Abstract
Introduction
Microbial cells secrete many metabolites during growth, including important intermediates of the central carbon metabolism. This has not been taken into account by researchers when modeling microbial metabolism for metabolic engineering and systems biology studies.
Materials and Methods
The uptake of metabolites by microorganisms is well studied, but our knowledge of how and why they secrete different intracellular compounds is poor. The secretion of metabolites by microbial cells has traditionally been regarded as a consequence of intracellular metabolic overflow.
Conclusions
Here, we provide evidence based on time-series metabolomics data that microbial cells eliminate some metabolites in response to environmental cues, independent of metabolic overflow. Moreover, we review the different mechanisms of metabolite secretion and explore how this knowledge can benefit metabolic modeling and engineering.






Similar content being viewed by others
References
Agarwal, L., Isar, J., Meghwanshi, G. K., & Saxena, R. K. (2006). A cost effective fermentative production of succinic acid from cane molasses and corn steep liquor by Escherichia coli. Journal of Applied Microbiology, 100, 1348–1354. https://doi.org/10.1111/j.1365-2672.2006.02894.x.
Airich, L. G., Tsyrenzhapova, I. S., Vorontsova, O. V., Feofanov, A. V., Doroshenko, V. G., & Mashko, S. V. (2010). Membrane topology analysis of the Escherichia coli aromatic amino acid efflux protein YDDG. Journal of Molecular Microbiology and Biotechnology, 19, 189–197. https://doi.org/10.1159/000320699.
Ajinomoto. (2009). Fact Sheet: Amino Acids Business [online]. 2015. Retrieved August 19, 2002 from https://www.ajinomoto.com/en/. Accessed 10 Oct 2017.
Allen, J., Davey, H.M., Broadhurst, D., Heald, J.K., Rowland, J.J., Oliver, S.G., et al. (2003). High-throughput classification of yeast mutants for functional genomics using metabolic footprinting. Nature Biotechnology, 21, 692–696.
Aung, H. W., Henry, S. A., & Walker, L. P. (2013). Revising the representation of fatty acid, glycerolipid, and glycerophospholipid metabolism in the consensus model of yeast metabolism. Industrial Biotechnology, 9, 215–228. https://doi.org/10.1007/s11306-014-0721-3.
Aurich, M. K., Paglia, G., Rolfsson, Ó, Hrafnsdóttir, S., Magnúsdóttir, M., Stefaniak, M. M., et al. (2014). Prediction of intracellular metabolic states from extracellular metabolomic data. Metabolomics, 11, 603–619. https://doi.org/10.1007/s11306-014-0721-3.
Bass, R. B., Strop, P., Barclay, M., & Rees, D. C. (2002). Crystal structure of Escherichia coli MscS, a voltage-modulated and mechanosensitive channel. Science, 298, 1582–1587. https://doi.org/10.1126/science.1077945.
Becker, M., Börngen, K., Nomura, T., Battle, A. R., Marin, K., Martinac, B., et al. (2013). Glutamate efflux mediated by Corynebacterium glutamicum MSCCG, Escherichia coli MSCS, and their derivatives. BBA-Biomembrane, 1828, 1230–1240. https://doi.org/10.1016/j.bbamem.2013.01.001.
Beese-Sims, S. E., Lee, J., & Levin, D. E. (2011). Yeast Fps1 glycerol facilitator functions as a homotetramer. Yeast, 28, 815–819. https://doi.org/10.1002/yea.1908.
Börngen, K., Battle, A. R., Möker, N., Morbach, S., Marin, K., Martinac, B., et al. (2010). The properties and contribution of the Corynebacterium glutamicum MSCS variant to fine-tuning of osmotic adaptation. BBA-Biomembrane, 1798, 2141–2149. https://doi.org/10.1016/j.bbamem.2010.06.022.
Boudker, O., & Verdon, G. (2010). Structural perspectives on secondary active transporters. Trends in Pharmacological Science, 31, 418–426. https://doi.org/10.1016/j.tips.2010.06.004.
Braga, R. M., Dourado, M. N., & Araujo, W. L. (2016). Microbial interactions: Ecology in a molecular perspective. Brazilian Journal of Microbiology, 47, 86–98.
Broda, P. (1968). Ribonucleic acid synthesis and glutamate excretion in Escherichia coli. Journal of Bacteriology, 96, 1528–1534.
Burkovski, A., & Krämer, R. (2002). Bacterial amino acid transport proteins: Occurrence, functions, and significance for biotechnological applications. Applied Microbiology and Biotechnology, 58, 265–274. https://doi.org/10.1007/s00253-001-0869-4.
Carneiro, S., Villas-Bôas, S. G., Ferreira, E. C., & Rocha, I. (2011). Metabolic footprint analysis of recombinant Escherichia coli strains during fed-batch fermentations. Molecular Biosystems, 7, 899–910. https://doi.org/10.1039/c0MB00143k.
Carneiro, S., Villas-Bôas, S. G., Ferreira, E. C., & Rocha, I. (2012). Influence of the RelA activity on E. coli metabolism by metabolite profiling of glucose-limited chemostat cultures. Metabolites, 2, 717–732. https://doi.org/10.3390/metabo2040717.
Chubukov, V., Gerosa, L., Kochanowski, K., & Sauer, U. (2014). Coordination of microbial metabolism. Nature Reviews Microbiology, 12, 327–340. https://doi.org/10.1038/nrmicro3238.
Chumnanpuen, P., Hansen, M. A. E., Smedsgaard, J., & Nielsen, J. (2014). Dynamic metabolic footprinting reveals the key components of metabolic network in yeast Saccharomyces cerevisiae. International Journal of Genomics 2014: Article ID 894296. https://doi.org/10.1155/2014/894296.
Cocaign-Bousquet, M., & Lindley, N. D. (1995). Pyruvate overflow and carbon flux within the central metabolic pathways of Corynebacterium glutamicum during growth on lactate. Enzyme and Microbial Technology, 17, 260–267. https://doi.org/10.1016/0141-0229(94)00023-K.
Crabtree, H. G. (1929). Observations on the carbohydrate metabolism of tumours. Biochemical Journal, 23, 536–545.
Daßler, T., Maier, T., Winterhalter, C., & Böck, A. (2000). Identification of a major facilitator protein from Escherichia coli involved in efflux of metabolites of the cysteine pathway. Molecular Microbiology, 36, 1101–1112.
Dörries, K., & Lalk, M. (2013). Metabolic footprint analysis uncovers strain specific overflow metabolism and d-isoleucine production of Staphylococcus aureus COL and HG001. PLoS ONE. https://doi.org/10.1371/journal.pone.0081500.
dos Santos, S. C., & Sa-Correia, I. (2011). A genome-wide screen identifies yeast genes required for protection against or enhanced cytotoxicity of the antimalarial drug quinine. Molecular Genetics and Genomics, 286, 333–346.
dos Santos, S. C., & Sa-Correia, I. (2015). Yeast toxicogenomics: Lessons from a eukaryotic cell model and cell factory. Current Opinion in Biotechnology, 33, 183–191.
dos Santos, S. C., Teixeira, M. C., Dias, P. J., & Sa-Correia, I. (2014). MFS transporters required for multidrug/multixenobiotic (MD/MX) resistance in the model yeast: Understanding their physiological function through post-genomic approaches. Frontiers in Physiology, 5, 180.
Driessen, A. J. M., & Konings, W.,N. (1990). Energetic problems of bacterial fermentations extrusion of metabolic end products. In T. A. Krulwich (Ed.), The bacteria: A treatise on structure and function. San Diego: Academic Press, Inc.
Düring-Olsen, L., Regenberg, B., Gjermansen, C., Kielland-Brandt, M. C., & Hansen, J. (1999). Cysteine uptake by Saccharomyces cerevisiae is accomplished by multiple permeases. Current Genetics, 35, 609–617.
Eggeling, L., & Sahm, H. (2003). New ubiquitous translocators: Amino acid export by Corynebacterium glutamicum and Escherichia coli. Archives of Microbiology, 180, 155–160. https://doi.org/10.1007/s00203-003-0581-0.
Fell, D. A. (1992). Metabolic control analysis—A survey of its theoretical and experimental development. Biochemical Journal, 286, 313–330.
Forrest, L. R., Krämer, R., & Ziegler, C. (2011). The structural basis of secondary active transport mechanisms. BBA-Bioenergetics, 1807, 167–188. https://doi.org/10.1016/j.bbabio.2010.10.014.
Forsberg, H., & Ljungdahl, P. O. (2001). Sensors of extracellular nutrients in Saccharomyces cerevisiae. Current Genetics, 40, 91–109. https://doi.org/10.1007/s002940100244.
Franke, S., Grass, G., Rensing, C., & Nies, D. H. (2003). Molecular analysis of the copper-transporting efflux system cuscfba of Escherichia coli. Journal of Bacteriology, 185, 3804–3812. https://doi.org/10.1128/JB.185.13.3804-3812.2003.
Fu, Z., Verderame, T. D., Leighton, J. M., Sampey, B. P., Appelbaum, E. R., Patel, P. S., et al. (2014). Exometabolome analysis reveals hypoxia at the up-scaling of a Saccharomyces cerevisiae high-cell density fed-batch biopharmaceutical process. Microbial Cell Factories, 13, 32. https://doi.org/10.1186/1475-2859-13-32.
Fukui, K., Koseki, C., Yamamoto, Y., Nakamura, J., Sasahara, A., Yuji, R., et al. (2011). Identification of succinate exporter in Corynebacterium glutamicum and its physiological roles under anaerobic conditions. Journal of Bacteriology, 154, 25–34. https://doi.org/10.1016/j.jbiotec.2011.03.010.
Geijer, C., Ahmadpour, D., Palmgren, M., Filipsson, C., Klein, D. M., et al. (2012). Yeast aquaglyceroporins use the transmembrane core to restrict glycerol transport. Journal of Biological Chemistry, 287, 23562–23570. https://doi.org/10.1074/jbc.M112.353482.
Granucci, N., Pinu, F. R., Han, T. L., & Villas-Boas, S. G. (2015). Can we predict the intracellular metabolic state of a cell based in extracellular metabolite data? Molecular BioSystems, 11, 3297–3304. https://doi.org/10.1039/C5MB00292C.
Guzmán, G. I., Utrilla, J., Nurk, S., Brunk, E., Monk, J. M., Ebrahim, A., et al. (2015). Model-driven discovery of underground metabolic functions in Escherichia coli. Proceedings of National Academy of Science of the United States of America 112, 929–934. https://doi.org/10.1073/pnas.1414218112.
Hagman, A., Säll, T., & Piškur, J. (2014). Analysis of the yeast short-term crabtree effect and its origin. FEBS Journal, 281, 4805–4814. https://doi.org/10.1111/febs.13019.
Han, T. L., Tumanov, S., Cannon, R. D., & Villas-Boas, S.,G. (2013). Metabolic response of Candida albicans to phenylethyl alcohol under hyphae-inducing conditions. PLoS ONE. https://doi.org/10.1371/journal.pone.0071364.
Hermann, T., & Krämer, R. (1996). Mechanism and regulation of isoleucine excretion in Corynebacterium glutamicum. Applied and Environmental Microbiology, 62, 3238–3244.
Hoffmann, T., Von Blohn, C., Stanek, A., Moses, S., Barzantny, H., & Bremer, E. (2012). Synthesis, release, and recapture of compatible solute proline by osmotically stressed Bacillus subtilis cells. Applied and Environmental Microbiology, 78, 5753–5762. https://doi.org/10.1128/AEM.01040-12.
Holme, T. (1957). Continuous culture studies on glycogen synthesis in Escherichia coli B. Acta Chemica Scandenavica, 11, 763–775.
Ingram, L. O. (1976). Adaptation of membrane lipids to alcohols. Journal of Bacteriology, 125, 670–678.
Kell, D. B., Brown, M., Davey, H. M., Dunn, W. B., Spasic, I., & Oliver, S. G. (2005). Metabolic footprinting and systems biology: The medium is the message. Nature Reviews Microbiology, 3, 557–565. https://doi.org/10.1038/nrmicro1177.
Kell, D. B., & Oliver, S. G. (2014). How drugs get into cells: Tested and testable predictions to help discriminate between transporter-mediated uptake and lipoidal bilayer diffusion. Frontiers in Pharmacology, 5, 231.
Kell, D. B., Peck, M. W., Rodger, G., & Morris, J. G. (1981). On the permeability to weak acids and bases of the cytoplasmic membrane of Clostridium pasteurianum. Biochemical and Biophysical Research Communications, 99(1), 81–88.
Kell, D. B., Swainston, N., Pir, P., & Oliver, S. G. (2015). Membrane transporter engineering in industrial biotechnology and whole cell biocatalysis. Trends in Biotechnology, 33, 237–246.
Kell, D. B., & Westerhoff, H. V. (1986). Metabolic control-theory—Its role in microbiology and biotechnology. Fems Microbiology Letters, 39, 305–320.
Kiefer, P., Heinzle, E., Zelder, O., & Wittmann, C. (2004). Comparative metabolic flux analysis of lysine-producing Corynebacterium glutamicum cultured on glucose or fructose. Applied and Environmental Microbiology, 70, 229–239. https://doi.org/10.1128/AEM.70.1.229-239.2004.
Kinclova-Zimmermannova, O., & Sychrova, H. (2007). Plasma-membrane Cnh1 Na/H antiporter regulates potassium homeostasis in Candida albicans. Microbiology, 153, 2603–2612. https://doi.org/10.1099/mic.0.2007/008011-0.
Konings, W. N., Poolman, B., & Driessen, A. M. (1992). Can the excretion of metabolites by bacteria be manipulated? FEMS Microbiology Letters, 88:, 93–108. https://doi.org/10.1111/j.1574-6968.1992.tb04959.x.
Krämer, R. (1994). Secretion of amino acids by bacteria: Physiology and mechanism. FEMS Microbiology Reviews, 13, 75–94. https://doi.org/10.1111/j.1574-6976.1994.tb00036.x.
Krämer, R. (1996). Analysis and modeling of substrate uptake and product release by prokaryotic and eukaryotic cells. Advances in Biochemical Engineering and Biotechnology, 54, 31–74.
Krämer, R. (2004). Production of amino acids: Physiological and genetic approaches. Food Biotechnology, 18, 171–216. https://doi.org/10.1081/FBT-200025664.
Kubicek, C. P. (1987). The role of the citric acid cycle in fungal organic acid fermentations. Biochemical Society Symposium, 54, 113–126.
Lamark, T., Styrvold, O. B., & Strøm, A. R. (1992). Efflux of choline and glycine betaine from osmoregulating cells of Escherichia coli. FEMS Microbiology Letters, 96, 149–154. https://doi.org/10.1016/0378-1097(92)90395-5.
Lee, D., Smallbone, K., Dunn, W.B., Murabito, E., Winder, C.L., Kell, D.B., et al. (2012). Improving metabolic flux predictions using absolute gene expression data. BMC Systems Biology, 6, 73.
Legiša, M., & Mattey, M. (2007). Changes in primary metabolism leading to citric acid overflow in Aspergillus niger. Biotechnology Letters, 29, 181–190. https://doi.org/10.1007/s10529-006-9235-z.
Lepore, B. W., Indic, M., Pham, H., Hearn, E. M., Patel, D. R., & van den Berg, B. (2011). Ligand-gated diffusion across the bacterial outer membrane. Proceedings of the National Academy of Sciences of the United States of America, 108, 10121–10126.
Liang, L., Liu, R., Li, F., Wu, M., Chen, K., Ma, J., et al. (2013). Repetitive succinic acid production from lignocellulose hydrolysates by enhancement of atp supply in metabolically engineered Escherichia coli. Bio resource Technology, 143, 405–412. https://doi.org/10.1016/j.biortech.2013.06.031.
Liu, J. Y., Miller, P. F., Willard, J., & Olson, E. R. (1999). Functional and biochemical characterization of Escherichia coli sugar efflux transporters. Journal of Biological Chemistry, 274, 22977–22984. https://doi.org/10.1074/jbc.274.33.22977.
Livshits, V. A., Zakataeva, N. P., Aleshin, V. V., & Vitushkina, M. V. (2003). Identification and characterization of the new gene rhta involved in threonine and homoserine efflux in Escherichia coli. Research in Microbiology, 154, 123–135. https://doi.org/10.1016/S0923-2508(03)00036-6.
Ljungdahl, P. O., & Daignan-Fornier, B. (2012). Regulation of amino acid, nucleotide, and phosphate metabolism in Saccharomyces cerevisiae. Genetics, 190, 885–929. https://doi.org/10.1534/genetics.111.133306.
Magasanik, B., & Kaiser, C. A. (2002). Nitrogen regulation in Saccharomyces cerevisiae. Gene, 290, 1–18. https://doi.org/10.1016/S0378-1119(02)00558-9.
Martín, J. F., Casqueiro, J., & Liras, P. (2005). Secretion systems for secondary metabolites: How producer cells send out messages of intercellular communication. Current Opinion on Microbiology, 8, 282–293. https://doi.org/10.1016/j.mib.2005.04.009.
Mattey, M. (1992). The production of organic acids. Critical Reviews on Biotechnology, 12, 87–132. https://doi.org/10.3109/07388559209069189.
Mccloskey, D., Palsson, B. O., & Feist, A. M. (2013). Basic and applied uses of genome-scale metabolic network reconstructions of Escherichia coli. Molecular System Biology, 9, 661. https://doi.org/10.1038/msb.2013.18.
Mitsuhashi, S. (2014). Current topics in the biotechnological production of essential amino acids, functional amino acids, and dipeptides. Current Opinion in Biotechnology, 26, 38–44.
Mo, M. L., Palsson, B., & Herrgård, M. J. (2009). Connecting extracellular metabolomic measurements to intracellular flux states in yeast. BMC System Biology. https://doi.org/10.1186/1752-0509-3-37.
Molenaar, D., Van Berlo, R., De Ridder, D., & Teusink, B. (2009). Shifts in growth strategies reflect tradeoffs in cellular economics. Molecular System Biology, 5, 323. https://doi.org/10.1038/msb.2009.82.
Moreno-Sanchez, R., Saavedra, E., Rodriguez-Enriquez, S., & Olin-Sandoval, V. (2008). Metabolic control analysis: A tool for designing strategies to manipulate metabolic pathways. Journal of Biomedicine and Biotechnology, 2008: 597913.
Nakamura, J., Hirano, S., Ito, H., & Wachi, M. (2007). Mutations of the Corynebacterium glutamicum ncgl1221 gene, encoding a mechanosensitive channel homolog, induce l-glutamic acid production. Applied and Environmental Microbiology, 73, 4491–4498. https://doi.org/10.1128/AEM.02446-06.
Nehls, U., Mikolajewski, S., Magel, E., & Hampp, R. (2001). Carbohydrate metabolism in ectomycorrhizas: Gene expression, monosaccharide transport and metabolic control. New Phytologist, 150, 533–541. https://doi.org/10.1046/j.1469-8137.2001.00141.x.
Neijssel, O. M., & Tempest, D. W. (1975). Production of gluconic acid and 2 ketogluconic acid by Klebsiella aerogenes NCTC 418. Archives in Microbiology, 105, 183–185.
Neijssel, O. M., & Tempest, D. W. (1976). Role of energy-spilling reactions in growth of klebsiella-aerogenes NCTC-418 in aerobic chemostat culture. Archives of Microbiology, 110, 305–311.
Netik, A., Torres, N. V., Riol, J. M., & Kubicek, C. P. (1997). Uptake and export of citric acid by Aspergillus niger is reciprocally regulated by manganese ions. BBA-Biomembranes, 1326, 287–294. https://doi.org/10.1016/S0005-2736(97)00032-1.
Nikaido, H. (1993). Transport across the bacterial outer-membrane. Journal of Bioenergetics and Biomembranes, 25, 581–589.
Notebaart, R. A., Szappanos, B., Kintses, B., Pál, F., Györkei, Á, & Bogos, B., et al. (2014). Network-level architecture and the evolutionary potential of underground metabolism. Proceedings of National Academy of Science of the United States of America 111, 11762–11767. https://doi.org/10.1073/pnas.1406102111.
Orth, J. D., Conrad, T. M., Na, J., Lerman, J. A., Nam, H., Feist, A. M., et al. (2011). A comprehensive genome-scale reconstruction of Escherichia coli metabolism-2011. Molecular System Biology, 7, 535. https://doi.org/10.1038/msb.2011.65.
Orth, J. D., Thiele, I., & Palsson, B. O. (2010). What is flux balance analysis? Nature Biotechnology, 28, 245–248. https://doi.org/10.1038/nbt.1614.
Paczia, N., Nilgen, A., Lehmann, T., Gätgens, J., Wiechert, W., & Noack, S. (2012). Extensive exometabolome analysis reveals extended overflow metabolism in various microorganisms. Microbial Cell Factories, 11, 122. https://doi.org/10.1186/1475-2859-11-122.
Perez-Garcia, O., Villas-Boas, S. G., & Singhal, N. (2014). A method to calibrate metabolic network models with experimental datasets. 8th International Conference on Practical Applications of Computational Biology and Bioinformatics, PACCB 2014, 294, 183–190. https://doi.org/10.1007/978-3-319-07581-5_22.
Pinu, F. R., & Villas-Boas, S. G. (2017). Extracellular microbial metabolomics: The state of the art. Metabolites, 7, 3.
Ponomarova, O., & Patil, K. R. (2015). Metabolic interactions in microbial communities: Untangling the Gordian knot. Current Opinion in Microbiology, 27, 37–44.
Poole, K. (2004). Efflux-mediated multiresistance in gram-negative bacteria. Clinical Microbiology and Infection, 10, 12–26. https://doi.org/10.1111/j.1469-0691.2004.00763.x.
Pronk, J. T., Steensma, H. Y., & Van Dijken, J. P. (1996). Pyruvate metabolism in Saccharomyces cerevisiae. Yeast, 12, 1607–1633. https://doi.org/10.1002/(SICI)1097-0061(199612)12:16<1607::AID-YEA70>3.0.CO;2-4
Rancourt, D. E., Stephenson, J. T., & Vickell, G. A. (1984). Proline excretion by Escherichia coli K12. Biotechnology and Bioengineering, 26, 74–80. https://doi.org/10.1002/bit.260260114.
Reaves, M. L., Young, B. D., Hosios, A. M., Xu, Y. F., & Rabinowitz, J. D. (2013). Pyrimidine homeostasis is accomplished by directed overflow metabolism. Nature, 500, 237–241. https://doi.org/10.1038/nature12445.
Roberts, S. B., Gowen, C. M., Brooks, J. P., & Fong, S. S. (2010). Genome-scale metabolic analysis of Clostridium thermocellum for bioethanol production. BMC System Biology, 4, 31. https://doi.org/10.1186/1752-0509-4-31.
Saier, M. H., Jr. (2000). A functional-phylogenetic classification system for transmembrane solute transporters. Microbiology and Molecular Biology Reviews, 64, 354–411. https://doi.org/10.1128/MMBR.64.2.354-411.2000.
Saier, M. H., Jr., Tran, C. V., & Barabote, R. D. (2006). TCDB: The transporter classification database for membrane transport protein analyses and information. Nucleic Acids Research, 34, D181–D186. https://doi.org/10.1093/nar/gkj001.
Schink, B. (2002). Synergistic interactions in the microbial world. Antonie Van Leeuwenhoek International Journal of General and Molecular Microbiology, 81, 257–261.
Schweikhard, E. S., & Ziegler, C. M. (2012). Amino acid secondary transporters: Toward a common transport mechanism. Current Topics in Membranes, 70, 1–28. https://doi.org/10.1016/B978-0-12-394316-3.00001-6.
Segrè, D., Vitkup, D., & Church, G. M. (2002). Analysis of optimality in natural and perturbed metabolic networks. Proceedings of National Academy of Science of the United States of America, 99, 15112–15117. https://doi.org/10.1073/pnas.232349399.
Segura, A., Molina, L., Fillet, S., Krell, T., Bernal, P., Muñoz-Rojas, J., et al. (2012). Solvent tolerance in gram-negative bacteria. Current Opinion in Biotechnology, 23, 415–421. https://doi.org/10.1016/j.copbio.2011.11.015.
Shankaranand, V. S., & Lonsane, B. K. (1994). Ability of Aspergillus niger to tolerate metal ions and minerals in a solid-state fermentation system for the production of citric acid. Process Biochemistry, 29, 29–37. https://doi.org/10.1016/0032-9592(94)80056-1.
Shinfuku, Y., Sorpitiporn, N., Sono, M., Furusawa, C. T. H., & Shimizu, H. (2009). Development and experimental verification of a genome-scale metabolic model for Corynebacterium glutamicum. Microbial Cell Factories, 8, 43. https://doi.org/10.1186/1475-2859-8-43.
Shlykov, M. A., Zheng, W. H., Wang, E., Nguyen, J. D., & Saier, M. H., Jr. (2013). Transmembrane molecular transporters facilitating export of molecules from cells and organelles. In E. W. Yu, Q. Zhang & M. H. Brown (Eds.), Microbial efflux pumps: Current research. Norfolk: Caister Academic Press.
Simic, P., Sahm, H., & Eggeling, L. (2001). L-threonine export: Use of peptides to identify a new translocator from Corynebactedum glutamicum. Journal of Bacteriology, 183, 5317–5324. https://doi.org/10.1128/JB.183.18.5317-5324.2001.
Smith, D., Artursson, P., Avdeef, A., Di, L., Ecker, G.F., Faller, B., et al. (2014). Passive lipoidal diffusion and carrier-mediated cell uptake are both important mechanisms of membrane permeation in drug disposition. Molecular Pharmaceutics, 11, 1727–1738.
Soccol, C. R., Vandenberghe, L. P. S., Rodrigues, C., & Pandey, A. (2006). New perspectives for citric acid production and application. Food Technology and Biotechnology, 44, 141–149.
Sonenshein, A. L. (2007). Control of key metabolic intersections in Bacillus subtilis. Nature Reviews Microbiology, 5, 917–927. https://doi.org/10.1038/nrmicro1772.
Stäbler, N., Oikawa, T., Bott, M., & Eggeling, L. (2011). Corynebacterium glutamicum as a host for synthesis and export of d-amino acids. Journal of Bacteriology, 193,, 1702–1709. https://doi.org/10.1128/JB.01295-10.
Tempest, D. W., & Neijssel, O. M. (1979). Overflow metabolism in aerobic micro-organisms. Biochemical Society Transactions, 7, 82–85.
Trötschel, C., Deutenberg, D., Bathe, B., Burkovski, A., & Krämer, R. (2005). Characterization of methionine export in Corynebacterium glutamicum. Journal of Bacteriology, 187, 3786–3794. https://doi.org/10.1128/JB.187.11.3786-3794.2005.
Valgepea, K., Adamberg, K., Nahku, R., Lahtvee, P., Arike, L., & Vilu, R. (2010). Systems biology approach reveals that overflow metabolism of acetate in Escherichia coli is triggered by carbon catabolite repression of acetyl-CoA synthetase. BMC System Biology. https://doi.org/10.1186/1752-0509-4-166.
van Berlo, R. J. P., de Ridder, D., Daran, J. M., Daran-Lapujade, P. A. S., Teusink, B., & Reinders, M. J. T. (2011). Predicting metabolic fluxes using gene expression differences as constraints. IEEE-ACM Transactions on Computational Biology and Bioinformatics, 8, 206–216.
Van Dyk, T. K. (2008). Bacterial efflux transport in biotechnology. Advanced Applied Microbiology, 63, 231–247. https://doi.org/10.1016/S0065-2164(07)00006-8.
Van Dyk, T. K., Templeton, L. J., Cantera, K. A., Sharpe, P. L., & Sariaslani, F. S. (2004). Characterization of the Escherichia coli AAEAB efflux pump: A metabolic relief valve? Journal of Bacteriology, 186, 7196–7204. https://doi.org/10.1128/JB.186.21.7196-7204.2004.
Van Hoek, M. J. A., & Merks, R. M. H. (2012). Redox balance is key to explaining full vs. Partial switching to low-yield metabolism. BMC System Biology, 6, 22. https://doi.org/10.1186/1752-0509-6-22.
Velasco, I., Tenreiro, S., Calderon, I. L., & André, B. (2004). Saccharomyces cerevisiae Aqr1 is an internal-membrane transporter involved in excretion of amino acids. Eukaryotic Cell, 3, 1492–1503. https://doi.org/10.1128/EC.3.6.1492-1503.2004.
Vemuri, G. N., Altman, E., Sangurdekar, D. P., Khodursky, A. B., & Eiteman, M. A. (2006a). Overflow metabolism in Escherichia coli during steady-state growth: Transcriptional regulation and effect of the redox ratio. Applied and Environmental Microbiology, 72, 3653–3661. https://doi.org/10.1128/AEM.72.5.3653-3661.2006.
Vemuri, G. N., Eiteman, M. A., & Altman, E. (2006b). Increased recombinant protein production in Escherichia coli strains with overexpressed water-forming NADH oxidase and a deleted ARCA regulatory protein. Biotechnology and Bioengineering, 94, 538–542. https://doi.org/10.1002/bit.20853.
Verkhovskaya, M. L., Barquera, B., & Wikström, M. (2001). Deletion of one of two Escherichia coli genes encoding putative Na/H exchangers (ycgO) perturbs cytoplasmic alkali cation balance at low osmolarity. Microbiology, 147, 3005–3013. https://doi.org/10.1099/00221287-147-11-3005.
Villas-Bôas, S. G., Moon, C. D., Noel, S., Hussein, H., Kelly, W. J., et al. (2008). Phenotypic characterization of transposon-inserted mutants of Clostridium proteoclasticum B316T using extracellular metabolomics. Journal of Bacteriology, 134, 55–63. https://doi.org/10.1016/j.jbiotec.2008.01.010.
Villas-Bôas, S. G., Noel, S., Lane, G. A., Attwood, G., & Cookson, A. (2006). Extracellular metabolomics: A metabolic footprinting approach to assess fiber degradation in complex media. Analytical Biochemistry, 349, 297–305. https://doi.org/10.1016/j.ab.2005.11.019.
Vrljic, M., Sahm, H., & Eggeling, L. (1996). A new type of transporter with a new type of cellular function: L-lysine export from Corynebacterium glutamicum. Molecular Microbiology, 22, 815–826. https://doi.org/10.1046/j.1365-2958.1996.01527.x.
Wachi, M. (2013). Amino acid exporters in Corynebacterium glutamicum. In H. Yukawa & M. Inui (Eds.), Corynebacterium glutamicum. Berlin: Springer.
Walter, A., & Gutknecht, J. (1984). Monocarboxylic acid permeation through lipid bilayer membranes. Journal of Membrane Biology, 77, 255–264. https://doi.org/10.1007/BF01870573.
Wendisch, V. F., Bott, M., & Eikmanns, B. J. (2006). Metabolic engineering of Escherichia coli and Corynebacterium glutamicum for biotechnological production of organic acids and amino acids. Current Opinion in Microbiology, 9, 268–274. https://doi.org/10.1016/j.mib.2006.03.001.
West, I. C. (1997). Ligand conduction and the gated-pore mechanism of transmembrane transport. BBA-Review Biomembranes, 1331, 213–234. https://doi.org/10.1016/S0304-4157(97)00007-5.
Wiebe, M. G., Rintala, E., Tamminen, A., Simolin, H., Salusjärvi, L., & Toivari, M., et al. (2008). Central carbon metabolism of Saccharomyces cerevisiae in anaerobic, oxygen-limited and fully aerobic steady-state conditions and following a shift to anaerobic conditions. FEMS Yeast Research, 8, 140–154. https://doi.org/10.1111/j.1567-1364.2007.00234.x.
Willemsen, A. M., Hendrickx, D. M., Hoefsloot, H. C. J., Hendriks, M. M. W. B., Wahl, S. A., Teusink, B., et al. (2015). METDFBA: Incorporating time-resolved metabolomics measurements into dynamic flux balance analysis. Molecular Biosystems, 11, 137–145. https://doi.org/10.1039/c4mb00510d.
Acknowledgements
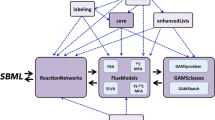
The authors are thankful to Mia Jüllig for assistance with Fig. 2. Callaghan Innovation and Bioresource Processing Alliance provided PhD stipends for James Daniell and Ninna Granucci respectively.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Pinu, F.R., Granucci, N., Daniell, J. et al. Metabolite secretion in microorganisms: the theory of metabolic overflow put to the test. Metabolomics 14, 43 (2018). https://doi.org/10.1007/s11306-018-1339-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11306-018-1339-7