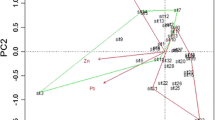
Abstract
Little is known about the microbiota that form symbioses with wetland plants. We describe how endophytes from leaves and roots of baldcypress trees changed along a nutrient gradient and differed in wastewater treated versus untreated wetlands. Cultured bacterial and fungal endophytes were most abundant and diverse in baldcypress roots (compared to leaves) for both treated and untreated wetlands. Bacterial endophyte abundance increased with increasing distance from the wastewater outfall pipe, while fungal endophytes decreased with increasing distance from the wastewater outfall pipe—showing a greater abundance of fungi where nutrients were greatest. Bacterial endophyte abundance and species richness were greater in the wastewater-treatment compared to untreated wetlands, but diversity metrics suggested this was due to incomplete sampling at some sites. Community composition of endophytes in the treated wetland differed from some but not all of the communities observed in untreated wetlands. Since baldcypress is a key restoration species for declining swamps in the southeastern US, our descriptive work provides a foundation for future studies to understand the functional roles of plant-microbial interactions and community patterns.




Similar content being viewed by others
References
Ali S, Charles TC, Glick BR (2014) Amelioration of high salinity stress damage by plant growth-promoting bacterial endophytes that contain ACC deaminase. Plant Physiol Biochem 80:160–167. https://doi.org/10.1016/J.Plaphy.2014.04.003
Arnold AE, Lutzoni F (2007) Diversity and host range of foliar fungal endophytes: are tropical leaves biodiversity hotspots? Ecology 88:541–549. https://doi.org/10.1890/05-1459
Arnold AE, Maynard Z, Gilbert GS, Coley PD, Kursar TA (2000) Are tropical fungal endophytes hyperdiverse? Ecol Lett 3:267–274. https://doi.org/10.1046/j.1461-0248.2000.00159.x
Bodker JE, Turner RE, Tweel A, Schulz C, Swarzenski C (2015) Nutrient-enhanced decomposition of plant biomass in a freshwater wetland. Aquat Bot 127:44–52. https://doi.org/10.1016/j.aquabot.2015.08.001
Boesch DF, Josselyn MN, Mehta AJ, Morris JT, Nuttle WK, Simenstad CA, Swift DJ (1994) Scientific assessment of coastal wetland loss, restoration and management in Louisiana. J Coastal Res 20:1–103
Bokulich NA et al (2018) Optimizing taxonomic classification of marker-gene amplicon sequences with QIIME 2’s q2-feature-classifier plugin. Microbiome 6:90
Camacho C, Coulouris G, Avagyan V, Ma N, Papadopoulos J, Bealer K, Madden TL (2009) BLAST+: architecture and applications. BMC Bioinform 10:421
Caporaso JG et al (2010) QIIME allows analysis of high-throughput community sequencing data. Nat Methods 7:335
Chao A, Jost L (2012) Coverage-based rarefaction and extrapolation: standardizing samples by completeness rather than size. Ecology 93:2533–2547
Chao A, Jost L (2015) Estimating diversity and entropy profiles via discovery rates of new species. Methods Ecol Evol 6:873–882
Chao A, Ma KH, Hsieh TC (2016) iNEXT (iNterpolation and EXTrapolation) Online: Software for Interpolation and Extrapolation of Species Diversity
Chowdhury SP, Hartmann A, Gao X, Borriss R (2015) Biocontrol mechanism by root-associated Bacillus amyloliquefaciens FZB42–a review. Front Microbiol 6:780
Clarke KR (1993) Non-parametric multivariate analyses of changes in community structure. Austral Ecol 18:117–143
Clarke KR, Gorley RN (2006) PRIMER v6: user manual/tutorial, 6th edn. PRIMER Ltd., Plymouth
Couvillion BR et al (2011) Land area change in coastal Louisiana from 1932 to 2010. US Geological Survey, Reston
Day JW, Shaffer GP, Britsch LD, Reed DJ, Hawes SR, Cahoon D (2000) Pattern and process of land loss in the Mississippi Delta: a spatial and temporal analysis of wetland habitat change. Estuaries 23:425–438. https://doi.org/10.2307/1353136
Day JW et al (2006) Effects of long-term municipal effluent discharge on the nutrient dynamics, productivity, and benthic community structure of a tidal freshwater forested wetland in Louisiana. Ecol Eng 27:242–257. https://doi.org/10.1016/j.ecoleng.2006.03.004
Day JW et al (2007) Restoration of the Mississippi delta: lessons from hurricanes Katrina and Rita. Science 315:1679–1684. https://doi.org/10.1126/science.1137030
Eriksson L, Hermens JL, Johansson E, Verhaar HJ, Wold S (1995) Multivariate analysis of aquatic toxicity data with PLS. Aquat Sci 57:217–241
Friesen ML, Porter SS, Stark SC, von Wettberg EJ, Sachs JL, Martinez-Romero E (2011) Microbially mediated plant functional traits. Annu Rev Ecol Evol Syst 42:23–46
Gibson A, Harper J (1985) Nitrate effect on nodulation of soybean by Bradyrhizobium japonicum. Crop Sci 25:497–501
Glöckner FO et al (2017) 25 years of serving the community with ribosomal RNA gene reference databases and tools. J Biotechnol 261:169–176
Hodge A, Campbell CD, Fitter AH (2001) An arbuscular mycorrhizal fungus accelerates decomposition and acquires nitrogen directly from organic material. Nature 413:297
Hoffman MT, Arnold AE (2010) Diverse bacteria inhabit living hyphae of phylogenetically diverse fungal endophytes. Appl Environ Microbiol 76:4063–4075. https://doi.org/10.1128/Aem.02928-09
Hunter R, Lane R, Day J, Lindsey J, Day J, Hunter M (2009) Nutrient removal and loading rate analysis of Louisiana forested wetlands assimilating treated municipal effluent. Environ Manage 44:865–873. https://doi.org/10.1007/s00267-009-9348-y
Hunter RG et al (2018) Using natural wetlands for municipal effluent assimilation: a half-century of experience for the Mississippi River Delta and surrounding environs. In: Nagabhatla N, Metcalfe CD (eds) Multifunctional wetlands. Springer, Cham, pp 15–81
Husband R, Herre EA, Turner SL, Gallery R, Young JPW (2002a) Molecular diversity of arbuscular mycorrhizal fungi and patterns of host association over time and space in a tropical forest. Mol Ecol 11:2669–2678. https://doi.org/10.1046/j.1365-294X.2002.01647.x
Husband R, Herre EA, Young JPW (2002b) Temporal variation in the arbuscular mycorrhizal communities colonising seedlings in a tropical forest. FEMS Microbiol Ecol 42:131–136. https://doi.org/10.1111/j.1574-6941.2002.tb01002.x
Ijaz A, Shabir G, Khan QM, Afzal M (2015) Enhanced remediation of sewage effluent by endophyte-assisted floating treatment wetlands. Ecol Eng 84:58–66
Imsande J (1986) Inhibition of nodule development in soybean by nitrate or reduced nitrogen. J Exp Bot 37:348–355
Jumpponen A, Mattson KG, Trappe JM (1998) Mycorrhizal functioning of Phialocephala fortinii with Pinus contorta on glacier forefront soil: interactions with soil nitrogen and organic matter. Mycorrhiza 7:261–265
Kandalepas D (2012) Effects of coastal dynamics on colonization of Louisiana wetland plants by fungal endophytes. Louisiana State University, Baton Rouge
Kandalepas D, Stevens KJ, Shaffer GP, Platt WJ (2010) How abundant are root-colonizing fungi in Southeastern Louisiana’s degraded marshes? Wetlands 30:189–199
Kandalepas D, Blum MJ, Van Bael SA (2015) Shifts in symbiotic endophyte communities of a foundational salt marsh grass following oil exposure from the Deepwater Horizon oil spill. PLoS ONE 10:e0122378. https://doi.org/10.1371/journal.pone.0122378
Kimbrough ER, Berlow ML, Van Bael SA (2019) Water level and salinity drive community structure of culturable baldcypress (Taxodiumdistichum) Endophytes in Southern Louisiana. Wetlands 39:329–335
Kõljalg U et al (2013) Towards a unified paradigm for sequence-based identification of fungi. Mol Ecol 22:5271–5277
Li J-y, Strobel G, Sidhu R, Hess W, Ford EJ (1996) Endophytic taxol-producing fungi from bald cypress, Taxodium distichum. Microbiology 142:2223–2226
Lundberg CJ, Shaffer GP, Wood WB, Day JW (2011) Growth rates of baldcypress (Taxodium distichum) seedlings in a treated effluent assimilation marsh. Ecol Eng 37:549–553. https://doi.org/10.1016/j.ecoleng.2010.11.012
Mandyam K, Jumpponen A (2005) Seeking the elusive function of the root-colonising dark septate endophytic fungi. Stud Mycol 5:173–189
Marques JM et al (2014) Plant age and genotype affect the bacterial community composition in the tuber rhizosphere of field-grown sweet potato plants. FEMS Microbiol Ecol 88:424–435. https://doi.org/10.1111/1574-6941.12313
Mayerhofer MS, Kernaghan G, Harper KA (2013) The effects of fungal root endophytes on plant growth: a meta-analysis. Mycorrhiza 23:119–128
Mugerwa TM, Saleeba J, McGee P (2013) A variety of melanised root-associated fungi from the Sydney basin form endophytic associations with Trifolium subterraneum. Fungal Ecol 6:70–82
Murray JD, Liu C-W, Chen Y, Miller AJ (2016) Nitrogen sensing in legumes. J Exp Bot 68(8):1919–1926
Newsham KK (2011) A meta-analysis of plant responses to dark septate root endophytes. New Phytol 190:783–793. https://doi.org/10.1111/j.1469-8137.2010.03611.x
Pedregosa F et al (2011) Scikit-learn: machine learning in Python. J Mach Learn Res 12:2825–2830
Peskan-Berghofer T et al (2004) Association of Piriformospora indica with Arabidopsis thaliana roots represents a novel system to study beneficial plant-microbe interactions and involves early plant protein modifications in the endoplasmic reticulum and at the plasma membrane. Physiol Plant 122:465–477. https://doi.org/10.1111/j.1399-3054.2004.00424.x
Petrini O, Carroll G (1981) Endophytic fungi in foliage of some Cupressaceae in Oregon. Can J Bot-Rev Can Bot 59:629–636. https://doi.org/10.1139/b81-089
Porras-Alfaro A, Bayman P (2011) Hidden fungi, emergent properties: endophytes and microbiomes. Annu Rev Phytopathol 49:291–315
Redman RS, Kim YO, Woodward CJDA, Greer C, Espino L, Doty SL, Rodriguez RJ (2011) Increased fitness of rice plants to abiotic stress via habitat adapted symbiosis: a strategy for mitigating impacts of climate change. PLoS ONE 6:e14823. https://doi.org/10.1371/journal.pone.0014823
Rehman K, Imran A, Amin I, Afzal M (2018) Inoculation with bacteria in floating treatment wetlands positively modulates the phytoremediation of oil field wastewater. J Hazard Mater 349:242–251
Rodriguez RJ, White JF, Arnold AE, Redman RS (2009) Fungal endophytes: diversity and functional roles. New Phytol 182:314–330. https://doi.org/10.1111/J.1469-8137.2009.02773.X
Saikkonen K, Faeth SH, Helander M, Sullivan TJ (1998) Fungal endophytes: a continuum of interactions with host plants. Annu Rev Ecol Syst 29:319–343
Saleem H, Arslan M, Rehman K, Tahseen R, Afzal M (2018) Phragmites australis—a helophytic grass—can establish successful partnership with phenol-degrading bacteria in a floating treatment wetland. Saudi J Biol Sci 26(6):1179–1186
Shaffer GP et al (2015) System response, nutria herbivory, and vegetation recovery of a wetland receiving secondarily-treated effluent in coastal Louisiana. Ecol Eng 79:120–131
Streeter J, Wong PP (1988) Inhibition of legume nodule formation and N2 fixation by nitrate. Crit Rev Plant Sci 7:1–23
Strickland ER (2015) Optimizing growth and survival of baldcypress (Taxodium distichum) and water tupelo (Nyssa aquatica) destined for restoration projects in nutrient-rich, freshwater areas. M. S. Southeastern Louisiana University, Hanmond
Syranidou E, Christofilopoulos S, Gkavrou G, Thijs S, Weyens N, Vangronsveld J, Kalogerakis N (2016) Exploitation of endophytic bacteria to enhance the phytoremediation potential of the wetland helophyte Juncus acutus. Front Microbiol 7:1016
R Team (2019) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. URL https://www.R-project.org/. R Foundation for Statistical Computing, Vienna, Austria
U’ren JM, Dalling JW, Gallery RE, Maddison DR, Davis EC, Gibson CM, Arnold AE (2009) Diversity and evolutionary origins of fungi associated with seeds of a Neotropical pioneer tree: a case study for analysing fungal environmental samples. Mycol Res 113:432–449. https://doi.org/10.1016/j.mycres.2008.11.015
Weishampel PA, Bedford BL (2006) Wetland dicots and monocots differ in colonization by arbuscular mycorrhizal fungi and dark septate endophytes. Mycorrhiza 16:495–502. https://doi.org/10.1007/s00572-006-0064-7
Weller M, Bossart J (2017) Insect community diversity tracks degradation and recovery of a wastewater assimilation marsh in southeast Louisiana. Wetlands 37:661–673
Wilson D (1995) Endophyte—the evolution of a term, and clarification of its use and definition. Oikos 73:274–276. https://doi.org/10.2307/3545919
Acknowledgements
For help in the lab and field we thank Mae Berlow, Kim Mighell, Peter Tellez, Kathalina Tran, Josh Lerner, Casey Gu, James Sebes, Emma Tower, Elaine Broussard, George Washburn, and Julie Whitbeck. Collections that occurred at Jean Lafitte National Historical Park and Preserve were authorized by permit # JELA-2014-SCI-0012.
Funding
Funds were provided by the School of Science and Engineering at Tulane University to S. Van Bael, as well as a Louisiana Board of Regents SURE (EPSCOR) award to J. Janowsky and a Tulane Honors College award to J. Janowsky.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11273_2019_9688_MOESM2_ESM.jpg
Supplementary material 2—Figure S1. Map of transects and reference trees at the treated site. Yellow circles indicate where the samples were collected. The reference area was outside of the area in which treated wastewater would have had contact. The labels 0, 200, 400 and 600 m refer to the distance from the wastewater outfall pipe. (JPEG 1555 kb)
11273_2019_9688_MOESM3_ESM.jpg
Supplementary material 3—Figure S2. Map of the five sample sites for baldcypress endophytes in southeastern Louisiana. Sample sites are designated with a yellow circle. (JPEG 1570 kb)
11273_2019_9688_MOESM4_ESM.jpg
Supplementary material 4—Figure S3. Species diversity estimates for fungal endophytes of baldcypress. (a) All endophytes at the Hammond site only, showing empirical species diversity for the Hill numbers 0 < q < 3. For example, species richness at q = 0 is 32 OTUs, with a value for Shannon diversity (q = 1) of 17.11 and a value for Simpson’s diversity (q = 2) of 8.51. (b) Hammond site only, showing the estimated species diversity for the Hill numbers 0 < q < 3. (c) A comparison of empirical species diversity using Hill numbers 0 < q < 3 for all of the sites in the study. Although the empirical observation of species richness (q = 0) makes the sample at Hammond appear more diverse than the other sites, the overlapping 95% confidence intervals for q > 0.5 illustrate that there is not a significant difference in species diversity among sites. All shaded areas represent 95% confidence intervals based on 100 bootstrap replications (JPEG 219 kb)
Rights and permissions
About this article
Cite this article
Janowsky, J., Kimbrough, E., Kandalepas, D. et al. Bacterial and fungal endophyte communities differ in trees of natural versus wastewater-treatment wetlands. Wetlands Ecol Manage 27, 711–723 (2019). https://doi.org/10.1007/s11273-019-09688-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-019-09688-x