Abstract
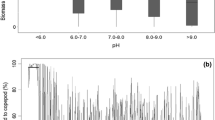
Microcrustacean species (Cladocera, Copepoda) as a significant component of the plankton assembly play diverse roles in floodplain ecosystems and cope with the heterogenic environment differently. Since alterations in the hydro regime are considered disturbance events for zooplankton development, we assessed whether and to what extent are Cladocera and Copepoda exposed to disturbances caused by hydro regime alterations in a floodplain lake. To evaluate the disturbance levels for individual microcrustacean assemblage in different water layers and between different hydrological phases, we applied the abundance/biomass comparison (ABC) method. Recorded microcrustacean assemblages highly depended on the hydrological state of the waterbody and diverged accordingly. Cladocera demonstrated to be more sensitive to change in specific environmental parameters like conductivity, temperature and transparency, while Copepoda exhibited no limitation by ecological variables. Although both groups were influenced by the bottom–up and top–down control, different disturbance intensity was recorded for the individual microcrustacean class. An undisturbed condition during isolation of the Lake allowed Cladocera to reach equilibrium while both microcrustacean groups exhibited moderate to high levels of disturbance following inundation. Regarding disturbance intensity, Cladocera expressed a weak distinction following hydro regime and water layer replacement. On the other hand, Copepoda disturbance levels notably depended on the position in the water column within a specific hydrological phase. Results of the ABC analyses gave a good representation of a real community dispersal in the studied Lake. However, more research is needed to re-evaluate here given levels of disturbance properly.





Similar content being viewed by others
References
Adamczuk M, Mieczan T (2013) Spatial distribution of brood-bearing females of limnetic species of Cladocera. CR Biol. https://doi.org/10.1016/j.crvi.2013.09.003
Amoros C (1984) Crustaces Cladoceres. Introduction Pratique a la Systematique des Organismes des Eaux Continentales Francaises. Université Claude Bernard, Lyon
APHA (American Public Health Association) (1992) Standard methods for the examination of water and wastewater. American Public Health Association, Washington, DC
Armstead MY, Bitzer-Creathers L, Wilson M (2016) The effects of elevated specific conductivity on the chronic toxicity of mining influenced streams using Ceriodaphnia dubia. PLoS ONE 11(11):e0165683. https://doi.org/10.1371/journal.pone.0165683
Baldwin DS, Mitchell AM (2000) The effects of drying and re-flooding on the sediment and soil nutrient dynamics of lowland river-floodplain systems: a synthesis. Regul River 16:457–467
Baranyi C, Hein T, Holarek C, Keckeis S, Schiemer F (2002) Zooplankton biomass and community structure in a Danube River floodplain system: effects of hydrology. Freshw Biol 47:473–482
Bianchi G, Hamukuaya H, Alvheim O (2001) On the dynamics of demersal fish assemblages off Namibia in the 1990s. S Afr J Marine Sci 23:419–428
Bollens SM, Frost BW (1989) Predator-induced diet vertical migration in a planktonic copepod. J Plankton Res 11(5):1047–1065
Borcherding J, Murawski S, Arndt H (2006) Population ecology, vertical migration and feeding of the Ponto-Caspian invader Hemimysis anomala in a gravel-pit lake connected to the River Rhine. Freshw Biol. https://doi.org/10.1111/j.1365-2427.2006.01666.x
Bottrell HH, Duncan A, Gliwicz ZM, Grygierek F, Herzig A, Hillbricht-Ilkowska A, Kurasawa H, Larsson P, Weglenska T (1976) A review of some problems in zooplankton production studies. Nor J Zool 24:419–456
Butorina LG (1971) On the diurnal migration of Polyphemus pediculus (L.). In: Vainshtain BA (ed) Biologia i Fiziologia Presnovodnykh Organizmov. Nauka, Leningrad, pp 94–105 (in Russian)
Butorina LG (1978) On the use of 14C for the investigation of horizontal migrations of Polyphemus pediculus (L.), Cladocera. Biologia vnutrennikh vod 37:42–45 (in Russian)
Clarke KR (1990) Comparison of dominance curves. J Exp Mar Biol Ecol. https://doi.org/10.1016/0022-0981(90)90181-B
Clarke KR, Warwick RM (2001) Change in marine communities: an approach to statistical analysis and interpretation. Primer-E Ltd, Plymouth
Coeck J, Vandelannoote A, Yseboodt R, Verheyen RF (1993) Use of the abundance/biomass method for comparison of fish communities in regulated and unregulated lowland rivers in Belgium. River Res Appl 8(1–2):73–82
Connell JH (1978) Diversity of tropical rainforests and coral reefs. Science 199:1304–1310
Cowles TJ, Olson RJ, Chisholm SW (1988) Food selection by copepods discrimination an the basis of food quality. Mar Biol 100:41–50
Davis SR, Brown AG, Dinnin MH (2007) Floodplain connectivity, disturbance and change: a palaeoenthomological investigation of floodplain ecology from south-west England. J Anim Ecol 76:276–288
de Carvalho MAJ (1984) On the feeding behaviour of Thermocyclops crassus. Crustaceana. Studies on Copepoda II. In Proceedings of the First International Conference on Copepoda, pp. 122–125
Dehghan-Madiseh S, Nabavi SMB, Ghofleh-Marammazi J, Jahani N, Koochaknejad E (2012) Application of abundance biomass curve in ecological health assessment of Khure-Mussa (Northwest of the Persian Gulf). J Persian Gulf 3(7):1–10
Dumont HJ, Van de Velde I, Dumont S (1975) The dry weight estimate of biomass in a selection of Cladocera, Copepoda and Rotifera from the plankton, periphyton and benthos of continental waters. Oecologia 19:75–97
Echevarria F, Carrilo FJ, Sanchez-Castillo P, Cruz-Pizarro L, Rodrigues J (1990) The sizeabundance distribution and taxonomic composition of plankton in a oligotrophic, high mountain lake (La Caldera, Sierra Nevada, Spain). J Plankton Res 12(2):415–422
Einsle U (1993) Crustacea, Copepoda, Calanoida und Cyclopoida. Gustav Fischer Verlag, Berlin
Farkas T, Herodek S (1967) Investigations of the fatty acid composition of fishes from Lake Balaton. Ann Inst Biol 34:139–146
Galir A, Palijan G (2012) Change in metazooplankton abundance in response to flood dynamics and trophic relations in Danubian floodplain lake (Kopački rit, Croatia). Pol J Ecol 60:777–787
Gama-Flores JL, Huidobro-Salasa ME, Sarma SSS, Nandini S, Zepeda-Mejia Gulatic RD (2015) Temperature and age affect the life history characteristics and fatty acid profiles of Moina macrocopa (Cladocera). J Therm Biol 53:135–142
Gliwicz ZM (1990) Why do cladocerans fail to control algal blooms? Hydrobiologia 200:83–97
Goździejewska A, Glińska-Lewczuk K, Obolewski K, Grzybowski M, Kujawa R, Lew S, Grabowska M (2016) Effects of lateral connectivity on zooplankton community structure in floodplain lakes. Hydrobiologia 774:7–21
Grosholz E, Galo E (2006) The influence of flood cycle and fish predation on invertebrate production on a restored California floodplain. Hydrobiologia 568:91–109
Härkönen L, Pekcan-Hekim Z, Hellén N, Ojala A, Horppila J (2014) Combined effects of turbulence and different predation regimes on zooplankton in highly colored water—implications for environmental change in lakes. PLoS ONE 9(11):e111942. https://doi.org/10.1371/journal.pone.0111942
Harper D (1992) Eutrophication of freshwaters: principles, problems and restoration. Chapman and Hall, London
Hein T, Baranyi C, Reckendorfer W, Schiemer F (2004) The impact of surface water exchange on the nutrient and particle dynamics in side-arms along the River Danube, Austria. Sci Total Environ 328:207–218
Hillbricht-Ilkowska A (1977) Trophic relations and energy flow in pelagic plankton. Polish Scientific Publishers, Warszawa
Ismail HA, Adnan AAM (2016) Zooplankton composition and abundance as indicators of eutrophication in two small man-made lakes. Trop Life Sci Res 27:31–38. https://doi.org/10.21315/tlsr2016.27.3.5
Jackson JM, Lenz PH (2016) Predator–prey interactions in the plankton: larval fish feeding on evasive copepods. Sci Rep 6:33585. https://doi.org/10.1038/srep33585
Jiang X, Yang W, Zhang L, Chen L, Niu Y (2014) Predation and cyanobacteria jointly facilitate competitive dominance of small-bodied cladocerans. J Plankton Res 36(4):956–965. https://doi.org/10.1093/plankt/fbu032
Keckeis S, Baranyi C, Hein T, Holarek C, Riedler P, Schiemer F (2003) The significance of zooplankton grazing in a floodplain system of the River Danube. J Plankton Res 25(3):243–253
Kirk KL, Gilbert JJ (1990) Suspended clay and the population dynamics of planktonic rotifers and cladocerans. Ecology 71:1741–1755
Kotta J, Orav-Kotta H, Pärnoja M (2013) Role of physical water properties and environmental disturbances on the diversity of coastal macrophyte and invertebrate communities in a brackish water ecosystem. WIT Trans Ecol Environ 169:77–88
Kulkarni VA, Naidu VS, Jagtap TG (2011) Marine ecological habitat: a case study on projected thermal power plant around Dharamtar creek, India. J Environ Biol 32:213–219
Lambshead PJD, Platt HM, Shaw KM (1983) The detection of differences among assemblages of marine benthic species based on an assessment of dominance and diversity. J Nat Hist 17:859–874
Lampert W, Loose CL (1992) Plankton towers: bridging the gap between laboratory and field experiments. Arch Hydrobiol 126:53–66
Lansac-Tôha FA, Thomaz SM, Lima AF, Roberto MC, Garcia AP (1995) Vertical distribution of some planktonic crustaceans in a varzea lake (Lake Pousada das Garças) of the floodplain of hight River Parana, MS, Brazil. J Ecol Environ Sci 21:67–78
Lawrence SG, Malley DF, Findlay WJ, MacIver MA, Delbaere IL (1987) Method for estimating dry weight of freshwater planktonic crustaceans from measures of lenght and shape. Can J Fish Aquat Sci 44:264–274
Lindenschmidt K-E, Chorus I (1998) The effect of water column mixing on phytoplankton succession, diversity and similarity. J Plankton Res 20:1927–1951
Lyu K, Zhang L, Gu L, Zhu X, Wilson AE, Yang Z (2017) Cladoceran offspring tolerance to toxic Microcystis is promoted by maternal warming. Environ Pollut. https://doi.org/10.1016/j.envpol.2017.04.095
Margaritoria F (1983) Cladoceri (Crustacea: Cladocera). Guide per il Reconoscimiento delle Specie Animali delle Acque Interne Italiane. Consiglio Nazionale delle Ricerche, Roma
Masson S, Desrosiers M, Pinel-Alloul B, Martel L (2010) Relating macroinvertebrate community structure to environmental characteristics and sediment contamination at the scale of the St. Lawrence River. Hydrobiologia 647:35–50
McCauley E (1984) The estimation of the abundance and biomass of zooplankton in samples. In: Downing JA, Rigler FH (eds) A manual on methods for the assessment of secondary productivity in fresh waters. Blackwell Scientific Publishers, Oxford, pp 228–265
Meire PM, Dereu J (1990) Use of the abundance/biomass comparison method for detecting environmental stress: some considerations based on intertidal macrozoobenthos and bird communites. J Appl Ecol 27:210–223
Mihaljević M, Stević F (2011) Cyanobacterial blooms in a temperate river-floodplain ecosystem: the importance of hydrological extremes. Aquat Ecol 45:335–349. https://doi.org/10.1007/s10452-011-9357-9
Mihaljević M, Stević F, Horvatić J, Hackenberger Kutuzović B (2009) Dual impact of the flood pulses on the phytoplankton assemblages in a Danubian floodplain lake (Kopacki Rit Nature Park, Croatia). Hydrobiologia 618:77–88
Mihaljević M, Špoljarić D, Stević F, Žuna Pfeiffer T (2013a) Assessment of flood-induced changes of phytoplankton along a river-floodplain system using the morpho-functional approach. Environ Monit Assess 185:8601–8619
Mihaljević M, Žuna Pfeiffer T, Stević F, Špoljarić D (2013b) Dynamics of phytoplankton and periphytic algae in a danubian floodplain lake: a comparative study under altered hydrological conditions. Fresen Environ Bull 22(9):2516–2523
Mihaljević M, Stević F, Špoljarić D, Žuna Pfeiffer T (2015) Spatial pattern of phytoplankton based on the morphology-based functional approach along a river-floodplain gradient. River Res Appl 31:228–238
Neto AJG, Silva LC, Saggio AA, Rocha O (2014) Zooplankton communities as eutrophication bioindicators in tropical reservoirs. Biota Neotrop 14(4):e20140018
Özen A, Bucak T, Tavşanoǧlu ÜN, Çakıroǧlu AÏ, Levi EE, Coppens J, Jeppesen E, Beklioǧlu M (2014) Water level and fish-mediated cascading effects on the microbial community in eutrophic warm shallow lakes: a mesocosm experiment. Hydrobiologia 740:25–35
Palijan G (2010) Different impact of the flood dynamics on the development of culturable planktonic and biofilm bacteria in the floodplain lake. Pol J Ecol 58:439–448
Pasternak AF, Mikheev VN, Wanzenböck J (2006) How plankton copepods avoid fish predation: from individual responses to variations of the life cycle. J Ichthyol 46:220–226
Penczak T, Kruk A (1999) Applicabiliy af the abundance/biomass comparison method for detecting human impacts on fish populations in the Pilica River, Poland. Fish Res 39:229–240
Perticarrari A, Arcifa MS, Rodrigues RA (2004) Diel vertical migration of copepods in a Brazilian lake: a mechanism for decreasing risk of Chaoborus predation? Braz J Biol 64(2):289–298
Picapedra PHS, Lansac-Tôha FA, Bialetzki A (2015) Diel vertical migration and spatial overlap between fish larvae and zooplankton in two tropical lakes, Brazil. Braz J Biol 75:352–361. https://doi.org/10.1590/1519-6984.13213
Pinel-Alloul B, Méthot G, Malinsky-Rushansky NZ (2004) A short-term study of vertical and horizontal distribution of zooplankton during thermal stratification in Lake Kinneret, Israel. Hydrobiologia 526:85–98
Reckendorfer W, Keckeis H, Winkler G, Schiemer F (1999) Zooplankton abundance in the river Danube, Austria: the significance of inshore retention. Freshw Biol 41:583–591
Richardson WB (1992) Microcrustacea in flowing water: experimental analysis of washout times and a field test. Freshw Biol 28:217–230. https://doi.org/10.1111/j.1365-2427.1992.tb00578.x
Schiemer F, Hein T (2007) The ecological significance of hydraulic retention zones. In: Wood PJ, Hannah DM, Sadler JP (eds) Hydroecology and ecohydrology: past, present and future. Wiley, Chichester, pp 405–420
Serizawa H, Amemiya T, Itoha K (2010) Effects of buoyancy, transparency and zooplankton feeding on surface maxima and deep maxima: comprehensive mathematical model for vertical distribution in cyanobacterial biomass. Ecol Model 221:2028–2037
Shannon CE (1948) A mathematical theory of communication. Bell Syst Tech J 27:379–423
Simões NR, Ribeiro SMMS, Sonoda SL (2011) Diversity and structure of microcrustacean assemblages (Cladocera and Copepoda) and limnological variability in perennial and intermittent pools in a semi-arid region, Bahia, Brazil, Iheringia. Série Zoologia 101(4):317–324
Simpson JH, Lucas NS, Powell B, Maberl SC (2014) Dissipation and mixing during the onset of stratification in a temperate lake, Windermere. Limnol Oceanogr 60:29–41
Sousa W (1984) The role of disturbance in natural communities. Annu Rev Ecol Evol Syst 15:353–391
Strickland JDH, Parsons TR (1968) A practical hand-book of seawater analysis. Fish Res Board Can Bull 167:1–310
Tadić D, Vidaček Ž (1999) Klimatske, hidrološke i pedološke značajke. In: Mihaljević M, Getz D, Tadić Z, Živanović B, Gucunski D, Topić J, Kalinović I, Mikuška J (eds) Kopački rit - Pregled istraživanja i bibliografija. Hrvatska akademija znanosti i umjetnosti, Zagreb, pp 23–28 (in Croatian)
ter Braak CJF, Šmilauer P (2002) CANOCO Reference Manual and CanoDraw for Windows User’s Guide: Software for Canonical Community Ordination (version 4.5). Microcomputer Power, Ithaca NY
Tockner K, Malard F, Ward JV (2000) An extension of the flood pulse concept. Hydrol Process 14:2861–2883
UNESCO (1966) Determinations of photosynthetic pigments in seawater. Report of SCOR—UNESCO
Urabe J (1991) Effect of food concentration on growth, reproduction and survivorship of Bosmina longirostris (Cladocera): an experimental study. Freshw Biol 25:1–8
Vadadi-Fülöp C (2009) Zooplankton (Cladocera, Copepoda) dynamics in the River Danube upstream and downstream of Budapest, Hungary. Opuscula Zoologica (Budapest) 40(2):87–98
Vadadi-Fülöp C (2013) Microcrustacean assemblages in a large river: on the importance of the flow regime. Hydrobiologia 702:129–140
Vidaković J, Bogut I (2007) Periphyton nematode assemblages in association with Myriophyllum spicatum L. in Lake Sakadaš, Croatia. Russ J Nematol 15:78–88
Vranovsky M (1995) The effects of current velocity upon the biomass of zooplankton in the River Danube side arms. Biologia 50:461–464
Warwick RM (1986) A new method for detecting pollution effects on marine macrobenthic communities. Mar Biol 92:557–562
Warwick RM, Clarke KR (1994) Relearning the ABC: taxonomic changes and abundance/biomass relationships in disturbed benthic communities. Mar Biol 118:739–744
Weigelhofer G, Preiner S, Funk A, Bondar-Kunze E, Hein T (2014) The hydrochemical response of small and shallow floodplain water bodies to temporary surface water connections with the main river. Freshw Biol. https://doi.org/10.1111/fwb.12532
Weithoff G, Walz N, Gaedke U (2001) The intermediate disturbance hypothesis—species diversity or functional diversity? J Plankton Res 23(10):1147–1155. https://doi.org/10.1093/plankt/23.10.1147
Wetzel RG (2001) Limnology: lake and river ecosystems. Academic press, San Diego
Wüest A, Piepke G, Van Senden DC (2000) Turbulent kinetic energy balance as a tool for estimating vertical diffusivity in wind-forced stratified waters. Limnol Oceanogr 45:1388–1400
Yemane D, Field JG, Leslie RW (2005) Exploring the effects of fishing on fish assemblages using abundance biomass comparison (ABC) curves. ICES J Mar Sci 62:374–379. https://doi.org/10.1016/j.icesjms.2005.01.009
Zehetner F, Lair GJ, Maringer F-J, Gerzabek MH, Hein T (2008) From sediment to soil: floodplain phosphorus transformations at the Danube river. Biogeochemistry 88:117–126. https://doi.org/10.1007/s10533-008-9198-3
Zhao J, Ramin M, Cheng V, Arhonditsis GB (2008) Plankton community patterns across a trophic gradient: the role of zooplankton functional groups. Ecol Model 213:417–436
Acknowledgements
This study was supported by the Croatian Ministry of Science, Education and Sports, research Project No. 285-0000000-2674. We are grateful to project leader Prof. Jasna Vidaković for her support. Many thanks to Goran Palijan PhD, Filip Stević PhD, Dubravka Špoljarić Maronić PhD and Vanda Zahirović for field and laboratory assistance.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Rights and permissions
About this article
Cite this article
Galir Balkić, A., Ternjej, I. Assessing Cladocera and Copepoda individual disturbance levels in hydrologically dynamic environment. Wetlands Ecol Manage 26, 733–749 (2018). https://doi.org/10.1007/s11273-018-9604-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11273-018-9604-0