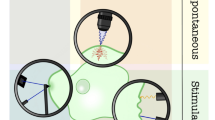
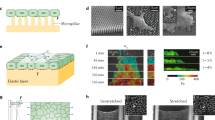
Abstract
Tribological evaluation of biological materials, such as cells and tissues, present both opportunities and challenges to experimentalists. When working with living materials, maintaining homeostasis during testing in vitro or in vivo often requires appropriate control of the environment, selection of the testing time and duration, applied loads, and shear stresses. This manuscript provides much of the background and design information used in the development of a microtribometer that has been modified to perform biotribology measurements in vitro. The focus of this manuscript is on mammalian cells in monolayer, and a series of order-of-magnitude calculations are used to inform future instrument designs and considerations, including: sliding speed ranges from 10 nm/s to 100 mm/s, contact pressures less than 6 kPa, temperature ~ 37 °C, and contact areas on the order of 1,000’s of µm2. The design and development of these biotribology instruments enable in situ fluorescence microscopy and allow for statistically significant gene expression analyses such as quantitative reverse-transcription polymerase chain-reaction (RT-qPCR) and enzyme-linked immunosorbent assay (ELISA).



Similar content being viewed by others
References
Reeve, L., Baldrick, P.: Biocompatibility assessments for medical devices – evolving regulatory considerations. Exp. Rev. Med. Devices. 14, 161–167 (2017). https://doi.org/10.1080/17434440.2017.1280392
Center for Devices and Radiological Health U.S. Food and Drug Administration. Anaplastic Large Cell Lymphoma (ALCL) In Women with Breast Implants: Preliminary FDA Findings and Analyses, http://wayback.archive-it.org/7993/20171115053750/https:/www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/BreastImplants/ucm239996.htm. Accessed Nov 9 2017
U.S. Food and Drug Administration. Medical Device Reports of Breast Implant-Associated Anaplastic Large Cell Lymphoma, https://www.fda.gov/MedicalDevices/ProductsandMedicalProcedures/ImplantsandProsthetics/BreastImplants/ucm481899.htm. Accessed April 15 2018
Nichols, J.J., Willcox, M.D.P., Bron, A.J., Belmonte, C., Ciolino, J.B., Craig, J.P., Dogru, M., Foulks, G.N., Jones, L., Nelson, J.D., Nichols, K.K., Purslow, C., Schaumberg, D.A., Stapleton, F., Sullivan, D.A.: The TFOS international workshop on contact lens discomfort: executive summary. Investig. Ophthalmol. Vis. Sci. 54, 7–13: (2013). https://doi.org/10.1167/iovs.13-13212
Efron, N.: Contact lens wear is intrinsically inflammatory. Clin. Exp. Optom. 100, 3–19 (2017). https://doi.org/10.1111/cxo.12487
Efron, N.: Rethinking contact lens discomfort. Clin. Exp. Optom. 101, 1–3 (2018). https://doi.org/10.1111/cxo.12629
Qin, G., Baidouri, H., Glasser, A., Raghunathan, V., Morris, C., Maltseva, I., McDermott, A.M.: Development of an in vitro model to study the biological effects of blinking. Ocul. Surf. (2018). https://doi.org/10.1016/j.jtos.2017.12.002
Pitenis, A.A., Urueña, J.M., Hart, S.M., O’Bryan, C.S., Marshall, S.L., Levings, P.P., Angelini, T.E., Sawyer, W.G.: Friction-induced inflammation. Tribol. Lett. 66, 81 (2018). https://doi.org/10.1007/s11249-018-1029-7
Cobb, J.A., Dunn, A.C., Kwon, J., Sarntinoranont, M., Sawyer, W.G., Tran-Son-Tay, R.: A novel method for low load friction testing on living cells. Biotechnol. Lett. 30, 801–806 (2008). https://doi.org/10.1007/s10529-007-9623-z
Dunn, A.C., Zaveri, T.D., Keselowsky, B.G., Sawyer, W.G.: Macroscopic friction coefficient measurements on living endothelial cells. Tribol. Lett. 27, 233–238 (2007). https://doi.org/10.1007/s11249-007-9230-0
Dunn, A.C., Cobb, J.A., Kantzios, A.N., Lee, S.J., Sarntinoranont, M., Tran-Son-Tay, R., Sawyer, W.G.: Friction coefficient measurement of hydrogel materials on living epithelial cells. Tribol. Lett. 30, 13–19 (2008). https://doi.org/10.1007/s11249-008-9306-5
Straehla, J.P., Limpoco, F.T., Dolgova, N.V., Keselowsky, B.G., Sawyer, W.G., Perry, S.S.: Nanomechanical probes of single corneal epithelial cells: Shear stress and elastic modulus. Tribol. Lett. 38, 107–113 (2010). https://doi.org/10.1007/s11249-010-9579-3
Sterner, O., Aeschlimann, R., Zürcher, S., Scales, C., Riederer, D., Spencer, N.D., Tosatti, S.G.P.: Tribological classification of contact lenses: from coefficient of friction to sliding work. Tribol. Lett. 63, 9 (2016). https://doi.org/10.1007/s11249-016-0696-5
Pitenis, A.A., Urueña, J.M., Hormel, T.T., Bhattacharjee, T., Niemi, S.R., Marshall, S.L., Hart, S.M., Schulze, K.D., Angelini, T.E., Sawyer, W.G.G.: Corneal cell friction: survival, lubricity, tear films, and mucin production over extended duration in vitro studies. Biotribology. 11, 77–83 (2017). https://doi.org/10.1016/j.biotri.2017.04.003
Pitenis, A.A., Urueña, J.M., McGhee, E.O., Hart, S.M., Reale, E.R., Kim, J., Schulze, K.D., Marshall, S.L., Bennett, A.I., Niemi, S.R., Angelini, T.E., Sawyer, W.G., Dunn, A.C.: Challenges and opportunities in soft tribology. Tribol. Mater. Surfaces Interfaces. 11, 180–186 (2017). https://doi.org/10.1080/17515831.2017.1400779
Zehnder, S.M., Wiatt, M.K., Uruena, J.M., Dunn, A.C., Sawyer, W.G., Angelini, T.E.: Multicellular density fluctuations in epithelial monolayers. Phys. Rev. E. 92, 032729 (2015). https://doi.org/10.1103/PhysRevE.92.032729
Schulze, K.D., Zehnder, S.M., Urueña, J.M., Bhattacharjee, T., Sawyer, W.G., Angelini, T.E.: Elastic modulus and hydraulic permeability of MDCK monolayers. J. Biomech. 53, 210–213 (2017). https://doi.org/10.1016/j.jbiomech.2017.01.016
Rennie, A.C., Dickrell, P.L., Sawyer, W.G.: Friction coefficient of soft contact lenses: measurements and modeling. Tribol. Lett. 18, 499–504 (2005). https://doi.org/10.1007/s11249-005-3610-0
Krick, B.A., Vail, J.R., Persson, B.N.J., Sawyer, W.G.: Optical in situ micro tribometer for analysis of real contact area for contact mechanics, adhesion, and sliding experiments. Tribol. Lett. 45, 185–194 (2012). https://doi.org/10.1007/s11249-011-9870-y
Urueña, J.M., Pitenis, A.A., Harris, K.L., Sawyer, W.G.: Evolution and wear of fluoropolymer transfer films. Tribol. Lett. 57, 9 (2015). https://doi.org/10.1007/s11249-014-0453-6
Pitenis, A.A., Urueña, J.M., Schulze, K.D., Nixon, R.M., Dunn, A.C., Krick, B.A., Sawyer, W.G., Angelini, T.E., Sawyer, G., Angelini, T.E.: Polymer fluctuation lubrication in hydrogel gemini interfaces. Soft Matter. 10, 8955–8962 (2014). https://doi.org/10.1039/C4SM01728E
Urueña, J.M., Pitenis, A.A., Nixon, R.M., Schulze, K.D., Angelini, T.E., Sawyer, W.G.: Mesh Size control of polymer fluctuation lubrication in gemini hydrogels. Biotribology. 1–2, 24–29 (2015). https://doi.org/10.1016/j.biotri.2015.03.001
Pitenis, A.A., Manuel Urueña, J., Cooper, A.C., Angelini, T.E., Sawyer, W.G.: Superlubricity in gemini hydrogels. J. Tribol. 138, 042103 (2016). https://doi.org/10.1115/1.4032890
Schmitz, T.L., Action, J.E., Ziegert, J.C., Sawyer, W.G.: The difficulty of measuring low friction: Uncertainty analysis for friction coefficient measurements. J. Tribol. 127, 673 (2005). https://doi.org/10.1115/1.1843853
Burris, D.L., Sawyer, W.G.: Addressing practical challenges of low friction coefficient measurements. Tribol. Lett. 35, 17–23 (2009). https://doi.org/10.1007/s11249-009-9438-2
Schulze, K.D., Hart, S.M., Marshall, S.L., O’Bryan, C.S., Urueña, J.M., Pitenis, A.A., Sawyer, W.G., Angelini, T.E.: Polymer osmotic pressure in hydrogel contact mechanics. Biotribology. 11, 3–7 (2017). https://doi.org/10.1016/j.biotri.2017.03.004
Urueña, J.M., McGhee, E.O., Angelini, T.E., Dowson, D., Sawyer, W.G., Pitenis, A.A.: Normal load scaling of friction in gemini hydrogels. Biotribology. 13, 30–35 (2018). https://doi.org/10.1016/j.biotri.2018.01.002
Johnson, K.L.: Contact Mechanics. Cambridge University Press, Cambridge (1985)
Garcia, M., Schulze, K.D., O’Bryan, C.S., Bhattacharjee, T., Sawyer, W.G., Angelini, T.E.: Eliminating the surface location from soft matter contact mechanics measurements. Tribol. Mater. Surfaces Interfaces. 11, 187–192 (2017). https://doi.org/10.1080/17515831.2017.1397908
Marshall, S.L., Schulze, K.D., Hart, S.M., Urueña, J.M., McGhee, E.O., Bennett, A.I., Pitenis, A.A., O’Bryan, C.S., Angelini, T.E., Sawyer, W.G.: Spherically capped membrane probes for low contact pressure tribology. Biotribology. 11, 69–72 (2017). https://doi.org/10.1016/j.biotri.2017.03.008
Bhattacharjee, T., Zehnder, S.M., Rowe, K.G., Jain, S., Nixon, R.M., Sawyer, W.G., Angelini, T.E.: Writing in the granular gel medium. Sci. Adv. 1, e1500655–e1500655 (2015). https://doi.org/10.1126/sciadv.1500655
Bhattacharjee, T., Gil, C.J., Marshall, S.L., Urueña, J.M., O’Bryan, C.S., Carstens, M., Keselowsky, B., Palmer, G.D., Ghivizzani, S., Gibbs, C.P., Sawyer, W.G., Angelini, T.E.: Liquid-like solids support Cells in 3D. ACS biomater. Sci. Eng. 2, 1787–1795 (2016). https://doi.org/10.1021/acsbiomaterials.6b00218
O’Bryan, C.S., Bhattacharjee, T., Hart, S., Kabb, C.P., Schulze, K.D., Chilakala, I., Sumerlin, B.S., Sawyer, W.G., Angelini, T.E.: Self-assembled micro-organogels for 3D printing silicone structures. Sci. Adv. 3, e1602800 (2017). https://doi.org/10.1126/sciadv.1602800
O’Bryan, C.S., Bhattacharjee, T., Niemi, S.R., Balachandar, S., Baldwin, N., Ellison, S.T., Taylor, C.R., Sawyer, W.G., Angelini, T.E.: Three-dimensional printing with sacrificial materials for soft matter manufacturing. MRS Bull. 42, 571–577 (2017). https://doi.org/10.1557/mrs.2017.167
Svoboda, K., Block, S.M.: Force and velocity measured for single kinesin molecules. Cell. 77, 773–784 (1994). https://doi.org/10.1016/0092-8674(94)90060-4
Dunn, A.C., Tichy, J.A., Uruenã, J.M., Sawyer, W.G.G., Urueña, J.M., Sawyer, W.G.G.: Lubrication regimes in contact lens wear during a blink. Tribol. Int. 63, 45–50 (2013). https://doi.org/10.1016/j.triboint.2013.01.008
Dowson, D.: Paper 12: Modes of Lubrication in Human Joints. Proc. Inst. Mech. Eng. Conf. Proc. 181, 45–54: (1966). https://doi.org/10.1243/PIME_CONF_1966_181_206_02
Hamrock, B.J., Dowson, D.: Elastohydrodynamic Lubrication of elliptical contacts for materials of low elastic modulus I—fully flooded conjunction. J. Lubr. Technol. 100, 236 (1978). https://doi.org/10.1115/1.3453152
Kapitza, P.L.: Hydrodynamic theory of lubrication during rolling. Zh. Tekh. Fiz. 25, 747–762 (1955)
Dunn, A.C., Urueña, J.M., Huo, Y., Perry, S.S., Angelini, T.E., Sawyer, W.G.: Lubricity of surface hydrogel layers. Tribol. Lett. 49, 371–378 (2013). https://doi.org/10.1007/s11249-012-0076-8
Schulze, K.D., Bennett, A.I., Marshall, S., Rowe, K.G., Dunn, A.C.: Real area of contact in a soft transparent interface by particle exclusion microscopy. J. Tribol. 138, 041404 (2016). https://doi.org/10.1115/1.4032822
McGhee, E.O., Pitenis, A.A., Urueña, J.M., Schulze, K.D., McGhee, A.J., O’Bryan, C.S., Bhattacharjee, T., Angelini, T.E., Sawyer, W.G.: In situ measurements of contact dynamics in speed-dependent hydrogel friction. Biotribology. 13, 23–29 (2018). https://doi.org/10.1016/j.biotri.2017.12.002
Lichtman, J.W., Conchello, J.-A.: Fluorescence microscopy. Nat. Methods. 2, 910–919 (2005). https://doi.org/10.1038/nmeth817
Shaner, N.C., Steinbach, P.A., Tsien, R.Y.: A guide to choosing fluorescent proteins. Nat. Methods. 2, 905–909 (2005). https://doi.org/10.1038/nmeth819
Berridge, M.J., Bootman, M.D., Roderick, H.L.: Calcium signalling: dynamics, homeostasis and remodelling. Nat. Rev. Mol. Cell Biol. 4, 517–529 (2003). https://doi.org/10.1038/nrm1155
de Oliveira, C.M.B., Sakata, R.K., Issy, A.M., Gerola, L.R., Salomão, R.: Cytokines and Pain. Rev. Bras. Anestesiol. 61, 255–265 (2011). https://doi.org/10.1016/S0034-7094(11)70029-0
Zhang, J.-M., An, J.: Cytokines, inflammation, and pain. Int. Anesthesiol. Clin. 45, 27–37 (2007). https://doi.org/10.1097/AIA.0b013e318034194e
Milo, R., Phillips, R.: Cell biology by the numbers. Garland Science, Taylor & Francis Group, New York (2016)
Ramsköld, D., Luo, S., Wang, Y.-C., Li, R., Deng, Q., Faridani, O.R., Daniels, G.A., Khrebtukova, I., Loring, J.F., Laurent, L.C., Schroth, G.P., Sandberg, R.: Full-length mRNA-Seq from single-cell levels of RNA and individual circulating tumor cells. Nat. Biotechnol. 30, 777–782 (2012). https://doi.org/10.1038/nbt.2282
Shapiro, E., Biezuner, T., Linnarsson, S.: Single-cell sequencing-based technologies will revolutionize whole-organism science. Nat. Rev. Genet. 14, 618–630 (2013). https://doi.org/10.1038/nrg3542
Milo, R.: What is the total number of protein molecules per cell volume? A call to rethink some published values. BioEssays. 35, 1050–1055 (2013). https://doi.org/10.1002/bies.201300066
Acknowledgements
This work was supported by Alcon Laboratories.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Urueña, J.M., Hart, S.M., Hood, D.L. et al. Considerations for Biotribometers: Cells, Gels, and Tissues. Tribol Lett 66, 141 (2018). https://doi.org/10.1007/s11249-018-1094-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s11249-018-1094-y