Abstract
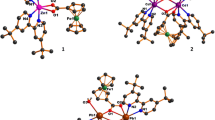
A new ligand 1-carboxymethyl-5-methyl-1H-pyrazole-4-carboxylic acid (H2L1) and its isomer 1-carboxymethyl-3-methyl-1H-pyrazole-4-carboxylic acid (H2L2) have been synthesized and characterized by IR and 1H NMR. Three complexes of these ligands, [M(HL1)2(H2O)2] (1: M = Zn; 2: M = Cu) and [Cd(HL2)2(H2O)2] (3) were synthesized by the reactions of H2L1 or H2L2 with the corresponding M(NO3)2·6H2O. The complexes were characterized by physico-chemical and spectroscopic methods, single-crystal X-ray diffraction, and thermogravimetric analysis. Complexes 1 and 2 are isostructural, while complex 3 crystallizes in the triclinic system. The M(II) atom in each of the complexes has a distorted octahedral geometry involving two oxygen and two nitrogen atoms from two anionic (HL1)− or (HL2)− ligands and two oxygen atoms from water ligands. Adjacent monomeric components are connected by hydrogen bonds to form a 3D supramolecular network. The free ligands and complexes 1 and 3 show dual-emissive luminescence in the solid state at room temperature. The luminescence intensity of solid complex 1 is weakened in the presence of NH3.
Graphical Abstract
Two new isomers, H2L1 and H2L2, and three complexes, [Zn(HL1)2(H2O)2], [Cu(HL1)2(H2O)2], and [Cd(HL2)2(H2O)2] have been synthesized, which display dual-emissive luminescence in the solid state.







Similar content being viewed by others
References
Wang B, Côt AP, Furukawa H, O′Keeffe M, Yaghi OM (2008) Nature 453:207–211
Park HJ, Suh MP (2010) Chem Commun 46:610–612
Liu D, Lang JP, Abrahams BF (2011) J Am Chem Soc 133:11042–11045
Li JR, Sculley J, Zhou HC (2012) Chem Rev 112:869–932
Cao JW, Gao YF, Wang YQ, Du CF, Liu ZL (2013) Chem Commun 49:6897–6899
Cui YJ, Yue YF, Qian GD, Chen BL (2012) Chem Rev 112:1126–1162
Fu RB, Hu SM, Wu XT (2012) Cryst Eng Comm 14:3478–3483
Ma LQ, Abney C, Lin WB (2009) Chem Soc Rev 38:1248–1256
Uraguchi D, Ueki Y, Ooi T (2009) Science 326:120–123
Liu Q, Yu LL, Wang Y, Ji YZ, Horvat J, Cheng ML, Jia XY, Wang GX (2013) Inorg Chem 52:2817–2822
Zhang GQ, Palmer GM, Dewhirst MW, Fraser CL (2009) Nat Mater 8:747–751
Lo KKW, Zhang KY, Leung SK, Tang MC (2008) Angew Chem Int Ed 47:2213–2216
Li LN, Zhang SQ, Han L, Sun ZH, Luo JH, Hong MC (2013) Cryst Growth Des 13:106–110
Cheng ML, Zhu EJ, Liu Q, Chen SC, Chen Q, He MY (2011) Inorg Chem Commun 14:300–303
Hong J, Cheng ML, Liu Q, Han W, Zhang YP, Ji YZ, Jia XY, Li ZF (2013) Trans Met Chem 38:385–392
Chen LT, Tao F, Wang LD, Hong J, Jia XY, Bao JT, Ji YZ, Cheng ML, Liu Q (2013) Z Anorg Allg Chem 639:552–557
Wang LD, Tao F, Cheng ML, Liu Q, Han W, Wu YJ, Yang DD, Wang LJ (2012) J Coord Chem 65:923–933
Han W, Cheng ML, Liu Q, Wang LD, Wu YJ (2012) Chin J Inorg Chem 28:1997–2004
Bao JT, Cheng ML, Liu Q, Han W, Zhai CW, Hong J, Sun XQ (2013) Chin J Inorg Chem 29:1504–1512
Zhou XH, Du XD, Li GN, Zou JL, You XZ (2009) Cryst Growth Des 9:4487–4496
Xie CZ, Li RF, Wang LY, Zhang QQ (2010) Z Anorg Allg Chem 636:657–661
Xia J, Zhao B, Wang HS, Shi W, Ma Y, Song HB, Cheng P, Liao DZ, Yan SP (2007) Inorg Chem 46:3450–3458
Lincke J, Lässig D, Kobalz M, Bergmann J, Handke M, Mollmer J, Langer M, Roth C, Möller A, Staudt R, Krautscheid H (2012) Inorg Chem 51:7579–7586
Gadzikwa T, Farha OK, Mulfort KL, Hupp JT, Nguyen ST (2009) Chem Commun 25:3720–3722
Cohen SM (2011) Chem Rev 112:970–1000
Yakimovich SI, Zerova IV, Pakal’nis VV (2008) Russ J Org Chem 44:621–623
Sheldrick GM (1997) SHELXTL-97, program for X-ray crystal structure determination. University of Göttingen, Germany
Lässig D, Lincke J, Griebel J, Kirmse R, Krautscheid H (2011) Inorg Chem 50:213–219
Ni TJ, Xing FF, Shao M, Zhao YM, Zhu SR, Li MX (2011) Cryst Growth Des 11:2999–3012
Xi PM, Wang SS, Gao DZ, Wang XG, Sun YQ, Zhang GY, Xu YY (2013) Transit Met Chem 38:873–881
Li ZF, Chen CJ, Yan LH, Li G, Wu CJ, Lu HJ (2011) Inorg Chim Acta 377:42–49
Li M, Ling Q, Yang Z, Li BL, Li HY (2013) Cryst Eng Comm 15:3630–3639
Zhao J, Wang XL, Shi X, Yang QH, Li C (2011) Inorg Chem 50:3198–3205
Baho N, Zargarian D (2007) Inorg Chem 46:299–308
Wu JJ, Ye YX, Qiu YY, Qiao ZP, Cao ML, Ye BH (2013) Inorg Chem 52:6450–6456
Acknowledgments
The authors are particularly grateful to the Natural Science Foundation of China (Nos. 20971060 and 21101018), the Project Funded by the Priority Academic Program Development of Jiangsu Higher Education Institutions, and the Natural Science Foundation of State Key Laboratory of Coordination Chemistry for the financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11243_2014_9833_MOESM1_ESM.doc
CCDC 958411, 962642, and 973934 contain the supplementary crystallographic data for the three complexes 1, 2, and 3. These data can be obtained free of charge at www.ccdc.cam.ac.uk/conts/retrieving.html, or from the Cambridge Crystallographic Data Center, 12 Union Road, Cambridge CB2 1EZ, UK; Fax: (internet) þ 44-1223/336-033; E-mail: deposit@ccdc.cam.ac.uk. (DOC 3276 kb)
Rights and permissions
About this article
Cite this article
Su, S., Cheng, Ml., Ren, Y. et al. Three transition metal complexes with uncoordinated carboxyl groups: synthesis, structures, and luminescence properties. Transition Met Chem 39, 559–566 (2014). https://doi.org/10.1007/s11243-014-9833-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11243-014-9833-x