Abstract
Purpose
The purpose of the study was to assess the differences in the concentration and function of urinary proteins between patients with cystine stones (CYS) and healthy controls (HC). We postulated that CYS and HC groups would demonstrate different proteomic profiles.
Methods
A pilot study was performed comparing urinary proteomes of 10 patients with CYS and 10 age- and gender-matched HC, using liquid chromatography-mass spectrometry. Proteins which met the selection criteria (i) ≥ 2 unique peptide identifications; (ii) ≥ twofold difference in protein abundance; and (iii) ≤ 0.05 p value for the Fisher’s Exact Test were analyzed using Gene Ontology classifications.
Results
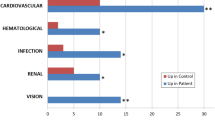
Of the 2097 proteins identified by proteomic analysis, 398 proteins were significantly different between CYS and HC. Of those, 191 were involved in transport processes and 61 in inflammatory responses. The majority were vesicle-mediated transport proteins (78.5%), and 1/3 of them were down-regulated; of those, 12 proteins were involved in endosomal transport (including 6 charged multivesicular body proteins (CHMP) and 3 vacuolar sorting-associated proteins) and 9 in transmembrane transport. Myosin-2 and two actin-related proteins were significantly up-regulated in the vesicle-mediated transport group.
Conclusion
We provide proteomic evidence of impaired endocytosis, dysregulation of actin and myosin cytoskeleton, and inflammation in CYS. Endosomal transport proteins were down-regulated mainly through defective CHMP. These findings may contribute to further understanding of the pathogenesis of CYS, potentially affecting its management.


Similar content being viewed by others
References
Chillarón J, Font-Llitjós M, Fort J, Zorzano A, Goldfarb DS, Nunes V, Palacín M (2010) Pathophysiology and treatment of cystinuria. Nat Rev Nephrol 6(7):424–424
Pras E, Arber N, Aksentijevich I, Katz G, Schapiro JM, Prosen L, Gruberg L, Harel D, Liberman U, Weissenbach J (1994) Localization of a gene causing cystinuria to chromosome 2p. Nat Genet 6(4):415–419
Knoll T, Zollner A, Wendt-Nordahl G, Michel MS, Alken P (2005) Cystinuria in childhood and adolescence: recommendations for diagnosis, treatment, and follow-up. Pediatr Nephrol 20:19–24
Dahlberg PJ, Van den Berg CJ, Kurtz SB, Wilson DM, Smith LH (1977) Clinical features and management of cystinuria. Mayo Clin Proc 52:533–542
Assimos DG, Leslie SW, Ng C, Streem SB, Hart LJ (2002) The impact of cystinuria on renal function. J Urol 168:27–20
Thongboonkerd V (2008) Proteomics and kidney stone disease. Contrib Nephrol 160:142–148
Cadieux PA, Beiko DT, Watterson JD, Burton JP, Howard JC, Knudsen BE, Gan BS, McCormick JK, Chambers AF, Denstedt JD, Reid G (2004) Surface- enhanced laser desorption/ionization-time of flight-mass spectrometry (SELDI-TOF-MS): a new proteomic urinary test for patients with urolithiasis. J Clin Lab Anal 18(3):170–175
Thongboonkerd V (2007) Practical points in urinary proteomics. J Proteome Res 6(10):3881–3880
Piper RC, Katzmann DJ (2007) Biogenesis and function of multivesicular bodies. Annu Rev Cell Dev Biol 23:519–547
Eshbach ML, Weisz OA (2017) Receptor-mediated endocytosis in the proximal tubule. Annu Rev Physiol 79:425–448
Bagga HS, Chi T, Miller J, Stoller ML (2013) New insights into the pathogenesis of renal calculi. Urol Clin N Am 40(1):1–12
Evan AP, Coe FL, Lingeman JE, Shao Y, Matlaga BR, Kim SC, Bledsoe SB, Sommer AJ, Grynpas M, Phillips CL, Worcester EM (2006) Renal crystal deposits and histopathology in patients with cystine stones. Kidney Int 69(12):2227–2235
Sabharanjak S, Sharma P, Parton RG (2002) GPI-anchored proteins are delivered to recycling endosomes via a distinct cdc42-regulated, clathrin-independeent pinocytic pathway. Dev Cell 2:411–423
Kovacevic L, Lu H, Caruso JA, Lakshmanan Y (2016) Renal tubular dysfunction in pediatric urolithiasis: proteomic evidence. Urology 92:100–105
Brooks ER, Hoppe B, Milliner DS, Salido E, Rim J, Krevitt LM, Olson JB, Price HE, Vural G, Langman CB (2016) Assessment of urine proteomics in type 1 primary hyperoxaluria. Am J Nephrol 43(4):293–303
Nagamori S, Wiriyasermkul P, Guarch ME, Okuyama H, Nakagomi S, Tadagaki K, Nishinaka Y, Bodoy S, Takafuji K, Okuda S, Kurokawa J, Ohgaki R, Nunes V, Palacín M, Kanai Y (2016) Novel cystine transporter in renal proximal tubule identified as a missing partner of cystinuria-related plasma membrane protein rBAT/SLC3A1. Proc Natl Acad Sci USA 113(3):775–780
Bourderioux M, Nguyen-Khoa T, Chhuon C, Jeanson L, Tondelier D, Walczak M, Ollero M, Bekri S, Knebelmann B, Escudier E, Escudier B, Edelman A, Guerrera IC (2014) A new workflow for proteomic analysis of urinary exosomes and assessment in cystinuria patients. J Proteome Res 14(1):567–777
Funding
This study was funded by the Rare Kidney Stone Consortium (5U54DK083908-05), a part of the Rare Diseases Clinical Research Network (RDCRN), an initiative of the Office of Rare Diseases Research (ORDR), National Center for Advancing Translational Sciences’ (NCATS). This consortium is funded through a collaboration between NCATS, and National Institute of Diabetes and Digestive and Kidney Diseases (NIDDK). The Wayne State University Proteomics Core is supported through the NIH Center Grant P30 ES 020957, the NIH Cancer Center Support Grant P30 CA 022453 and the NIH Shared Instrumentation Grant S10 OD 010700. We are thankful to Frank Modersitzki for his help with urine specimen collection and processing and with data collection.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
D.S. Goldfarb is a consultant for Retrophin and is owner of Ravine Group. The rest of the authors have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed consent
The urine was obtained for routine urinalysis for clinical care, and would have then been discarded; it was sent to the proteomics laboratory without any personal health identification information. Under these circumstances, analysis of the urine is not considered human subjects research. and therefore informed consent was not required from the participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Kovacevic, L., Caruso, J.A., Lu, H. et al. Urine proteomic profiling in patients with nephrolithiasis and cystinuria. Int Urol Nephrol 51, 593–599 (2019). https://doi.org/10.1007/s11255-018-2044-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-018-2044-1