Abstract
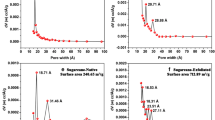
The integrated experimental methods were used to analyze the physicochemical properties and structural characteristics and to build the 2D structural model of two kinds of biochars. Corn straw and poplar leaf biochars were gained by pyrolysing the raw materials slowly in a furnace at 300, 500, and 700 °C under oxygen-deficient conditions. Scanning electron microscope was applied to observe the surface morphology of the biochars. High temperatures destroyed the pore structures of the biochars, forming a particle mixture of varying sizes. The ash content, yield, pH, and surface area were also observed to describe the biochars’ properties. The yield decreases as the pyrolysis temperature increases. The biochars are neutral to alkaline. The biggest surface area is 251.11 m2/g for 700 °C corn straw biochar. Elemental analysis, infrared microspectroscopy, solid-state C-13 NMR spectroscopy, and pyrolysis gas chromatography-mass spectrometry (Py-GC-MS) were also used to study the structural characteristics and build the 2D structural models of biochars. The C content in the corn straw and poplar leaf biochars increases with the increase of the pyrolysis temperature. A higher pyrolysis temperature makes the aryl carbon increase, and C=O, OH, and aliphatic hydrocarbon content decrease in the IR spectra. Solid-state C-13 NMR spectra show that a higher pyrolysis temperature makes the alkyl carbon and alkoxy carbon decrease and the aryl carbon increase. The results of IR microspectra and solid-state C-13 NMR spectra reveal that some noticeable differences exist in these two kinds of biochars and in the same type of biochar but under different pyrolysis temperatures. The conceptual elemental compositions of 500 °C corn straw and poplar leaf biochars are C61H33NO13 and C59H41N3O12, respectively. Significant differences exist in the SEM images, physicochemical properties, and structural characteristics of corn straw and poplar leaf biochars.






Similar content being viewed by others
References
Amir S, Jouraiphy A, Meddich A, Gharous ME, Winterton P, Hafidi M (2010) Structural study of humic acids during composting of activated sludge-green waste: elemental analysis, FTIR and 13C NMR. J Hazard Mater 177(1-3):524–529. https://doi.org/10.1016/j.jhazmat.2009.12.064
Baldock JA, Smernik RJ (2002) Chemical composition and bioavailability of thermally altered Pinus resinosa (red pine) wood. Organic Geochem 33(9):1093–1109. https://doi.org/10.1016/S0146-6380(02)00062-1
Chai XL, Hao YX, Liu GX, Zhao X, Zhao YC (2013) Spectroscopic studies of the effect of aerobic conditions on the chemical characteristics of humic acid in landfill leachate and its implication for the environment. Chemosphere 91:1058–1063
Chen B, Zhou D, Zhu L (2008) Transitional adsorption and partition of nonpolar and polar aromatic contaminants by biochars of pine needles with different pyrolytic temperatures. Environ Sci Technol 42(14):5137–5143. https://doi.org/10.1021/es8002684
Chen XC, Chen GC, Chen LG, Chen YX, Lehmann J, McBride MB, Hay AG (2011) Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour Technol 102(19):8877–8884. https://doi.org/10.1016/j.biortech.2011.06.078
Chen YQ, Yang HP, Wang XH, Zhang SH, Chen HP (2012) Biomass-based pyrolytic polygeneration system on cotton stalk pyrolysis: influence of temperature. Bioresour Technol 107:411–418. https://doi.org/10.1016/j.biortech.2011.10.074
Chun Y, Sheng GY, Chiou CT, Xing BS (2004) Compositions and sorptive properties of crop residue-derived chars. Environ Sci Technol 38(17):4649–4655. https://doi.org/10.1021/es035034w
Cimò G, Kucerik J, Berns AE, Schaumann GE, Alonzo G, Conte P (2014) Effect of heating time and temperature on the chemical characteristics of biochar from poultry manure. J Agr Food Chem 62(8):1912–1918. https://doi.org/10.1021/jf405549z
Conte P, Schmidt HP, Cimò G (2016) Research and application of biochar in Europe. In: Guo M, He Z, Uchimiya M (eds) Agricultural and environmental applications of biochar: advances and barriers. SSSA special publication 63 5585 Guilford Rd, Madison, WI 53711, USA, DOI: https://doi.org/10.2136/sssaspecpub63.2014.0050
De Pasquale C, Marsala V, Berns AE, Valagussa M, Pozzi A, Alonzo G, Conte P (2012) Fast field cycling NMR relaxometry characterization of biochars obtained from an industrial thermochemical process. J Soils Sediment 12(8):1211–1221. https://doi.org/10.1007/s11368-012-0489-x
Demirbas A (2004) Effects of temperature and particle size on bio-char yield from pyrolysis of agricultural residues. J Anal Appl Pyrol 72(2):243–248. https://doi.org/10.1016/j.jaap.2004.07.003
Diallo MS, Simpson A, Gassman PL, Faulon JL, Johnson JH, Goddard WA, Hatcher PG (2003) 3-D structural modeling of humic acids through experimental characterization, computer assisted structure elucidation and atomistic simulations. 1. Chelsea soil humic acid. Environ Sci Technol 37(9):1783–1793. https://doi.org/10.1021/es0259638
Hatfield GR, Maciel GE, Erbatur O, Erbatur G (1987) Qualitative and quantitative analysis of solid lignin samples by carbon-13 nuclear magnetic resonance spectroscopy. Anal Chem 59(1):172–179. https://doi.org/10.1021/ac00128a036
Hossain MK, Strezov V, Chan KY, Ziolkowski A, Nelson PF (2011) Influence of pyrolysis temperature on production and nutrient properties of wastewater sludge biochar. J Environ Manag 92(1):223–228. https://doi.org/10.1016/j.jenvman.2010.09.008
Jiang J, Yuan M, RK X, Bish DL (2015) Mobilization of phosphate in variable-charge soils amended with biochars derived from crop straws. Soil Till Res 146:139–147. https://doi.org/10.1016/j.still.2014.10.009
Jin H, Wang XM, Shen YB, ZG G (2014) A high-performance carbon derived from corn stover via microwave and slow pyrolysis for supercapacitors. J Anal Appl Pyrol 110:18–23. https://doi.org/10.1016/j.jaap.2014.07.010
Kim KH, Kim JY, Cho TS, Choi JW (2012) Influence of pyrolysis temperature on physicochemical properties of biochar obtained from the fast pyrolysis of pitch pine (Pinus rigida). Bioresour Technol 118:158–162. https://doi.org/10.1016/j.biortech.2012.04.094
Lu HL, Zhang WH, Yang YX, Huang XF, Wang SZ, Qiu RL (2012) Relative distribution of Pb2+ sorption mechanisms by sludge-derived biochar. Water Res 46:854–862
Mao JD, Kong XQ, Schmidt-Rohr K, Pignatello JJ, Perdue EM (2012) Advanced solid-state NMR characterization of marine dissolved organic matter isolated using the coupled reverse osmosis/electrodialysis method. Environ Sci Technol 46(11):5806–5814. https://doi.org/10.1021/es300521e
Miao Q, Bi E, Li B (2017) Roles of polar groups and aromatic structures of biochar in 1-methyl-3-octylimidazolium chloride ionic liquid adsorption: pH effect and thermodynamics study. Environ Sci Pollut Res 24(28):22265–22274. https://doi.org/10.1007/s11356-017-9886-4
Mohan D, Sharma R, Singh VK, Steele P, Pittman Jr CU (2012) Fluoride removal from water using bio-char, a green waste, low-cost adsorbent: equilibrium uptake and sorption dynamics modeling. Ind Eng Chem Res 51(2):900–914. https://doi.org/10.1021/ie202189v
Nguyen MV, Lee BK (2012) Improvement of yields and surface areas of biochar from chicken manure. J Biobased Mater Bio 6(6):714–716. https://doi.org/10.1166/jbmb.2012.1305
Nishimiya K (1998) Analysis of chemical structure of wood charcoal by X-ray photoelectron spectroscopy. J Wood Sci 44(1):56–61. https://doi.org/10.1007/BF00521875
Özçimen D, Ersoy-Meriçboyu A (2010) Chracterization of biochar and bio-oil samples obtained from carbonation of various biomass materials. Renew Energ 35(6):1319–1324. https://doi.org/10.1016/j.renene.2009.11.042
Purevsuren B, Avid B, Tesche B, Davaajav YA (2003) A biochar from casein and its properties. J Mater Sci 38(11):2347–2351. https://doi.org/10.1023/A:1023980429410
Rao MA, Simeone GDR, Scelza R, Conte P (2017) Biochar based remediation of water and soil contaminated by phenanthrene and pentachlorophenol. Chemosphere 186:193–201. https://doi.org/10.1016/j.chemosphere.2017.07.125
Srinivasan P, Sarmah AK (2015) Characterisation of agricultural waste-derived biochars and their sorption potential for sulfamethoxazole in pasture soil: a spectroscopic investigation. Sci Total Environ 502:471–480. https://doi.org/10.1016/j.scitotenv.2014.09.048
Sun JK, Lian F, Liu ZQ, Zhu LY, Song ZG (2014) Biochars derived from various crop straws: characterization and Cd (II) removal potential. Ecotox Environ Safe 106:226–231. https://doi.org/10.1016/j.ecoenv.2014.04.042
Tsaia WT, Liu SC, Chen HR, Chang YM, Yi-Lin Tsai YL (2012) Textural and chemical properties of swine-manure-derived biochar pertinent to its potential use as a soil amendment. Chemosphere 89(2):198–203. https://doi.org/10.1016/j.chemosphere.2012.05.085
Uchimiya M, Chang S, Klasson KT (2011) Screening biochars for heavy metal retention in soil: role of oxygen functional groups. J Hazard Mater 190(1-3):432–441. https://doi.org/10.1016/j.jhazmat.2011.03.063
Wang XL, Xing BS (2007) Importance of structural makeup of biopolymers for organic contaminant sorption. Environ Sci Technol 41(10):3559–3565. https://doi.org/10.1021/es062589t
Wang XS, Chen LF, Li FY, Chen KL, Wan WY, Tang YJ (2010) Removal of Cr (VI) with wheat-residue derived black carbon: reaction mechanism and adsorption performance. J Hazard Mater 175(1-3):816–822. https://doi.org/10.1016/j.jhazmat.2009.10.082
Yang GX, Jiang H (2014) Amino modification of biochar for enhanced adsorption of copper ions from synthetic wastewater. Water Res 48:396–405. https://doi.org/10.1016/j.watres.2013.09.050
Yuan HR, Lu T, Wang YZ, Huang HY, Chen Y (2014) Influence of pyrolysis temperature and holding time on properties of biochar derived from medicinal herb (radix isatidis) residue and its effect on soil CO2 emission. J Anal Appl Pyrol 110:277–284. https://doi.org/10.1016/j.jaap.2014.09.016
Yuan JH, RK X, Zhang H (2011) The forms of alkalis in the biochar produced from crop residues at different temperatures. Bioresour Technol 102(3):3488–3497. https://doi.org/10.1016/j.biortech.2010.11.018
Zhang J, Liu J, Liu RL (2015) Effects of pyrolysis temperature and heating time on biochar obtained from the pyrolysis of straw and lignosulfonate. Bioresour Technol 176:288–291. https://doi.org/10.1016/j.biortech.2014.11.011
Zhang JJ, Hu F, Li HX, Gao Q, Song XY, Ke XK, Wang LC (2011) Effects of earthworm activity on humus composition and humic acid characteristics of soil in a maize residue amended rice-wheat rotation agroecosystem. Appl Soil Ecol 51:1–8. https://doi.org/10.1016/j.apsoil.2011.08.004
Zhao N, Lv YZ (2012) Effects of different fertilization treatments on soil humic acids structure characteristics. Spectrosc Spect Anal 32(7):1586–1589
Zhao N, Lv YZ, Li GJ (2013) Characterization and three-dimensional structural modeling of humic acid via molecular mechanics and molecular dynamics simulation. Chem Res Chin Univ 29(6):1180–1184. https://doi.org/10.1007/s40242-013-3156-x
Zhao N, Lv YZ, Yang XX (2017) A new 3D conceptual structures modeling of biochars by molecular mechanics and molecular dynamic simulation. J Soil Sediment 17:641–655
Zheng W, Guo MX, Chow T, Bennett N, Rajagopalan N (2010) Sorption properties of greenwaste biochar for two triazine pesticides. J Hazard Mater 181(1-3):121–126. https://doi.org/10.1016/j.jhazmat.2010.04.103
Funding
This research was supported by the National Natural Science Foundation of China (41571317, 41271331) and China Postdoctoral Science Foundation (2016 M591267).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible editor: Philippe Garrigues
Electronic supplementary material
Table S1
(DOCX 288 kb)
Rights and permissions
About this article
Cite this article
Zhao, N., Lv, Y., Yang, X. et al. Characterization and 2D structural model of corn straw and poplar leaf biochars. Environ Sci Pollut Res 25, 25789–25798 (2018). https://doi.org/10.1007/s11356-017-0959-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0959-1