Abstract
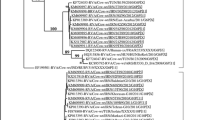
Neonatal diarrhea is the main cause of morbidity and mortality in calves up to 30 days old, and rotavirus A (RVA) is the main viral etiology. RVA vaccines are one of the main tools for diarrhea control in neonates. The aim of this cross-sectional study was to monitor by RT-PCR the G and P genotypes of RVA strains identified in dairy cattle herds regularly vaccinated with the RVA UK strain (G6P[5]). Of the 14 randomly selected herds, two were excluded because no calf was diagnosed with diarrhea on the day of fecal collection. Another six herds were also excluded from the study because all 20 diarrheic fecal samples evaluated were RT-PCR-negative. In the remaining six herds, 17 (25.4%) of the 67 diarrheic samples were RVA-positive. One G and P amplicon from each herd were selected for nucleotide sequencing. In the phylogenetic analysis, five RVA strains presented the G6P[11] genotype, and one presented the G10P[11] genotype. The G6 genotype present in all RVA field strains clustered into a distinct phylogenetic arrangement (lineage III) of the UK vaccine strain (lineage IV), characterizing the emergence of a phylogenetically distant G6 strain. In addition, we observed the emergence of strains with G10 and P[11] genotypes characterizing failure in heterologous immune protection. These results show the epidemiological importance of constant monitoring of RVA strains in vaccinated cattle herds and the low frequencies of diarrhea and diagnosis of RVA suggest that a regular vaccination program reduces the frequency and severity of RVA diarrhea in suckling calves.

Similar content being viewed by others
References
Adah, M.I., Nagashima, S., Wakuda, M., and Taniguchi, K., 2003. Close relationship between G8-serotype bovine and human rotaviruses isolated in Nigeria. Journal of Clinical Microbiology, 41, 3945-3950
Al Mawly, J., Grinberg, A., Prattley, D., Moffat, J., Marshall, J., and French, N., 2015. Risk factors for neonatal calf diarrhoea and enteropathogen shedding in New Zealand dairy farms. The Veterinary Journal, 203, 155-160
Alfieri, A.F., Alfieri, A.A., Barreiros, M.A., Leite, J.P., and Richtzenhain, L.J., 2004. G and P genotypes of group A rotavirus strains circulating in calves in Brazil, 1996-1999. Veterinary Microbiology, 99, 167-173
Alfieri, A.A., Parazzi, M.E., Takiuchi, E., Medici, K.C., and Alfieri, A.F., 2006. Frequency of group A rotavirus in diarrhoeic calves in Brazilian cattle herds, 1998-2002. Tropical Animal Health and Production, 38, 521-526
Alkan, F., Ozkul, A., Oguzoglu, T.C., Timurkan, M.O., Caliskan, E., Martella, V., and Burgu, I., 2010. Distribution of G (VP7) and P (VP4) genotypes of group A bovine rotaviruses from Turkish calves with diarrhea, 1997-2008. Veterinary Microbiology, 141, 231-237
Badaracco, A., Garaicoechea, L., Rodriguez, D., Uriarte, E.L., Odeon, A., Bilbao, G., Galarza, R., Abdala, A., Fernandez, F., and Parreno, V., 2012. Bovine rotavirus strains circulating in beef and dairy herds in Argentina from 2004 to 2010. Veterinary Microbiology, 158, 394-399
Badaracco, A., Garaicoechea, L., Matthijnssens, J., Louge Uriarte, E., Odeon, A., Bilbao, G., Fernandez, F., Parra, G.I., and Parreno, V., 2013a. Phylogenetic analyses of typical bovine rotavirus genotypes G6, G10, P[5] and P[11] circulating in Argentinean beef and dairy herds. Infection, Genetics and Evolution, 18, 18-30
Badaracco, A., Matthijnssens, J., Romero, S., Heylen, E., Zeller, M., Garaicoechea, L., Van Ranst, M., and Parreno, V., 2013b. Discovery and molecular characterization of a group A rotavirus strain detected in an Argentinean vicuna (Vicugna vicugna). Veterinary Microbiology, 161, 247-254
Banyai, K., Kemenesi, G., Budinski, I., Foldes, F., Zana, B., Marton, S., Varga-Kugler, R., Oldal, M., Kurucz, K., and Jakab, F., 2017. Candidate new rotavirus species in Schreiber’s bats, Serbia. Infection, Genetics and Evolution, 48, 19-26
Barreiros, M.A., Alfieri, A.F., Medici, K.C., Leite, J.P., and Alfieri, A.A., 2004. G and P genotypes of group A rotavirus from diarrhoeic calves born to cows vaccinated against the NCDV (P[1],G6) rotavirus strain. Journal of Veterinary Medicine, 51, 104-109
Bartels, C.J., Holzhauer, M., Jorritsma, R., Swart, W.A., and Lam, T.J., 2010. Prevalence, prediction and risk factors of enteropathogens in normal and non-normal faeces of young Dutch dairy calves. Preventive Veterinary Medicine, 93, 162-169
Buzinaro, M.G., Samara, S.I., Pereira, E.A.S., Fuentes, D.B., and Oliveira, M.C.S., 2009. Ocorrência dos Genótipos G e P de Rotavirus do grupo A em Bezerros de Rebanhos de Corte no Estado de São Paulo, Brasil. Arquivos do Instituto Biológico, 76, 99-105
Cashman, O., Lennon, G., Sleator, R.D., Power, E., Fanning, S., and O'Shea, H., 2010. Changing profile of the bovine rotavirus G6 population in the south of Ireland from 2002 to 2009. Veterinary Microbiology, 146, 238-244
Chang, K.O., Parwani, A.V., and Saif, L.J., 2000. Comparative sequence analysis of the VP7 genes of G6, G8 and G10 bovine group A rotarviruses and further characterization of G6 subtypes. Archives of Virology, 145, 725-737
Cho, Y.I., and Yoon, K.J., 2014. An overview of calf diarrhea - infectious etiology, diagnosis, and intervention. Journal of Veterinary Science, 15, 1-17
Conner, M.E., Crawford, S.E., Barone, C., and Estes, M.K., 1993. Rotavirus vaccine administered parenterally induces protective immunity. Journal of Virology, 67, 6633-6641
Cortese, V.S., 2009. Neonatal immunology, Veterinary Clinics of North America: Food Animal Practice, 25, 221-227
Coura, F.M., Freitas, M.D., Ribeiro, J., de Leme, R.A., de Souza, C., Alfieri, A.A., Facury Filho, E.J., de Carvalho, A.U., Silva, M.X., Lage, A.P., and Heinemann, M.B., 2015. Longitudinal study of Salmonella spp., diarrheagenic Escherichia coli, Rotavirus, and Coronavirus isolated from healthy and diarrheic calves in a Brazilian dairy herd. Tropical Animal Health and Production, 47, 3-11
Durel, L., Rose, C., Bainbridge, T., Roubert, J., Dressel, K.-U., Bennemann, J., Rückner, A., Vahlenkamp, T., and Maillard, R., 2017. Immune response of mature cows subjected to annual booster vaccination against neonatal calf diarrhoea with two different commercial vaccines: A non-inferiority study. Livestock Science, 204, 52-58
Gentsch, J.R., Glass, R.I., Woods, P., Gouvea, V., Gorziglia, M., Flores, J., Das, B.K., and Bhan, M.K., 1992. Identification of group A rotavirus gene 4 types by polymerase chain reaction. Journal of Clinical Microbiology, 30, 1365-1373
Gonzalez, D.D., Mozgovoj, M.V., Bellido, D., Rodriguez, D.V., Fernandez, F.M., Wigdorovitz, A., Parreno, V.G., and Dus Santos, M.J., 2010. Evaluation of a bovine rotavirus VP6 vaccine efficacy in the calf model of infection and disease. Veterinary Immunology and Immunopathology, 137, 155-160
Gouvea, V., Glass, R.I., Woods, P., Taniguchi, K., Clark, H.F., Forrester, B., and Fang, Z.Y., 1990. Polymerase chain reaction amplification and typing of rotavirus nucleic acid from stool specimens. Journal of Clinical Microbiology, 28, 276-282
Gurgel, R.Q., Cuevas, L.E., Vieira, S.C., Barros, V.C., Fontes, P.B., Salustino, E.F., Nakagomi, O., Nakagomi, T., Dove, W., Cunliffe, N., and Hart, C.A., 2007. Predominance of rotavirus P[4]G2 in a vaccinated population, Brazil. Emerging Infectious Diseases Journal, 13, 1571-1573
Kaplon, J., Fremy, C., Bernard, S., Rehby, L., Aho, S., Pothier, P., and Ambert-Balay, K., 2013. Impact of rotavirus vaccine on rotavirus genotypes and caliciviruses circulating in French cattle. Vaccine, 31, 2433-2440
Karayel, I., Feher, E., Marton, S., Coskun, N., Banyai, K., and Alkan, F., 2017. Putative vaccine breakthrough event associated with heterotypic rotavirus infection in newborn calves, Turkey, 2015. Veterinary Microbiology, 201, 7-13
Kumar, S., Stecher, G., and Tamura, K., 2016. MEGA7: Molecular Evolutionary Genetics Analysis Version 7.0 for Bigger Datasets. Molecular Biology and Evolution, 33, 1870-1874
Lorenzetti, E., da Silva Medeiros, T.N., Alfieri, A.F., and Alfieri, A.A., 2011. Genetic heterogeneity of wild-type G4P[6] porcine rotavirus strains detected in a diarrhea outbreak in a regularly vaccinated pig herd. Veterinary Microbiology, 154, 191-196
Malik, Y.S., Sharma, K., Vaid, N., Chakravarti, S., Chandrashekar, K.M., Basera, S.S., Singh, R., Minakshi, Prasad, G., Gulati, B.R., Bhilegaonkar, K.N., and Pandey, A.B., 2012. Frequency of group A rotavirus with mixed G and P genotypes in bovines: predominance of G3 genotype and its emergence in combination with G8/G10 types. Journal of Veterinary Science, 13, 271-278
Martella, V., Ciarlet, M., Banyai, K., Lorusso, E., Cavalli, A., Corrente, M., Elia, G., Arista, S., Camero, M., Desario, C., Decaro, N., Lavazza, A., and Buonavoglia, C., 2006. Identification of a novel VP4 genotype carried by a serotype G5 porcine rotavirus strain. Virology, 346, 301-311
Martella, V., Banyai, K., Matthijnssens, J., Buonavoglia, C., and Ciarlet, M., 2010. Zoonotic aspects of rotaviruses. Veterinary Microbiology, 140, 246-255
Matsuda, Y., Isegawa, Y., Woode, G.N., Zheng, S., Kaga, E., Nakagomi, T., Ueda, S., and Nakagomi, O., 1993. Two-way cross-neutralization mediated by a shared P (VP4) serotype between bovine rotavirus strains with distinct G (VP7) serotypes. Journal of Clinical Microbiology, 31, 354-358
Matthijnssens, J., Bilcke, J., Ciarlet, M., Martella, V., Banyai, K., Rahman, M., Zeller, M., Beutels, P., Van Damme, P., and Van Ranst, M., 2009. Rotavirus disease and vaccination: impact on genotype diversity. Future Microbiology, 4, 1303-1316
Matthijnssens, J., Otto, P.H., Ciarlet, M., Desselberger, U., Van Ranst, M., and Johne, R., 2012. VP6-sequence-based cutoff values as a criterion for rotavirus species demarcation. Archives of Virology, 157, 1177-1182
Medeiros, T.N.S., Lorenzetti, E., Alfieri, A.F., and Alfieri, A.A., 2015. Phylogenetic analysis of a G6P[5] bovine rotavirus strain isolated in a neonatal diarrhea outbreak in a beef cattle herd vaccinated with G6P[1] and G10P[11] genotypes. Archives of Virology, 160, 447-451
Meganck, V., Hoflack, G., Piepers, S., and Opsomer, G., 2015. Evaluation of a protocol to reduce the incidence of neonatal calf diarrhoea on dairy herds. Preventive Veterinary Medicine, 118, 64-70
Mihalov-Kovacs, E., Gellert, A., Marton, S., Farkas, S.L., Feher, E., Oldal, M., Jakab, F., Martella, V., and Banyai, K., 2015. Candidate new rotavirus species in sheltered dogs, Hungary. Emerging Infectious Diseases Journal, 21, 660-663
Otto, P.H., Rosenhain, S., Elschner, M.C., Hotzel, H., Machnowska, P., Trojnar, E., Hoffmann, K., and Johne, R., 2015. Detection of rotavirus species A, B and C in domestic mammalian animals with diarrhoea and genotyping of bovine species A rotavirus strains. Veterinary Microbiology, 179, 168-176
Papp, H., Laszlo, B., Jakab, F., Ganesh, B., De Grazia, S., Matthijnssens, J., Ciarlet, M., Martella, V., and Banyai, K., 2013. Review of group A rotavirus strains reported in swine and cattle. Veterinary Microbiology, 165, 190-199
Parreno, V., Bejar, C., Vagnozzi, A., Barrandeguy, M., Costantini, V., Craig, M.I., Yuan, L., Hodgins, D., Saif, L., and Fernandez, F., 2004. Modulation by colostrum-acquired maternal antibodies of systemic and mucosal antibody responses to rotavirus in calves experimentally challenged with bovine rotavirus. Veterinary Immunology and Immunopathology, 100, 7-24
RCWG, 2017. Rotavirus Classification Working Group. Newly assigned genotypes - June 30th 2017.
Rocha, T.G., Silva, F.D., Gregori, F., Alfieri, A.A., Buzinaro, M.D., and Fagliari, J.J., 2017. Longitudinal study of bovine rotavirus group A in newborn calves from vaccinated and unvaccinated dairy herds. Tropical Animal Health and Production, 49, 783-790
Snodgrass, D.R., Terzolo, H.R., Sherwood, D., Campbell, I., Menzies, J.D., and Synge, B.A., 1986. Aetiology of diarrhoea in young calves. Veterinary Record, 119, 31-34
Swiatek, D.L., Palombo, E.A., Lee, A., Coventry, M.J., Britz, M.L., and Kirkwood, C.D., 2010. Detection and analysis of bovine rotavirus strains circulating in Australian calves during 2004 and 2005. Veterinary Microbiology, 140, 56-62
Taniguchi, K., Urasawa, T., Kobayashi, N., Ahmed, M.U., Adachi, N., Chiba, S., and Urasawa, S., 1991. Antibody response to serotype-specific and cross-reactive neutralization epitopes on VP4 and VP7 after rotavirus infection or vaccination. Journal of Clinical Microbiology, 29, 483-487
Usonis, V., Ivaskeviciene, I., Desselberger, U., Rodrigo, C., and Pediatric, R.E.C., 2012. The unpredictable diversity of co-circulating rotavirus types in Europe and the possible impact of universal mass vaccination programmes on rotavirus genotype incidence. Vaccine, 30, 4596-4605
Van Zaane, D., Ijzerman, J., and De Leeuw, P.W., 1986. Intestinal antibody response after vaccination and infection with rotavirus of calves fed colostrum with or without rotavirus antibody. Veterinary Immunology and Immunopathology, 11, 45-63
Windeyer, M.C., Leslie, K.E., Godden, S.M., Hodgins, D.C., Lissemore, K.D., and LeBlanc, S.J., 2014. Factors associated with morbidity, mortality, and growth of dairy heifer calves up to 3 months of age. Preventive Veterinary Medicine, 113, 231-240
Acknowledgements
The authors thank the following Brazilian Institutes for financial support: the National Council of Scientific and Technological Development (CNPq), the Brazilian Federal Agency for Support and Evaluation of Graduate Education (CAPES), and the Araucária Foundation (FAP/PR). Alfieri, A.A. and Alfieri, A.F are recipients of CNPq fellowships.
Funding
This study was funded by INCT Leite (Grant number 465725/ 2014–7)
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Fritzen, J.T.T., Lorenzetti, E., Oliveira, M.V. et al. Cross-sectional study of the G and P genotypes of rotavirus A field strains circulating in regularly vaccinated dairy cattle herds. Trop Anim Health Prod 51, 887–892 (2019). https://doi.org/10.1007/s11250-018-1769-2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11250-018-1769-2