, Wang B, Lu F (2009) Hexavalent chromium removal from aqueous solution by adsorption on aluminum magnesium mixed hydroxide. Water Res 43(12):3067–3075. https://doi.org/10.1016/j.watres.2009.04.008
Li YS, Qin JT, Han Y, Du JF, Dong ZB, Sun SF, Liu Y (2017) Controlled preparation and highly photocatalytic activity of portable MCC-g-GMA@ TiO2 photocatalyst by pre-radiation grafting-embedding method. Appl Catal B 218:101–110. https://doi.org/10.1016/j.apcatb.2017.03.083
Liu C, Jin RN, Ouyang XK, Wang YG (2017) Adsorption behavior of carboxylated cellulose nanocrystal–polyethyleneimine composite for removal of Cr (VI) ions. Appl Surf Sci 408:77–87. https://doi.org/10.1016/j.apsusc.2017.02.265
Luo X, Zeng J, Liu S, Zhang L (2015) An effective and recyclable adsorbent for the removal of heavy metal ions from aqueous system: magnetic chitosan/cellulose microspheres. Bioresour Technol 194:403–406. https://doi.org/10.1016/j.biortech.2015.07.044
Mittal A, Mittal J, Malviya A, Gupta VK (2009) Adsorptive removal of hazardous anionic dye “Congo red” from wastewater using waste materials and recovery by desorption. J Colloid Interface Sci 340(1):16–26. https://doi.org/10.1016/j.jcis.2009.08.019
Módenes AN, de Oliveira AP, Espinoza-Quiñones FR, Trigueros DEG, Kroumov AD, Bergamasco R (2017) Study of the involved sorption mechanisms of Cr (VI) and Cr (III) species onto dried Salvinia auriculata biomass. Chemosphere 172:373–383. https://doi.org/10.1016/j.chemosphere.2017.01.038
Nayak V, Jyothi MS, Balakrishna RG, Padaki M, Deon S (2017) Novel modified poly vinyl chloride blend membranes for removal of heavy metals from mixed ion feed sample. J Hazard Mater 331:289–299. https://doi.org/10.1016/j.jhazmat.2017.02.046
Owens DK, Wendt RC (1969) Estimation of the surface free energy of polymers. J Appl Polym Sci 13(8):1741–1747. https://doi.org/10.1002/app.1969.070130815
Park D, Yun YS, Park JM (2008) XAS and XPS studies on chromium-binding groups of biomaterial during Cr (VI) biosorption. J Colloid Interface Sci 317(1):54–61. https://doi.org/10.1016/j.jcis.2007.09.049
Ren G, Wang X, Huang P, Zhong B, Zhang Z, Yang L, Yang X (2017) Chromium (VI) adsorption from wastewater using porous magnetite nanoparticles prepared from titanium residue by a novel solid-phase reduction method. Sci Total Environ 607:900–910
Rosales E, Meijide J, Tavares T, Pazos M, Sanromán MA (2016) Grapefruit peelings as a promising biosorbent for the removal of leather dyes and hexavalent chromium. Process Saf Environ Prot 101:61–71
Shaikh SH, Kumar SA (2017) Polyhydroxamic acid functionalized sorbent for effective removal of chromium from ground water and chromic acid cleaning bath. Chem Eng J 326:318–328. https://doi.org/10.1016/j.cej.2017.05.151
Stollenwerk KG, Grove DB (1985) Adsorption and desorption of hexavalent chromium in an alluvial aquifer near Telluride, Colorado. J Environ Qual 14(1):150–155. https://doi.org/10.2134/jeq1985.00472425001400010030x
Sun X, Yang L, Xing H, Zhao J, Li X, Huang Y, Liu H (2013) Synthesis of polyethylenimine-functionalized poly (glycidyl methacrylate) magnetic microspheres and their excellent Cr (VI) ion removal properties. Chem Eng J 234:338–345
Tan W, Li Q, Wang H, Liu Y, Zhang J, Dong F, Guo Z (2016) Synthesis, characterization, and antibacterial property of novel starch derivatives with 1, 2, 3-triazole. Carbohydr Polym 142:1–7
Theapsak S, Watthanaphanit A, Rujiravanit R (2012) Preparation of chitosan-coated polyethylene packaging films by DBD plasma treatment. ACS Appl Mater Interfaces 4:2474–2482
Wang J, Zhao L, Duan W, Han L, Chen Y (2012) Adsorption of aqueous Cr (VI) by novel fibrous adsorbent with amino and quaternary ammonium groups. Ind Eng Chem Res 51:13655–13662
Wang W, Zhou J, Achari G, Yu J, Cai W (2014) Cr (VI) removal from aqueous solutions by hydrothermal synthetic layered double hydroxides: adsorption performance, coexisting anions and regeneration studies. Colloids Surf A Physicochem Eng Asp 457:33–40. https://doi.org/10.1016/j.colsurfa.2014.05.034
Wu J, Wang XB, Zeng RJ (2017) Reactivity enhancement of iron sulfide nanoparticles stabilized by sodium alginate: taking Cr (VI) removal as an example. J Hazard Mater 333:275–284. https://doi.org/10.1016/j.jhazmat.2017.03.023
Yang R, Aubrecht KB, Ma H, Wang R, Grubbs RB, Hsiao BS, Chu B (2014) Thiol-modified cellulose nanofibrous composite membranes for chromium (VI) and lead (II) adsorption. Polymer 55(5):1167–1176. https://doi.org/10.1016/j.polymer.2014.01.043
Zhang H, Zhang F, Huang Q (2017) Highly effective removal of malachite green from aqueous solution by hydrochar derived from phycocyanin-extracted algal bloom residues through hydrothermal carbonization. RSC Adv 7:5790–5799
Zhang J, Shen H, Wang X, Wu J, Xue Y (2004) Effects of chronic exposure of 2, 4-dichlorophenol on the antioxidant system in liver of freshwater fish Carassius auratus. Chemosphere 55(2):167–174. https://doi.org/10.1016/j.chemosphere.2003.10.048
Zheng W, An Q, Lei Z, Xiao Z, Zhai S, Liu Q (2016) Efficient batch and column removal of Cr (VI) by carbon beads with developed nano-network. RSC Adv 6(106):104897–104910. https://doi.org/10.1039/C6RA14070J
Zu J, Liu R, Zhang J, Tang F, He L (2016) Adsorption of Re and 99Tc by means of radiation-grafted weak basic anion exchange resin. J Radioanal Nucl Chem 310(1):229–237. https://doi.org/10.1007/s10967-016-4819-0
Funding
This work was supported by the National Natural Science Foundation of China (grant numbers 11675247 and Y715191031).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Guilherme L. Dotto
Rights and permissions
About this article
Cite this article
Pang, Lj., Hu, Jt., Zhang, Mj. et al. An efficient and reusable quaternary ammonium fabric adsorbent prepared by radiation grafting for removal of Cr(VI) from wastewater. Environ Sci Pollut Res 25, 11045–11053 (2018). https://doi.org/10.1007/s11356-018-1355-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1355-1