Abstract
Purpose
Primary ADT (pADT) monotherapy is used significantly for patients with clinically localised disease in Asia and is acceptable even by guidelines, especially in intermediate- and high-risk disease. This occurs despite controversy in the West and data suggesting association with adverse effects, notably cardiovascular events. We therefore sought to assess the impact of pADT on all-cause mortality and prostate cancer-specific mortality (PCSM) in Asian men with high-risk and unfavourable intermediate-risk PCa.
Methods
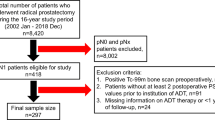
With cancer registry data, men from a single centre in Singapore with clinically localised high-risk/unfavourable intermediate-risk PCa diagnosed between 2004 and 2014 and either treated conservatively with no therapy or started on pADT within 1 year of diagnosis were followed up through January 2017. Patients with non-localised PCa (clinical stage T4, regional/distant lymph node involvement, metastases), or receipt of local therapy (radical prostatectomy/radiotherapy) were excluded. The primary outcomes of all-cause mortality and PCSM were analysed with Cox proportional hazards regression models.
Results
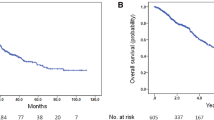
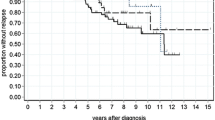
Three hundred and forty Asian men were analysed, and 177 (52.1%) were started on pADT, with mean age of 77 (49–98) years. There were 119 deaths in the cohort, and 68 (38.4%) occurred in patients treated with pADT (median follow-up, 4.4 years). After adjusting for comorbidities and clinical characteristics, pADT did not provide benefit to all-cause mortality, PCSM or cardiovascular mortality.
Conclusion
For clinically localised unfavourable intermediate-risk and high-risk PCa, starting pADT within 12 months of diagnosis is not associated with improved 5-year all-cause mortality or PCSM compared to patients treated conservatively with no therapy and should be discouraged due to lack of mortality benefit.


Similar content being viewed by others
References
Center MM, Jemal A, Lortet-Tieulent J et al (2012) International variation in prostate cancer incidence and mortality rates. Eur Urol 61:1079–1092
Immediate versus deferred treatment for advanced prostatic cancer: Initial results of the Medical Research Council Trial (1997) The Medical Research Council Prostate Cancer Working Party Investigators Group. Br J Urol 79:235–246
Loblaw DA, Virgo KS, Nam R, Somerfield MR, Ben-Josef E et al (2007) Initial hormonal management of androgen-sensitive metastatic, recurrent, or progressive prostate cancer: 2006 update of an American Society of Clinical Oncology practice guideline. J Clin Oncol 25:1596–1605
Messing EM, Manola J, Sarosdy M et al (1999) Immediate hormonal therapy compared with observation after radical prostatectomy and pelvic lymphadenectomy in men with node-positive prostate cancer. N Engl J Med 341:1781–1788
Jones CU, Hunt D, McGowan DG et al (2011) Radiotherapy and short-term androgen deprivation for localized prostate cancer. N Engl J Med 365:107–118
Lu-Yao GL, Albertsen PC, Moore DF et al (2008) Survival following primary androgen deprivation therapy among men with localized prostate cancer. JAMA 300:173–181
Tanaka N, Fujimoto K, Hirayama A et al (2010) Trends of the primary therapy for patients with prostate cancer in Nara Uro-Oncological Research Group (NUORG): a comparison between the CaPSURE data and the NUORG data. Jpn J Clin Oncol 40:588–592
Kapoor D, Malkin CJ, Channer KS, Jones TH (2005) Androgens, insulin resistance and vascular disease in men. Clin Endocrinol (Oxf) 63(3):239–250
D’Amico AV, Denham JW, Crook J et al (2007) Influence of androgen suppression therapy for prostate cancer on the frequency and timing of fatal myocardial infarctions. J Clin Oncol 25(17):2420–2425
Cooperberg M, Hinotsu S, Namiki M et al (2016) Trans-pacific variation in outcomes for men treated with primary androgen-deprivation therapy (ADT) for prostate cancer. BJU Int 117:102–109
Fukagai T, Namiki ST, Carlile R, Yoshida H, Namiki M (2006) Comparison of the clinical outcome after hormonal therapy for prostate cancer between Japanese and Caucasian men. BJU Int 97:1190–1193
National Comprehensive Cancer Network Guidelines Asia Consensus for Prostate Cancer. http://demo.nccn.org/professionals/physician_gls/PDF/prostate-asia.pdf
D’Amico AV, Whittington R, Malkowicz S et al (1998) Biochemical outcome after radical prostatectomy, external beam radiation therapy or, interstitial radiation therapy for clinically localized prostate cancer. JAMA 280(11):969–974. https://doi.org/10.1001/jama.280.11.969
Zumsteg ZS, Spratt DE, Pei I et al (2013) A new risk classification system for therapeutic decision making with intermediate-risk prostate cancer patients undergoing dose-escalated external-beam radiation therapy. Eur Urol 64:895–902
Charlson MEM (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chron Dis 40(5):373
Sammon J, Abdollah F, Reznor G et al (2015) Patterns of declining use and the adverse effects if primary androgen deprivation therapy on all-cause mortality in elderly men with prostate cancer. Eur Urol 68:32–39
Lu-Yao GL, Albertsen PC, Moore DF et al (2014) Fifteen-year survival outcomes following primary androgen-deprivation therapy for localized prostate cancer. JAMA 174(9):1460–1467
Potosky AL, Haque R, Cassidy-Bushrow AE et al (2014) Effectiveness of primary androgen-deprivation therapy for clinically localized prostate cancer. J Clin Oncol 32:1324–1330
Studer UE, Whelan P, Wimpissinger F et al (2014) Differences in time to disease progression do not predict for cancer-specific survival in patients receiving immediate or deferred androgen-deprivation therapy for prostate cancer: final results of EORTC randomized trial 30891 with 12 years of follow-up. Eur Urol 66:829–838
Wong YN, Freedland SJ, Egleston B et al (2009) The role of primary androgen deprivation therapy in localized prostate cancer. Eur Urol 56:609–616
Akaza H, Homma Y, Okada K et al (2003) A prospective and randomized study of primary hormonal therapy for patients with localized or locally advanced prostate cancer unsuitable for radical prostatectomy: results of the 5-year follow-up. BJU Int 91:33–36
Akaza H, Homma Y, Usami M et al (2006) Efficacy of primary hormone therapy for localized or locally advanced prostate cancer: results of a 10-year follow-up. BJU Int 98:573–579
Matsumoto K, Hagiwara M, Tanaka N et al (2014) Survival following primary androgen deprivation therapy for localized intermediate- or high-risk prostate cancer: comparison with the life expectancy of the age-matched normal population. Med Oncol 31:979
Akaza H, Hirao Y, Kim CS, Oya M, Ozono S, Ye D et al (2016) Asia prostate cancer study (A-CaP) launch symposium. Prostate Int 4:88–96
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
All authors involved in this study declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Declaration of Helsinki and its later amendments or comparable ethical standards. Informed consent was obtained from all participants included in this study.
Rights and permissions
About this article
Cite this article
Lee, H.J., Lee, A., Huang, H.H. et al. Primary androgen deprivation therapy as monotherapy in unfavourable intermediate- and high-risk localised prostate cancer: a Singaporean single-centre perspective. Int Urol Nephrol 50, 665–673 (2018). https://doi.org/10.1007/s11255-018-1802-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11255-018-1802-4