Abstract
Purpose
The basal ganglia have long been implicated in motor and cognitive function. Previous research has shown that regular forced treadmill running improves cognitive performance especially learning and memory. In this study, we examined the role of mild forced treadmill exercise on spatial learning and memory, and motor activity.
Methods
56 male Wistar rats were randomly split into seven groups. Animals received ibotenic acid infusions into striatum bilaterally. Spatial learning and memory for all rats were evaluated by Morris water maze test. On the following day, visible and probe tests were performed for all groups. Motor activity of rats was assessed by open-field apparatus.
Results
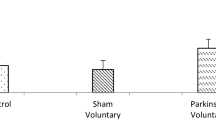
Our results showed that animals with striatal lesion were much slower at finding the platform. Mild treadmill exercise during training could decrease the distance traveled to find the hidden platform. While exercise has shown to improve learning/memory, it has not been able to completely treat the impairment caused by ibotenic acid injection into the striatum. Results of open-field test were congruent with the Morris water maze test, with exercise animals displaying higher levels of motor activity.
Conclusions
The present results suggest that striatum has a critical role in spatial learning and memory. However, mild treadmill exercise could not improve impaired spatial learning and memory in rats with striatum lesion.












Similar content being viewed by others
References
Aguiar AS, Araújo AL, da-Cunha TR, Speck AE, Ignácio ZM, De-Mello N, Prediger RD (2009) Physical exercise improves motor and short-term social memory deficits in reserpinized rats. Brain Res Bull 79:452–457
Alexander GE, DeLong MR, Strick PL (1986) Parallel organization of functionally segregated circuits linking basal ganglia and cortex. Ann Rev Neurosci 9:357–381
Alheid G, Heimer L (1988) New perspectives in basal forebrain organization of special relevance for neuropsychiatric disorders: the striatopallidal, amygdaloid, and corticopetal components of substantia. Innominata Neurosci 27:1–39
Ang E-T, Dawe GS, Wong PT, Moochhala S, Ng Y-K (2006) Alterations in spatial learning and memory after forced exercise. Brain Res 1113:186–193
Atallah HE, Frank MJ, O’Reilly RC (2004) Hippocampus, cortex, and basal ganglia: insights from computational models of complementary learning systems. Neurobiol Learn Memory 82:253–267
Baker PM, Ragozzino ME (2014) Contralateral disconnection of the rat prelimbic cortex and dorsomedial striatum impairs cue-guided behavioral switching. Learn Memory 21:368–379
Block F, Kunkel M, Schwarz M (1993) Quinolinic acid lesion of the striatum induces impairment in spatial learning and motor performance in rats. Neurosci Lett 149:126–128
Care IoLARCo A, Resources UoL NIoHDoR (1985) Guide for the care and use of laboratory animals. National Academies, Washington
Cassilhas R, Lee K, Fernandes J, Oliveira M, Tufik S, Meeusen R, De Mello M (2012) Spatial memory is improved by aerobic and resistance exercise through divergent. Mol Mech Neurosci 202:309–317
Cassilhas RC, Tufik S, de Mello MT (2016) Physical exercise, neuroplasticity, spatial learning and memory. Cell Mol Life Sci 73:975–983
Cassilhas RC, Viana VA, Grassmann V, Santos RT, Santos RF, Tufik S, Mello MT (2007) The impact of resistance exercise on the cognitive function of the elderly. Med Sci Sports Exerc 39:1401
Costa C et al (2012) Mechanisms underlying the impairment of hippocampal long-term potentiation and memory in experimental Parkinson’s disease Brain 101
David FJ et al (2015) Exercise improves cognition in Parkinson’s disease: the PRET-PD randomized, clinical trial. Mov Disord 30:1657–1663
de Senna PN et al (2015) Physical training improves non-spatial memory, locomotor skills and the blood brain barrier in diabetic rats. Brain Res 1618:75–82
Döbrössy MD, Dunnett SB (2003) Motor training effects on recovery of function after striatal lesions and striatal grafts. Exp Neurol 184:274–284
Esch T, Stefano GB, Fricchione GL, Benson H (2002) The role of stress in neurodegenerative diseases and mental disorders. Neuroendocrinol Lett 23:199–208
Espay AJ et al (2011) Differential response of speed, amplitude, and rhythm to dopaminergic medications in Parkinson’s disease. Mov Disord 26:2504–2508
Garcia PC, Real CC, Ferreira AF, Alouche SR, Britto LR, Pires RS (2012) Different protocols of physical exercise produce different effects on synaptic and structural proteins in motor areas of the rat brain. Brain Res 1456:36–48
Hoveida R, Alaei H, Oryan S, Parivar K, Reisi P (2011) Treadmill running improves spatial memory in an animal model of Alzheimer’s disease. Behav Brain Res 216:270–274
Hüttenrauch M et al (2016) Physical activity delays hippocampal neurodegeneration and rescues memory deficits in an Alzheimer disease mouse model. Transl Psychiatry 6:e800
Iodice P, Ferrante C, Brunetti L, Cabib S, Protasi F, Walton ME, Pezzulo G (2017) Fatigue modulates dopamine availability and promotes flexible choice reversals during decision making Scientific Reports 7
Kim S-H et al (2002) Treadmill exercise increases cell proliferation without altering of apoptosis in dentate gyrus of Sprague-Dawley rats. Life Sci 71:1331–1340
Kohl Z et al (2007) Physical activity fails to rescue hippocampal neurogenesis deficits in the R6/2 mouse model of Huntington’s disease. Brain Res 1155:24–33
Lee B, Sur B-J, Kwon S, Jung E, Shim I, Lee H, Hahm D-H (2011) Acupuncture stimulation alleviates corticosterone-induced impairments of spatial memory and cholinergic neurons in rats Evid Based Complement Altern Med 2012
Lupien SJ, McEwen BS, Gunnar MR, Heim C (2009) Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat Rev Neurosci 10:434–445
Mariotti R, Fattoretti P, Malatesta M, Nicolato E, Sandri M, Zancanaro C (2014) Forced mild physical training improves blood volume in the motor and hippocampal cortex of old mice. J Nutr Health aging 18:178–183
McDonald RJ, Devan BD, Hong NS (2004) Multiple memory systems: the power of interactions. Neurobiol Learn Memory 82:333–346
Morton AJ, Skillings E, Bussey TJ, Saksida LM (2006) Measuring cognitive deficits in disabled mice using an automated interactive touchscreen system. Nat Methods 3:767–767
Murray DK, Sacheli MA, Eng JJ, Stoessl AJ (2014) The effects of exercise on cognition in Parkinson’s disease: a systematic review Transl Neurodegener 3:1
Packard MG, Knowlton BJ (2002) Learning and memory functions of the basal ganglia. Annu Rev Neurosci, 25(1), 563–593
Potter MC, Yuan C, Ottenritter C, Mughal M, van Praag H (2010) Exercise is not beneficial and may accelerate symptom onset in a mouse model of Huntington’s disease PLOS Curr Huntington Dis
Prado-Alcala R, Grinberg Z, Arditti Z, Garcia M, Prieto H, Brust-Carmona H (1975) Learning deficits produced by chronic and reversible lesions of the corpus striatum in rats. Physiol Behav 15:283–287
Paxinos G, Watson CR, Emson PC (1980) AChE-stained horizontal sections of the rat brain in stereotaxic coordinates. J Neurosci methods 3(2):129–149
Rosenblum S, Samuel M, Zlotnik S, Erikh I, Schlesinger I (2013) Handwriting as an objective tool for Parkinson’s disease diagnosis. J Neurol 260:2357–2361
Tarsy D, Vitek JL, Lozano AM (Eds) (2002) Surgical treatment of Parkinson’s disease and other movement disorders. Springer Science & Business Media
Van Praag H (2008) Neurogenesis and exercise: past and future directions. Neuromol Med 10:128–140
Van Praag H, Christie BR, Sejnowski TJ, Gage FH (1999) Running enhances neurogenesis, learning, and long-term potentiation in mice Proc Natl Acad Sci 96:13427–13431
Van Praag H, Shubert T, Zhao C, Gage FH (2005) Exercise enhances learning and hippocampal neurogenesis in aged mice. J Neurosci 25:8680–8685
Vorhees CV, Williams MT (2006) Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nat Protoc 1:848–858
Whishaw I, Zeeb F, Erickson C, McDonald R (2007) Neurotoxic lesions of the caudate-putamen on a reaching for food task in the rat: acute sensorimotor neglect and chronic qualitative motor impairment follow lateral lesions and improved success follows. Medial Lesions Neurosci 146:86–97
Whishaw IQ, Mittleman G, Bunch ST, Dunnett SB (1987) Impairments in the acquisition, retention and selection of spatial navigation strategies after medial caudate-putamen lesions in rats. Behav Brain Res 24:125–138
Wichmann T, DeLong MR (1996) Functional and pathophysiological models of the basal ganglia. Curr Opin Neurobiol 6:751–758
Winocur G, Mills JA (1969) Effects of caudate lesions on avoidance behavior in rats. J Comp Physiol Psychol 68:552
Acknowledgements
The experiments were supported by grants from Pasteur Institute of Iran, and Tehran University.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors have declared no conflict of interest.
Ethical standard
These animal experiments were carried out in accordance with the recommendations of the Declaration of Helsinki and the internationally accepted principles for the use of experimental animals. All efforts were made to minimize animal suffering.
Informed Consent
For this type of study, formal consent is not required.
Rights and permissions
About this article
Cite this article
Modaberi, S., Shahbazi, M., Dehghan, M. et al. The role of mild treadmill exercise on spatial learning and memory and motor activity in animal models of ibotenic acid-induced striatum lesion. Sport Sci Health 14, 587–596 (2018). https://doi.org/10.1007/s11332-018-0467-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-018-0467-9