Abstract
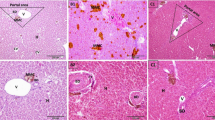
Although tannery effluents are known for being highly toxic to organisms, reports about the effects of the intake of these xenobiotics on experimental mammal models are recent. Studies about the damages the chronic intake of these effluents can cause in the liver of outbred mice remain an unexplored field. Thus, the aim of the present study is to assess (histological) the hepatic condition of Swiss mice (outbred strain) chronically exposed to the intake of different raw tannery effluent concentrations diluted in water for 150 days. Accordingly, the mice (males and females) were divided in the following groups: control group—animals treated with drinking water, only; and groups 5 and 10%—treated with raw tannery effluent diluted in water. After exposure, the animals were subjected to euthanasia for liver fragment sample collection and histological analysis, respectively. Moderate hydropic degeneration was observed in the centrilobular regions of the liver of mice exposed to 5 and 10% tannery effluent, as well as greater amounts of hepatocytes presenting karyomegaly and necrotic hepatocytes, and a smaller amount of Kuffer cells in the liver of mice exposed to the xenobiotic. Finally, animals exposed to 10% tannery effluent showed mild hyperplasia of the bile ducts in the portal areas and fibroblast proliferation around the bile ducts, thus suggesting a fibrous process. Except for the frequency of hepatocytes presenting karyomegaly (lower in females), the herein observed hepatic changes were similar in male and female Swiss mice. Accordingly, the present data support the hypothesis that the chronic intake of tannery effluent by outbred mice (Swiss) causes damages in the liver, a fact that broadens the knowledge about the toxic potential of this pollutant, which goes beyond that of C57Bl/6J male mice (inbred strain).



Similar content being viewed by others
Change history
27 March 2018
There is a problem in the original publication of this paper (Figure 2 Graphs are in Portuguese). Shown in this paper is the correct version.
References
Ajayan KV, Selvaraju M (2012) Heavy metal induced antioxidant defense system of green microalgae and its effective role in phycoremediation of tannery effluent. Pak J Biol Sci 15(22):1056–1062
Almeida SF, Rabelo LM, Souza JM, Ferreira RO, Guimarães ATB, Pereira CCO, Rodrigues ASL, Malafaia G (2016) Behavioral changes in female Swiss mice exposed to tannery effluents. Ambiente & Água 11(3):519–534
Ambreen K, Khan FH, Bhadauria S, Kumar S (2014) Genotoxicity and oxidative stress in chromium-exposed tannery workers in North India. Toxicol Ind Health 30(5):405–414
Behmer AO, Tolosa EM, Freitas-Neto AG (1976) Manual de técnicas para histologia normal e patológica. Ed. Art/Edusp, São Paulo, p 155
China CR, Ndaro MS (2015) A study on leather sector investment status in Tanzania. African Journal of Science and Research 6(4):19–22
Ferreira RO, Guimarães ATB, Silva BC, Silva WMA, Mendes BO, Rodrigues ASL, Almeida SF, Souza JM, Rabelo LM, Estrela DC, Malafaia G (2015) Análise de toxicidade aguda e determinação da dose letal mediana (DL50) de efluente de curtume em camundongos Swiss. Multi-Science Jounal 1(3):83–87
Guimarães AT, de Oliveira Ferreira R, de Souza JM, da Silva WA, da Silva AR, de Lima Rodrigues AS, de Melo E, Silva D, Costa RM, da Silva Castro AL, Malafaia G (2016b) Anxiety and memory deficits induced by tannery effluent in C57BL/6J female mice. Environ Sci Pollut Res 23(24):25323–25334. https://doi.org/10.1007/s11356-016-7746-2
Guimarães ATB, Ferreira RO, Rabelo LM, Silva BC, Souza JM, Silva WMA, Menezes IPP, Rodrigues ASL, Vaz BG, Costa DRO, Pereira I, Silva AR, Malafai G (2016a) The C57BL/6J mice offspring originated from a parental generation exposed to tannery effluents shows object recognition deficits. Chemosphere 164(13):593–602
Harada T, Enomoto A, Boorman GA, Maronpot RR (1999) Liver and gallbladder. In: Maronpot RR (ed) Pathology of the mouse. Cache River Press, Vienna, IL, pp 119–183
Inoue KM, Shibutani M, Masutomi N, Toyoda K, Takagi H, Uneyama C, Nishikawa A, Hirose M (2008) A 13-week subchronic toxicity study of madder color in F344 rats. Food Chem Toxicol 46(1):241–252
Inoue K, Yoshida M, Takahashi M, Shibutani M, Takagi H, Hirose M, Nishikawa A (2009) Induction of kidney and liver cancers by the natural food additive madder color in a two-year rat carcinogenicity study. Food Chem Toxicol 47(6):184–191
Kitaura K, Chone Y, Satake N, Akagi A, Ohnishi T, Suzuki Y, Izumi K (1999) Role of copper accumulation in spontaneous renal carcinogenesis in Long-Evans Cinnamon rats. Jpn J Cancer Res 90(4):385–392
Lee J, Prokopec SD, Watson JD, Sun RX, Pohjanvirta R, Boutros PC (2015) Male and female mice show significant differences in hepatic transcriptomic response to 2,3,7,8-tetrachlorodibenzo-p-dioxin. BMC Genomics 16(1):625
Luna A, Meister HP, Szanto PB (1968) Esophageal varices in the absence of cirrhosis: incidence and characteristics in congestive heart failure and neoplasm of the liver. Am J Clin Pathol 49(5):710–717
Manivasagam N (1987) Industrial effluents origin, characteristic effects, analysis and treatment Kovaipudur, India. Shakti Publication 42:1–476
Mishra A, Mishra KP (2015) Bacterial response as determinant of oxidative stress by heavy metals and antibiotic. J Innov Pharm Biol Sci 2(3):229–239
Mosher B, Dean R, Harkema J, Remick D, Palma J, Crockett E (2001) Inhibition of Kupffer cells reduced CXC chemokine production and liver injury. J Surg Res 99(2):201–210
Percy DH, Barthold SW (2001) Hamster. In: Pathology of laboratory rodents and rabbits. Blackwell Publishing, Ames, pp 194–195
Prabakaran M, Binuramesh C, Steinhagen D, Dinakaran Michael R (2007) Immune response in the tilapia, Oreochromis mossambicus on exposure to tannery effluent. Ecotoxicol Environ Saf 68(3):372–378
Rabelo LM, Silva BC, Almeida SF, Silva WAM, Mendes BO, Guimarães ATB, Silva AR, Castro ALS, Rodrigues ASL, Malafaia G (2016) Memory deficit in Swiss mice exposed to tannery effluent. Neurotoxicol Teratol 55(55):45–49
Roy S, Nagarchi L, Das I, Achuthananthan JM, Krishnamurthy S (2015). Cytotoxicity, genotoxicity, and phytotoxicity of tannery effluent discharged into Palar River basin, Tamil Nadu, India. J Toxicol. 2015(504360):9. https://doi.org/10.1155/2015/504360
Sabumon PC (2016) Perspectives on biological treatment of tannery effluent. Adv Recycl Waste Manag: Open Acess 1(1):3–10
Silva WAM, Mendes BO, Guimarães ATB, Rabelo LM, Ferreira RO, Silva BC, Soouza JM, Menezes IPP, Rodrigues ASL, Malafaia G (2016) Dermal exposure to tannery effluent causes neurobehavioral changes in C57Bl/6J and Swiss mice. Chemosphere 160:237–243
Siqueira IR, Vanzella CB, Paula RR, Rodrigues MAS, Stülp S (2011) Anxiety-like behaviour in mice exposed to tannery wastewater: the effect of photoelectrooxidation treatment. Neurotoxicol Teratol 33(4):481–484
Souza JM, Guimarães ATB, Silva WAM, Mendes BO, Estrela DC, Rodrigues ASL, Santos AS, Malafaia G (2016b) Histopathological assessment of C57BL/6J mice organs exposed to tannery effluents. Revista Ambiente & Água 11(1):24–34
Souza JM, Silva WAM, Mendes BO, Guimarães ATB, Almeida SF, Estrela DC, Silva AR, Rodrigues ASL, Malafaia G (2016a) Neurobehavioral evaluation of C57BL/6J mice submitted to tannery effluents intake. JSM Anxiety and Depression 1(1):1006
Stalker MJ, Hayes MA (2007) Liver and biliary system. In: Maxie MG (ed) Jubb, Kennedy, and Palmer’s pathology of domestic animals, vol 2, 5th edn. Saunders Elsevier, Philadelphia, pp 297–388
Thoolen B, Maronpot RR, Harada T, Nyska A, Rousseaux C, Nolte T, Malarkey D, Kaufmann W, Kutter K, Deschl U, Nakae D, Gregson R, Winlove M, Brix A, Singl B, Belpoggi F, Ward JM (2010) Hepatobiliary lesion nomenclature and diagnostic criteria for lesions in rats and mice (INHAND). Toxicol Pathol 38:5S–81S
Funding
The authors are grateful to Brazilian National Council for Research (CNPq) (Brazilian research agency) (Proc. No. 467801/2014-2) and to Instituto Federal Goiano for the financial support. Moreover, the authors are grateful to CNPq for granting a scholarship to the student who developed the current study.s
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
All procedures were approved by the Institutional Committee for Animal Care and Use of Goiano Federal Institute (Goiás State, Brazil) (protocol no. 17/2014) according to the Guide for Care and Use of Laboratory Animals, 8th edition. Animal handling and all the experiments were performed according to the International Guidelines for Animal Welfare.
Additional information
Responsible editor: Philippe Garrigues
A correction to this article is available online at https://doi.org/10.1007/s11356-018-1805-9.
Rights and permissions
About this article
Cite this article
Rabelo, L.M., Guimarães, A.T.B., de Souza, J.M. et al. Histological liver chances in Swiss mice caused by tannery effluent. Environ Sci Pollut Res 25, 1943–1949 (2018). https://doi.org/10.1007/s11356-017-0647-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11356-017-0647-1