Abstract
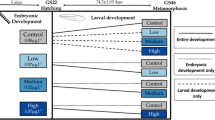
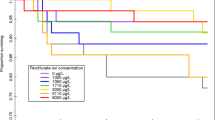
Antifouling chemicals have a long history of causing toxicity to aquatic organisms. We measured growth and developmental timing in wood frog tadpoles exposed to the antifouling chemical medetomidine (10 nM–10 μM) starting at two different developmental stages in static renewal experiments. For tadpoles hatched from egg masses and exposed for 3 weeks to 100 nM and 1 μM, head width/total body length ratio was significantly shorter compared to control. For field-collected tadpoles at Gosner stage 24–25 and exposed for 2 weeks, 1 and 10 μM medetomidine significantly slowed development as measured by Gosner stage. Medetomidine (1 and 10 μM) significantly increased the time to metamorphosis by over 16 days on average, and at 100 nM and 1 μM, it significantly decreased mass at metamorphosis. We discuss the possible effects of antifouling chemicals containing medetomidine on globally threatened groups such as amphibians.




Similar content being viewed by others
References
Ahren B (1985) Effects of alpha-adrenoceptor agonists and antagonists on thyroid hormone secretion. Acta Endocrinol 108(2):184–191
Barry MJ (2014) Fluoxetine inhibits predator avoidance behavior in tadpoles. Toxicol Environ Chem 96(4):641–649. https://doi.org/10.1080/02772248.2014.966713
Bellas J, Hilvarsson A, Birgersson G, Granmo A (2006) Effects of medetomidine, a novel antifouling agent, on the burrowing bivalve Abra nitida (Muller). Chemosphere 65(4):575–582. https://doi.org/10.1016/j.chemosphere.2006.02.045
Benard MF (2015) Warmer winters reduce frog fecundity and shift breeding phenology, which consequently alters larval development and metamorphic timing. Glob Chang Biol 21(3):1058–1065. https://doi.org/10.1111/gcb.12720
Berven KA (1982) The genetic basis of altitudinal variation in wood frog Rana sylvatica. I An experimental analysis of life history traits. Evolution 36(5):962–983. https://doi.org/10.1111/j.1558-5646.1982.tb05466.x
Boorse GC, Denver RJ (2004) Endocrine mechanisms underlying plasticity in metamorphic timing in spadefoot toads. Integr Comp Biol 43:646–657
Brown DD, Cai L (2007) Amphibian metamorphosis. Dev Biol 306(1):20–33. https://doi.org/10.1016/j.ydbio.2007.03.021
Bruhl CA, Schmidt T, Pieper S, Alscher A (2013) Terrestrial pesticide exposure of amphibians: an underestimated cause of global decline? Sci Rep 3(1). https://doi.org/10.1038/srep01113
Carew AC, Hoque ME, Metcalfe CD, Peyrot C, Wilkinson KJ, Helbing CC (2015) Chronic sublethal exposure to silver nanoparticles disrupts thyroid hormone signaling during Xenopus laevis metamorphosis. Aquat Toxicol 159:99–108. https://doi.org/10.1016/j.aquatox.2014.12.005
Carey C, Bryant C (1995) Possible interrelations among environmental toxicants, amphibian development, and decline of amphibian populations. Env Health Perspect 103(supplement 4):13–17
Carfagno GLF, Fong PP (2014) Growth inhibition of tadpoles exposed to sertraline in the presence of conspecifics. J Herpetol 48(4):571–576. https://doi.org/10.1670/13-058
Cleuvers M (2003) Aquatic toxicology of pharmaceuticals including the assessment of combination effects. Toxicol Lett 142(3):185–194. https://doi.org/10.1016/S0378-4274(03)00068-7
Collins SJ, Russell RW (2009) Toxicity of road salt to Nova Scotia amphibians. Environ Pollut 157(1):320–324. https://doi.org/10.1016/j.envpol.2008.06.032
Conners DE, Rogers ED, Armbrust KL, Kwon JW, Black MC (2009) Growth and development of tadpoles (Xenopus laevis) exposed to selective serotonin reuptake inhibitors fluoxetine and sertraline, throughout metamorphosis. Environ Toxicol Chem 28(12):2671–2676. https://doi.org/10.1897/08-493.1
European Chemicals Agency (2008) CLH report for medetomidine, proposal for harmonised classification and labeling, UK Competent Authority. Available from: https://echa.europa.eu/documents/10162/13626/clh_proposal_medetomidine_en.pdf
Floyd RH, Wade J, Crain DA (2008) Differential acute sensitivity of wild Rana sylvatica and laboratory Xenopus laevis tadpoles to the herbicide atrazine. Bios 79(3):115–119.
Fong PP, Thompson LB, Carfagno GLF, Sitton AJ (2016) Long-term exposure to gold nanoparticles accelerates larval metamorphosis without affecting mass in wood frogs (Lithobates sylvaticus) at environmentally relevant concentrations. Environ Toxicol Chem 35(9):2304–2310. https://doi.org/10.1002/etc.3396
Foster H, Burton GA, Basu N (2010) Chronic exposure to fluoxetine (Prozac) causes developmental delays in Rana pipiens larvae. Environ Toxicol Chem 29(12):2845–2850. https://doi.org/10.1002/etc.345
Fraker SL, Smith GR (2004) Direct and interactive effects of ecologically relevant concentrations of organic wastewater contaminants on Rana pipiens tadpoles. Environ Toxicol 19(3):250–256. https://doi.org/10.1002/tox.20017
Gipperth L (2009) The legal design of the international and European Union ban on tributyltin antifouling paint: direct and indirect effects. J Environ Manag 90(supple. 1):S86–S95
Gosner KL (1960) A simplified table for staging anuran embryos and larvae with notes on identification. Herpetologica 16(3):183–190
Hayden MT, Reeves MK, Holyoak M, Perdue M, King A, Tobin SC (2015) Thrice as easy to catch! Copper and temperature modulate predator-prey interactions in larval dragonflies and anurans. Ecosphere 6(4):1–17
Hayes TB, Fals P, Gallipeau S, Stice M (2010) The cause of global amphibian declines: a developmental endocrinologist’s perspective. J Exp Biol 213(6):921–933. https://doi.org/10.1242/jeb.040865
Hilvarsson A, Halldorsson HP, Granmo A (2007) Medetomidine as a candidate antifoulant: sublethal effects on juvenile turbot (Psetta maxima L.) Aquat Toxicol 83(3):238–246. https://doi.org/10.1016/j.aquatox.2007.04.008
Hilvarsson A, Ohlauson C, Blank H, Granmo A (2009) Bioaccumulation of the new antifoulant medetomidine in marine organisms. Mar Environ Res 68(1):19–24. https://doi.org/10.1016/j.marenvres.2009.03.007
Horsberg TE, Burka JF, Tasker RAR (1999) Actions and pharmacokinetic properties of the alpha-2-adrenergic agents, medetomidine and atipamezole, in rainbow trout (Oncorhynchus mykiss). Vet Anaesth Analg 26(1):18–22
LaFortune M, Mitchell MA, Smith JA (2001) Evaluation of medetomidine, clove oil and propofol for anesthesia of leopard frogs, Rana pipiens. J Herp Med Surg 11(4):13–18
Lefcort H, Thomson SM, Cowles EE, Harowicz HL, Livaudais BM, Roberts WE, Ettinger WF (1999) Ramifications of predator avoidance: predator and heavy metal-mediated competition between tadpoles and snails. Ecol Appl 9(4):1477–1489.
Lefcort H, Meguire RA, Wilson LH, Ettinger WF (1998) Heavy metals alter the survival, growth, metamorphosis, and antipredatory behavior of Columbia spotted frog (Rana luteiventris) tadpoles. Arch Environ Contam Toxicol 35(3):447–456. https://doi.org/10.1007/s002449900401
Lennquist A, Martensson Lindblad LG, Hedberg D, Kristiansson E, Forlin L (2010a) Colour and melanophore function in rainbow trout after long term exposure to the new antifoulant medetomidine. Chemosphere 80(9):1050–1055. https://doi.org/10.1016/j.chemosphere.2010.05.014
Lennquist A, Hilvarsson A, Forlin L (2010b) Responses in fish exposed to medetomidine, a new antifouling agent. Mar Environ Res 69:S43–S45. https://doi.org/10.1016/j.marenvres.2010.01.004
Lennquist A, Asker N, Kristiansson E, Brenthel A, Bjornsson BT, Kling P, Hultman M, Larsson DG, Forlin L (2011) Physiology and mRNA expression in rainbow trout (Oncorhynchus mykiss) after long-term exposure to the new antifoulant medetomidine. Comp Biochem Physiol C 154(3):234–241
Lind U, Rosenblad MA, Frank LH, Falkbring S, Brive L, Laurila JM, Pohjanoksa K, Vuorenpas A, Kukkonen JP, Gunnarsson L, Scheinin M, Lindblad LGEM, Blomberg A (2010) Octopamine receptors from the barnacle Balanus improvisus are activated by the alpha2-adrenoceptor agonist medetomidine. Mol Pharmacol 78:237–248
Marco A, Quilchano C, Blaustein AR (1999) Sensitivity to nitrate and nitrite in pond-breeding amphibians from the Pacific Northwest, USA. Environ Toxicol Chem 18(12):2836–2839. https://doi.org/10.1002/etc.5620181225
Mohammed A (2013) Why are early life stages of aquatic organisms more sensitive to toxicants than adults? In: Gowder S (ed) New insights into toxicity and drug testing. InTech Publishing, pp 49–62. https://doi.org/10.5772/55187
Nakano D, Strayer DL (2014) Biofouling animals in fresh water: biology, impacts, and ecosystem engineering. Front Ecol Environ 12:167–175
Ohlauson C, Eriksson KM, Blanck H (2012) Short-term effects of medetomidine on photosynthesis and protein synthesis in periphyton, epipsammon and plankton communities in relation to predicted environmental concentrations. Biofouling 28(5):491–499. https://doi.org/10.1080/08927014.2012.687048
Reeves MK, Perdue M, Blakemore GD, Rinella DJ, Holyoak M (2011) Twice as easy to catch? A toxicant and a predator cue cause additive reductions in larval amphibian activity. Ecosphere 2(6):1–20
Relyea RA (2009) A cocktail of contaminants: how mixtures of pesticides at low concentrations affect aquatic communities. Oecologia 159(2):363–376. https://doi.org/10.1007/s00442-008-1213-9
Relyea RA, Jones DK (2009) The toxicity of roundup original max to 13 species of larval amphibians. Environ Toxicol Chem 28(9):2004–2008. https://doi.org/10.1897/09-021.1
Silva LJG, Pereira AMPT, Meisel LM, Lino CM, Pena A (2015) Reviewing the serotonin reuptake inhibitors (SSRIs) footprint in the aquatic biota: uptake, bioaccumulation and ecotoxicology. Environ Pollut 197:127–143. https://doi.org/10.1016/j.envpol.2014.12.002
Stuart S, Chanson J, Cox N, Young B, Rodrigues A, Fischman D, Waller R (2004) Status and trends of amphibian declines and extinctions worldwide. Science 306(5702):1783–1786. https://doi.org/10.1126/science.1103538
Thompson LB, Carfagno GLF, Andresen K, Sitton AJ, Bury T, Lee LL, Lerner KT, Fong PP (2017) Differential uptake of gold nanoparticles by two species of tadpole, the wood frog (Lithobates sylvaticus) and the bullfrog (L. catesbeianus). Env Tox Chem 36(12):3351–3358. https://doi.org/10.1002/etc.3909
Vasconcelos V, Azevedo J, Silva M, Ramos V (2010) Effects of marine toxins on the reproduction and early stages development of aquatic organisms. Marine Drugs 8(1):59–79. https://doi.org/10.3390/md8010059
Weldon C, du Preez LH, Hyatt AD, Muller R, Spears R (2004) Origin of the amphibian chytrid fungus. Emerg Infect Dis 10(12):2100–2105. https://doi.org/10.3201/eid1012.030804
Wendt I, Backhaus T, Blanck H, Arrhenius A (2016) The toxicity of the three antifouling biocides DCOIT, TPBP and medetomidine to the marine pelagic copepod Acartia tonsa. Ecotoxicology 25(5):871–879. https://doi.org/10.1007/s10646-016-1644-8
Wilbur HM, Collins JP (1973) Ecological aspects of amphibian metamorphosis. Science 182(4119):1305–1314. https://doi.org/10.1126/science.182.4119.1305
Willis K, Woods C (2011) Managing invasive Styela clava populations: inhibiting larval recruitment with medetomidine. Aquat Invasions 6(4):511–514. https://doi.org/10.3391/ai.2011.6.4.16
Acknowledgments
We thank K. Hiraizumi for statistical advice and two anonymous reviewers for helpful comments.
Funding
This work was supported in part by a grant to Gettysburg College from the Howard Hughes Medical Institute through the Precollege and Undergraduate Science Education Program.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The collection and use of all animals was approved by the Institutional Animal Care and Use Committee (IACUC) of Gettysburg College.
Additional information
Responsible editor: Philippe Garrigues
Rights and permissions
About this article
Cite this article
Barr, J.M., Palmucci, J.R., Lambert, O.J. et al. Exposure to the antifouling chemical medetomidine slows development, reduces body mass, and delays metamorphosis in wood frog (Lithobates sylvaticus) tadpoles. Environ Sci Pollut Res 25, 10630–10635 (2018). https://doi.org/10.1007/s11356-018-1204-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-018-1204-2