Abstract
Objectives
Diabetes mellitus (DM) is associated with a broad range of complications, such as retinopathy, nephropathy, neuropathy, and cardiovascular disease. Therefore, predicting DM from head and neck images is a challenge for clinicians. The purpose of this study was to assess the mandibular condylar bone marrow in DM patients using computed tomography (CT) texture analysis.
Methods
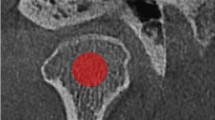
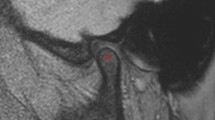
This retrospective study included 16 DM and age and sex matched 16 control patients (11 men, 5 women; mean age, 56.8 ± 14.4 years; range 31–78 years). Patients with Type I DM, prior history of taking bisphosphonates, osteoarthritis of the temporomandibular joint, and CT images with metal artifacts were excluded from this study. Bilateral mandibular condylar bone marrow was manually contoured on axial CT images. The presence or absence of DM is the primary predictor variable. Texture features of the region of interest was the outcome variable, that were analyzed using an open-access software, MaZda Ver.3.3. For each group, 20 features out of 279 parameters were selected with Fisher, probability of error and average correlation coefficient methods in MaZda. Bivariate statistics were computed with the Mann–Whitney U test and the P value was set at .05.
Results
One histogram feature, 15 Gy level co-occurrence matrix features, and four gray level run length matrix features showed differences between the DM patients and non-DM patients (P < 0.05).
Conclusions
Several texture features of the condyle demonstrated differences between the DM and non-DM patients. CT texture analysis may potentially detect DM from the condylar bone marrow.


Similar content being viewed by others
References
American Diabetes Association. Diagnosis and classification of diabetes mellitus. Diabetes Care. 2014;37(Supplement 1):S81–90.
Verhulst MJL, Loos BG, Gerdes VEA, Teeuw WJ. Evaluating all potential oral complications of diabetes mellitus. Front Endocrinol (Lausanne). 2019;10:56.
Bascones-Martínez A, González-Febles J, Sanz-Esporrín J. Diabetes and periodontal disease. Review of the literature. Am J Dent. 2014;27(2):63–7.
Mauri-Obradors E, Estrugo-Devesa A, Jané-Salas E, Viñas M, López-López J. Oral manifestations of diabetes mellitus. A systematic review. Med Oral Patol Oral Cir Bucal. 2017;22(5):e586–94.
Collin HL, Niskanen L, Uusitupa M, Töyry J, Collin P, Koivisto AM, Viinamäki H, Meurman JH. Oral symptoms and signs in elderly patients with type 2 diabetes mellitus. A focus on diabetic neuropathy. Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 2000;90(3):299–305.
Uemura M, Toda I, Kawashima W, Yoshimoto G, Fang YR, Xu YJ, Liu Y, Zhang L, Takemura A. Morphological study of the articular disc and capillary of the retrodiscal tissue in a type 2 spontaneous diabetes mellitus rat model. Okajimas Folia Anat Jpn. 2016;92(3–4):53–9.
Lambin P, Rios-Velazquez E, Leijenaar R, Carvalho S, van Stiphout RG, Granton P, Zegers CM, Gillies R, Boellard R, Dekker A, Aerts HJ. Radiomics: extracting more information from medical images using advanced feature analysis. Eur J Cancer. 2012;48:441–6.
Avanzo M, Stancanello J, El Naqa I. Beyond imaging: the promise of radiomics. Phys Med. 2017;38:122–39.
Valdora F, Houssami N, Rossi F, Calabrese M, Tagliafico AS. Rapid review: radiomics and breast cancer. Breast Cancer Res Treat. 2018;169:217–29.
Gao J, Jiang Q, Zhou B, Chen D. Convolutional neural networks for computer-aided detection or diagnosis in medical image analysis: an overview. Math Biosci Eng. 2019;16:6536–61.
Gentillon H, Stefańczyk L, Strzelecki M, Respondek-Liberska M. Parameter set for computer-assisted texture analysis of fetal brain. BMC Res Notes. 2016;9:496.
Lazli L, Boukadoum M, Ait MO. Computer-aided diagnosis system of Alzheimer’s disease based on multimodal fusion: tissue quantification based on the hybrid fuzzy-genetic-possibilistic model and discriminative classification based on the SVDD model. Brain Sci. 2019;9:e289.
Liu R, Li H, Liang F, Yao L, Liu J, Li M, Cao L, Song B. Diagnostic accuracy of different computer-aided diagnostic systems for malignant and benign thyroid nodules classification in ultrasound images: a systematic review and meta-analysis protocol. Medicine (Baltimore). 2019;98:e16227.
Mosquera-Lopez C, Agaian S, Velez-Hoyos A, Thompson I. Computer-aided prostate cancer diagnosis from digitized histopathology: a review on texture-based systems. IEEE Rev Biomed Eng. 2015;8:98–113.
Barry B, Buch K, Soto JA, Jara H, Nakhmani A, Anderson SW. Quantifying liver fibrosis through the application of texture analysis to diffusion weighted imaging. Magn Reson Imaging. 2014;32:84–90.
Alegro CM, Silva AV, Bando SY, et al. Texture analysis of high resolution MRI allows discrimination between febrile and afebrile initial precipitating injury in mesial temporal sclerosis. Magn Reson Med. 2012;68:1647–53.
Fujimoto K, Tonan T, Azuma S, et al. Evaluation of the mean and entropy of apparent diffusion coefficient values in chronic hepatitis C: correlation with pathologic fibrosis stage and inflammatory activity grade. Radiology. 2011;258:739–48.
Jirák D, Dezortová M, Taimr P, Hájek M. Texture analysis of human liver. J Magn Reson Imaging. 2002;15:68–74.
Mayerhoefer ME, Stelzeneder D, Bachbauer W, et al. Quantitative analysis of lumbar intervertebral disc abnormalities at 3.0 Tesla: value of T(2) texture features and geometric parameters. NMR Biomed. 2012;25:866–72.
Risse F, Pesic J, Young S, Olsson LE. A texture analysis approach to quantify ventilation changes in hyperpolarised 3He MRI of the rat lung in an asthma model. NMR Biomed. 2012;25:131–41.
Buch K, Fujita A, Li B, Kawashima Y, Qureshi MM, Sakai O. Using texture analysis to determine human papillomavirus status of oropharyngeal squamous cell carcinomas on CT. AJNR Am J Neuroradiol. 2015;36:1343–8.
Fujita A, Buch K, Li B, Kawashima Y, Qureshi MM, Sakai O. Difference between HPV-positive and HPV-negative non-oropharyngeal head and neck cancer: texture analysis features on CT. J Comput Assist Tomogr. 2016;40:43–7.
Kuno H, Qureshi MM, Chapman MN, et al. CT texture analysis potentially predicts local failure in head and neck squamous cell carcinoma treated with chemoradiotherapy. AJNR Am J Neuroradiol. 2017;38:2334–40.
American Diabetes Association. 2. Classification and diagnosis of diabetes: standards of medical care in diabetes—2020. Diabetes Care. 2020;43(Suppl 1):S14–31.
Szczypinski P, Strzelecki M, Materka A. MaZda—a Software for texture analysis. In: Proc. of ISITC 2007, November 23–23, 2007, Republic of Korea, p. 245–249.
Strzelecki M, Szczypinski P, Materka A, Klepaczko A. A software tool for automatic classification and segmentation of 2D/3D medical images. Nucl Instrum Methods Phy Res A. 2013;702:137–40.
Szczypinski P, Strzelecki M, Materka A, Klepaczko A. MaZda-A software package for image texture analysis. Comput Methods Programs Biomed. 2009;94:66–76.
Ito K, Muraoka H, Hirahara N, Tokunaga S, Sawada E, Kaneda T. Quantitative evaluation of nasopalatine duct using CT texture analysis: comparison of normal and pathological conditions of nasopalatine duct. Dent Radiol. 2020;60(1):5–11.
Mohanaiah P, Sathyanarayana P, GuruKumar L. Image texture feature extraction using GLCM approach. Int J Sci ResPubl. 2013;3:2250–3153.
Sharma EK, Priyanka E, Kalsh EK, et al. GLCM and its Features. Int J Adv Res Electron Commun Eng. 2015;4:2180–2.
Tsai A, Buch K, Fujita A, et al. Using CT texture analysis to differentiate between nasopharyngeal carcinoma and age-matched adenoid controls. Eur J Radiol. 2018;108:208–14.
Pang H, Chen T, Wang X, Chang Z, Shao S, Zhao J. Quantitative evaluation methods of skin condition based on texture feature parameters. Saudi J Biol Sci. 2017;24(3):514–8.
Takahara N, Nakagawa S, Sumikura K, Kabasawa Y, Sakamoto I, Harada H. Association of temporomandibular joint pain according to magnetic resonance imaging findings in temporomandibular disorder patients. J Oral Maxillofac Surg. 2017;75(9):1848–55.
Fave X, Mackin D, Yang J, et al. Can radiomics features be reproducibly measured from CBCT images for patients with non-small cell lung cancer? Med Phys. 2015;42(12):6784–97.
Acknowledgements
We would like to thank Editage (www.editage.com) for English language editing.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethics approval
This study was approved by the Ethics Committee of the University School of Dentistry (No. EC15-12-009-1).
Informed consent
The requirement to obtain written informed consent was waived for this retrospective study. All procedures followed the guidelines of the Declaration of Helsinki, Ethical Principles for Medical Research Involving Human Subjects.
Animal statements
This article does not contain any studies with animal subjects performed by the any of the authors.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Ito, K., Muraoka, H., Hirahara, N. et al. Computed tomography texture analysis of mandibular condylar bone marrow in diabetes mellitus patients. Oral Radiol 37, 693–699 (2021). https://doi.org/10.1007/s11282-021-00517-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11282-021-00517-7