Abstract
Objectives
There were limitations in the conventional treatments for depression. This study investigated if an aerobic exercise programme would be a beneficial adjunct for outpatients on treatment for depression in Hong Kong.
Methods
We performed a single blind randomized controlled trial on 34 adult patients (between 18 and 65) suffering from major depressive disorder. Their Hamilton Depression Rating Scale (HAM-D) scores were at least 14. They were randomly assigned to receive a 12-week aerobic exercise programme in addition to usual psychiatric care (intervention), or to continue with usual psychiatric care alone (control). The outcomes included depression severity, sleep quality, somatic symptoms distress level, and anxiety level.
Results
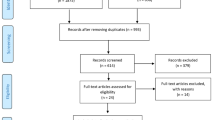
Seventeen participants were randomly allocated to each group. There was statistically significant reduction in the mean HAM-D scores in both intervention (18.5–9.8) and control (19.5–14.5) groups. There were statistically significant main effects for time, for group, and for time*group interaction. On the sleep quality measures, there was statistically significant improvement in the global Pittsburg Sleep Quality Index in the intervention group only (from 12.0 to 9.0).
Conclusion
This study provided suggestive evidence that aerobic exercise might be a beneficial adjunct treatment for depression.


Similar content being viewed by others
References
American Heart Association (2016) Target Heart Rate. http://www.heart.org/HEARTORG/HealthyLiving/PhysicalActivity/FitnessBasics/Target-Heart-Rates_UCM_434341_Article.jsp#.WigOUbaB36Y. Accessed 7 Dec 2017
Bagby RM, Ryder AG, Schuller DR, Marshall MB (2004) The Hamilton Depression Rating Scale: has the gold standard become a lead weight? Am J Psychiatry 2004(161):2163–2177
Bjelland I, Dahl AA, Haug TT, Neckelmann D (2002) The validity of the Hospital Anxiety and Depression Scale: an updated literature review. J Psychosom Res 2002(52):69–77
Blumenthal JA, Babyak MA, Moore KA, Craighead WE, Herman S, Hkatri P, Krishnan KR (1999) Effects of exercise training on older patients with major depression. Arch Intern Med 159(19):2349–2356
Callaghan P, Khalil E, Morres I, Carter T (2011) Pragmatic randomised controlled trial of preferred intensity exercise in women living with depression. BMC Public Health 11:465. http://www.biomedcentral.com/1471-2458/11/465
Campbell M, Fitzpatrick R, Haines A, Kinmonth AL, Sandercock P, Spiegelhalter D, Tyrer P (2000) Framework for design and evaluation of complex interventions to improve health. BMJ 2000(321):694–696
Chalder M, Wiles NJ, Campbell J, Hollinghurst SP, Haase AN, Taylor AH, Lewis G (2012) Facilitated physical activity as a treatment for depressed adults: randomised controlled trial. BMJ 2012(344):e2758. https://doi.org/10.1136/bmj.e2758
Chong AM, Cheung CK (2012) Factor structure of a Cantonese-version Pittsburgh Sleep Quality Index. Sleep and Biological Rhythms 10(2):118–125
Craft LL (2005) Exercise and clinical depression: examining two psychological mechanisms. Psychol Sport Exerc 2005(6):151–171
Craft LL, Perna FM (2004) The benefits of exercise for the clinically depressed. Prim Care Companion J Clin Psychiatry 6(3):104–111
Cuijpers P, van Straten A, Bohlmeijer E, Hollon SD, Andersson G (2010) The effects of psychotherapy for adult depression are overestimated: a meta-analysis of study quality and effect size. Psychol Med 2010(40):211–223. https://doi.org/10.1017/S0033291709006114
De Moor MH, Beem AL, Stubbe JH, Boomsma DI, De Geus EJ (2006) Regular exercise, anxiety, depression and personality: a population-based study. Prev Med 42(4):273–279
Dimeo F, Bauer M, Varahram I, Proest G, Halter U (2001) Benefits from aerobic exercise in patients with major depression: a pilot study. Br J Sports Med 2001(35):114–117
Dunn AL, Jewell JS (2010) The effect of exercise on mental health. Curr Sports Med Rep 9(4):202–207
Dunn AL, Trivedi MH, Kampert JB, Clark CG, Chambliss HO (2005) Exercise treatment for depression: efficacy and dose response. Am J Prev Med 28(1):1–8
Ernst C, Olson AK, Pinel JP, Lam RW, Christie BR (2006) Antidepressant effects of exercise: evidence for an adult-neurogenesis hypothesis? J Psychiatry Neurosci 31(2):84–92
Franzen PL, Buysee DJ (2008) Sleep disturbances and depression: risk relationships for subsequent depression and therapeutic implications. Dialogues Clin Neurosci 2008(10):473–481
Fournier JC, DeRubeis RJ, Hollon SD, Dimidjian S, Amsterdam JD, Shelton RC, Fawcett J (2010) Antidepressant drug effects and depression severity: a patient-level meta-analysis. J Am Medicat Assoc 303(1):47–53
Glass JM, Lyden AK, Petzke F, Stein P, Whalen G, Ambrose K, Clauw DJ (2004) The effect of brief exercise cessation on pain, fatigue, and mood symptom development in healthy, fit individuals. J Psychosom Res 2004(57):391–398
Hallgren M, Kraepelien M, Öjehagen A, Lindefors N, Zeebari Z, Kaldo V, Forsell Y (2015) Physical exercise and internet-based cognitive-behavioural therapy in the treatment of depression: randomised controlled trial. Br J Psychaitric 2015(207):227–234
Hamilton M (1960) A rating scale for depression. J Neurol Neurosurg Psychiatry 1960(23):56–62
Harvey SB, Hotopf M, Øverland S, Mykletun A (2010) Physical activity and common mental disorders. Br J Psychiatry 2010(197):357–364
Kessler RC, Berglund P, Demler O, Jin R, Koretz D, Merikangas KR, Wang PS (2003) The epidemiology of major depressive disorder: results from the National Comorbidity Survey Replication (NCS-R). J Am Med Assoc 289(23):3095–3105
Kinser PA, Robins JL (2013) Control group design: enhancing rigor in research of mind-body therapies for depression. Evid Based Complement Alternat Med. https://doi.org/10.1155/2013/140467
Lawlor DA, Hopker SW (2001) The effectiveness of exercise as an intervention in the management of depression: systematic review and meta-regression analysis of randomized controlled trials. BMJ 2001(322):1–8
Lee S, Ma YL, Tsang A (2011) Psychometric properties of the Chinese 15-item Patient Health Questionnaire in the general population of Hong Kong. J Psychosom Res 2011(71):69–73
Leung CM, Wing YK, Kwong PK, Lo A, Shum K (1999) Validation of the Chinese-Cantonese version of the Hospital Anxiety and Depression Scale and comparison with the Hamilton Rating Scale of Depression. Acta Psychiatr Scand 1999(100):456–461
Moskowitz DS, Young SN (2006) Ecological momentary assessment: what it is and why it is a method of the future in clinical psychopharmacology. J Psychiatry Neurosci 31(1):13–20
Mota-Pereira J, Silverio J, Carvalho S, Ribeiro JC, Fonte D, Ramos J (2011) Moderate exercise improves depression parameters in treatment-resistant patients with major depressive disorder. J Psychiatr Res 2011(45):1005–1011
Muntner P, Gu D, Wildman RP, Chen J, Qian W, Whelton PK, He J (2005) Prevalence of physical activity among Chinese adults: Results from the International Collaborative Study of Cardiovascular Disease in Asia. Am J Public Health 95(9):1631–1636
Mura G, Moro MF, Patten SB, Carta MG (2014) Exercise as an add-on strategy for the treatment of major depressive disorder: a systematic review. CNS Spectr 2014(3):1–13. https://doi.org/10.1017/S1092852913000953
Murray L, Mori I (2006) Sport, exercise and physical activity: public participation, barriers and attitudes. Scottish Executive Social Research, pp 19–23. Retrieved from http://www.scotland.gov.uk/Resource/Doc/932/0041468.pdf
NICE (2010) Depression: the treatment and management of depression in adults (updated edition). CG90. National Institute for Health and Care Excellence, London
Pampallona S, Bollini P, Tibaldi G, Kupelnick B, Munizza C (2002) Patient adherence in the treatment of depression. Br J Psychiatry 2002(180):104–109
Papakostas GI (2008) Tolerability of modern antidepressants. J Clin Psychiatry 69(Suppl E1):8–13
Pilu A, Sorba M, Hardoy MC, Floris AN, Mannu F, Maria LS, Carta MG (2007) Efficacy of physical activity in the adjunctive treatment of major depressive disorders: preliminary results. Clin Pract Epidemiol Ment Health 2007(3):8. https://doi.org/10.1186/1745-0179-3-8
Reid KJ, Baron KG, Lu B, Naylor E, Wolfe L, Zee PC (2010) Aerobic exercise improves self-reported sleep and quality of life in older adults with insomnia. Sleep Med 11(9):934–940. https://doi.org/10.1016/j.sleep.2010.04.014
Rimer J, Dwan K, Lawlor DA, Greig CA, McMurdo M, Morley W, Mead GE (2012) Exercise for depression. Cochrane Database Syst Rev 7:CD004366. https://doi.org/10.1002/14651858.cd004366.pub5
Seime RJ, Vickers KS (2006) The challenges of treating depression with exercise: from evidence to practice. Clin Psychol Sci Pract 13(2):194–197
Shek DT (1993) The Chinese version of the State-Trait Anxiety Inventory: its relationship to different measures of psychological well-being. J Clin Psychol 49(3):349–358
Singh NA, Stavrinos TM, Scarbek Y, Galambos G, Liber C, Singh MA (2005) A randomized controlled trial of high versus low intensity weight training versus general practitioner care in clinical depression in older adults. J Gerontol MEDICAL SCIENCES 60A 6:768–776
So E, Kam I, Leung CM, Chung D, Liu Z, Fong S (2003) The Chinese-bilingual SCID-I/P Project: stage 1—reliability for mood disorders and schizophrenia. Hong Kong J Psychiatry 2003(1):7–18
Ten Have M, de Graaf R, Monshouwer K (2011) Physical exercise in adults and mental health status: findings from the Netherlands Mental Health Survey and Incidence Study (NEMESIS). J Psychosom Res 2011(71):342–348
Trivedi MH, Greer TL, Grannemann BD, Chambliss HO, Jordan AN (2006) Exercise as an augmentation strategy for treatment of major depression. J Psychiat Pract 12(4):205–213
Veale D, Le Fevre K, Pantelis C, de Souza V, Mann A, Sargeant A (1992) Aerobic exercise in the adjunctive treatment of depression: a randomized controlled trial. J R Soc Med 85(9):541–544
Vos T, Flaxman AD, Maghavi M, Lozano R, Michaud C, Ezzati M, Murray CJ (2012) Years lived with disability (YLDs) for 1160 sequelae of 289 diseases and injuries 1990–2010: a systematic analysis for the Global Burden of Disease Study 2010. Lancet 380(9859):2163–2196
Warden D, Rush AJ, Trivedi MH, Fava M, Wisniewski SR (2007) The STAR*D project results: a comprehensive review of findings. Curr Psychiatry Rep 2007(9):449–459
Acknowledgements
We are grateful to Professor Linda Lam, Department of Psychiatry, the Chinese University of Hong Kong for her assistance in the planning and development of this exercise intervention research. We wish to acknowledge the help by Mr. Steve Wong in carrying out the exercise intervention, and we are thankful to other staff of the therapeutic physical mind exercise centre for their assistance.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical standards
The study protocol was approved by the Joint Chinese University of Hong Kong—New Territories East Cluster Clinical Research Ethics Committee. The trial was registered in the Centre for Clinical Research and Biostatistics, Clinical Trials Registry, Chinese University of Hong Kong. The clinical trial Registration Number was CUHK_CCT00375. The study was conducted in accordance with the principles of the Helsinki Declaration of the World Medical Association.
Informed consent
All participants were required to sign informed consent prior to the study and voluntarily participated in the study.
Conflict of interest
We have read and understood Sports Sciences for Health policy on declaration of interest and declare that we have no conflicts of interest.
Rights and permissions
About this article
Cite this article
Cheung, L.K., Lee, S. A randomized controlled trial on an aerobic exercise programme for depression outpatients. Sport Sci Health 14, 173–181 (2018). https://doi.org/10.1007/s11332-017-0423-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11332-017-0423-0