Abstract
Comparative effect of meta-topolin and other cytokinins was assessed to develop an efficient and reliable regeneration protocol for Tecoma stans, using mature nodal explants. The morphogenic effect of benzyl adenine (BA), kinetin (Kin), meta- topolin (mT) and 2-iP (2-iso pentenyl adenine) at various concentrations (1.0–10 µM) was studied individually or in combination with auxins (IAA, IBA or NAA). Superior multiplication rates were achieved on MS medium supplemented with mT and NAA. Of the tested combinations, maximum shoot regeneration (95%), mean shoot number (19.6 ± 0.60) and length (5.26 ± 0.73 cm) was recorded on MS medium supplemented with 7.5 µM mT + 0.5 µM NAA after 8 weeks of incubation. Among the different auxins employed for in vitro root induction, 92.5% microshoots rooted on MS medium enriched with 1.0 µM IBA with 10.8 ± 0.20 mean root number and 5.62 ± 0.17 cm length after 4 weeks of incubation. The acclimatized plants grew well in green house with 90% survival rate. The gas chromatography–mass spectrometry (GC–MS) analysis of ethanol leaf extract of in vitro-raised plants yielded a higher number of compounds than control plant. The assessment of genetic fidelity among regenerants, using ISSR markers did not reveal any somaclonal variation. Therefore, the protocol developed appears to be simple and reliable for mass production of clones with higher diversity of secondary metabolites.
Key message
Our studies based on interaction of mT with auxins for better shoot proliferation will undoubtedly be helpful in understanding many developmental processes in plant tissue culture. The regenerated plantlets survived well under field conditions and no somaclonal variation was detected. This study proved that in vitro raised plants are superior for the production of higher diversity of secondary metabolites than in vivo plant. Thus, our findings are helpful in generating an efficient micropropagation system for mass scale production of plants with higher diversity of bio-active molecules without losing their genetic stability.




Similar content being viewed by others
Abbreviations
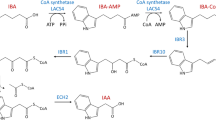
- IAA:
-
Indole -3- acetic acid
- BA:
-
Benzyl adenine
- IBA:
-
Indole-3-butyric acid
- NAA:
-
α-Naphthalene acetic acid
- Kin:
-
Kinetin
- 2iP:
-
2-iso pentenyl adenine
- mT:
-
Meta-topolin
- MS:
-
Murashige and Skoogs medium
- ISSR:
-
Inter-simple sequence repeat
References
Ahmad A, Anis M (2019) Meta-topolin improves in vitro morphogenesis, rhizogenesis and biochemical analysis in Pterocarpus marsupium Roxb.: a potential drug-yielding tree. J Plant Growth Regul. https://doi.org/10.1007/s00344-018-09910-9
Al- Azzawi AM, Al-Juboori AG (2012) Gas chromatography/mass spectroscopy for phytochemical screening of Tecoma stans. Can J Pure Appl Sci 6:1809–1813
Amoo SO, Van Staden J (2013) Influence of plant growth regulators on shoot proliferation and secondary metabolite production in micropropagated Huernia hystrix. Plant Cell Tiss Org Cult 112:249–256
Amoo SO, Finnie JF, Van Staden J (2011) The role of meta topolins in alleviating micropropagation problems. Plant Growth Regul 63:197–201
Anburaj G, Marimuthu M, Rajasudha V, Manikandan R (2016) Phytochemical screening and GC-MS analysis of ethanolic extract of Tecoma stans (Family: Bignoniaceae) Yellow Bell Flowers. J Pharmacog Phytochem 5:172–175
Andrade-Cetto A, Heinrich M (2005) Mexican plants with hypoglycemic effect used in the treatment of diabetes. J Ethnopharmacol 99:325–348
Anis M, Ahmad N (2016) Plant tissue culture: propagation conservation and Crop improvement. Springer, Singapore
Aremu AO, Bairu MW, Dolezal K, Finnie JF, Van Staden J (2012) Topolins: a panacea to plant tissue culture challenges? Plant Cell Tissue Organ Culture 108:1–16
Babu GA, Vinoth A, Ravindhran R (2018) Direct shoot regeneration and genetic fidelity analysis in finger millet using ISSR markers. Plant Cell Tissue Organ Cult 132:157–164
Baburaj S, Gunasekaran K (1993) Micropropagation of Tecoma stans (L.) Kunth. Geeobios (Jodhpur) 20:246–249
Bairu MW, Stirk WA, Dolezal K, Van Staden J (2007) Optimizing the micropropagation protocol for the endangered Aloe polyphylla: can meta-topolin and its derivatives serve as replacement for benzyladenine and zeatin? Plant Cell Tissue Organ Cult 90:15–23
Behera B, Sinha P, Gouda S, Rath SK, Barik DP, Jena PK, Panda PC, Naik SK (2018) In vitro propagation by axillary shoot proliferation, assessment of antioxidant activity, and genetic fidelity of micropropagated Paederia foetida L. J App Biol Biotech 6:41–49
Bhardwaj AK, Naryal A, Bhardwaj P, Warghat AR, Arora B, Dhiman S, Saxena S, Pati PK, Prakash Chaurasia OP (2018) High efficiency in vitro plant regeneration and secondary metabolite quantification from leaf explants of Rhodiola imbricate. Pharmacogn J 10:470–475
Bhattacharyya P, Kumaria S, Diengdoh R, Tandon P (2014) Genetic stability and phytochemical analysis of in vitro regenerated plants of Dendrobium nobile Lindl., an endangered medicinal orchid. Meta Gene 2:489–504
Coenen C, Lomax TL (1997) Auxin-cytokinin interactions in higher plants: old problems and new tools. Trends Plant Sci 2:351–356
Costantino L, Raimondi L, Parisino R, Brunetti T, Pessotto P, Giannessi F, Lins AP, Barlocco D, Antolini L, El-Abady SA (2003) Isolation and pharmacological activities of the Tecoma stans alkaloids. II Farmaco 58:781–785
Das C, Mohanty A, Dash S, Sahoo DC, Choudhury NSK, Patro VJ, Kanungo SK (2009) Phytochemical screening of crude extracts of Tecoma stans Linn. (Bignoniaceae). Res J Pharm Technol 2:816–818
Deventhiran M, Wyson WJ, Noor MS, Jaikumar K, Saravanan P, Anand D (2017) Comparative phytochemical analysis of wild and micropropagated Cleome viscosa L. J Appl Pharmaceut Sci 7:083–088
Doyle JJ, Doyle JL (1987) A rapid DNA isolation procedure for small quantities of fresh leaf tissue. Phytochem Bull 19:11–15
Escalona M, Cejas I, Gonzalez-Olmedo J, Capote I, Roels S, Canal MJ, Rodrıguez R, Sandoval J, Debergh P (2003) The effect of meta-topolin on plantain propagation using a temporary immersion bioreactor. InfoMusa 12:28–30
Fajinmi OO, Amoo SO, Finnie JF, Staden Van (2014) Optimization of in vitro propagation of Coleonema album, a highly utilized medicinal and ornamental plant. J S Afr J Bot 94:9–13
Goyal AK, Pradhan S, Basistha BC, Sen A (2015) Micropropagation and assessment of genetic fidelity of Dendrocalamus strictus (Roxb.) nees using RAPD and ISSR markers. 3 Biotech 5:473–482
Grzegorczyk I, Matkowski A, Wysokinska H (2007) Antioxidant activity of extracts from in vitro cultures of Salvia officinalis L. Food Chem 104:536–541
Gupta PK, Roy JK (2002) Molecular markers in crop improvement present status and future needs in India. Plant Cell Tissue Organ Cult 70:229–234
Hussain SA, Ahmad N, Anis M (2018) Synergetic effect of TDZ and BA on minimizing the post-exposure effects on axillary shoot proliferation and assessment of genetic fidelity in Rauvolfia tetraphylla (L.). Rendiconti Lincei Fisiche e Naturali 29:109–115
Hussain SA, Anis M, Alatar AA (2019) Efficient In Vitro Regeneration System for Tecoma stans L., using shoot tip and assessment of genetic fidelity among regenerants. Proc. Natl Acad Sci India Sect B Biol Sci 1:2–3. https://doi.org/10.1007/s40011-019-01093-x
Iiczuk A, Jacygard E (2016) In vitro propagation and assessment of genetic stability of acclimated plantlets of Cornus alba L. using RAPD and ISSR markers. Vitro Cell Dev Biol Plant 52:379–390
Kaminek M, Vanek T, Motyka V (1987) Cytokinin activities of N6-benzyladenosine derivatives hydroxylated on the side-chain phenyl ring. J Plant Growth Regul 6:113–120
Kant T, Prajapati UKT, Parmar AK (2010) In vitro propagation as a viable conservation strategy for Commiphora wightii, an endangered medicinally important desert tree. Conserv Evid 7:94–99
Kar P, Chakraborty AK, Bhattacharya M, Mishra T, Sen A (2019) Micropropagation, genetic fidelity assessment and phytochemical studies of Clerodendrum thomsoniae Balf. f. With special reference to its anti-stress properties. Res Plant Biol 9:9–15
Kumar R, Sing G (2014) Investigations into the cause of sterility Tecoma stans L. Bull Soc Bot Fr 135:131–135
Lakshmanan V, Venkataramareddy SR, Neelwarne N (2007) Molecular analysis of genetic stability in long-term micropropagated shoots of banana using RAPD and ISSR markers. Electron J Biotechnol 15:1–8
Larkin PJ, Scowcroft W (1981) Somaclonal variation—a novel source of variability from cell cultures for plant improvement. Theor Appl Genet 60:197–214
Lopez-Laredo AR, Ramirez-Flores FD, Sepulveda-Jimenez G, Trejo-Tapia G (2009) Comparison of metabolite levels in callus of Tecoma stans (L.) Juss. ex Kunth. Cultured in photoperiod and darkness. Vitro Cell Dev Biol Plant 45:550–558
Ludwig-Müller J (2000) Indole-3-butyric acid in plant growth and development. Plant Growth Regul 32:219–230
Mala J, Machova P, Cvrckova H, Karady M, Novak O, Mikulık J, Dostal J, Strnad M, Dolezal K (2013) The role of cytokinins during micropropagation of Wych elm. Biol Plant 57:174–178
Marzouk M, Gamal-Eldeen A, Mohamed M, El-Sayed M (2006) Anti-proliferative and antioxidant constituents from Tecoma stans. Z Naturforschung 61:783–791
Matkowski A (2008) Plant in vitro culture for the production of antioxidants—a review. Biotechnol Adv 26:548–560
Murashige T, Skoog F (1962) A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol Plant 15:473–497
Naaz A, Hussain SA, Anis M, Alatar AA (2019) Meta-topolin improved micropropagation in Syzygium cumini and acclimatization to ex vitro conditions. Biol Plant 63:174–182
Nordstrom AC, Jacobs FA, Eliasson L (1991) Effect of exogenous indole-3-acetic acid and indole-3-butyric acid on internal levels of the respective auxins and their conjugation with aspartic acid during adventitious root formation in pea cuttings. Plant Physiol 96:856–861
Perveen S, Javed SB, Anis M, Aref IM (2013) Rapid in vitro multiplication and ex vitro establishment of Caribbean copper plant (Euphorbia cotinifolia L.): an important medicinal shrub. Acta Physiol Plant 35:3391–3400
Petri C, Scorza R (2010) Factors affecting adventitious regeneration from in vitro leaf explants of ‘‘Improved French’’ plum, the most important dried plum cultivar in the USA. Ann Appl Biol 156:79–89
Robaszkiewicz A, Balcerczyk A, Bartosz G (2007) Antioxidative and prooxidative effects of quercetin on A549 cells. Cell Biol I 31:1245–1250
Saha S, Adhikari S, Dey T, Gosh P (2016) RAPD and ISSR based evaluation of genetic stability of micropropagated plantlets of Morus alba L. variety S-1. Meta Gene 7:7–13
Salvi ND, George L, Eapen S (2002) Micropropagation and field evaluation of micropropagated plants of turmeric. Plant Cell Tissue Organ Cult 68:143–151
Singh A, Nagori BP, Mathur PK (2013) Tecoma stans: an important medicinal plant. Int J Pharm Erud 3:13–21
Spethmann W, Hamzah A (1988) Growth hormone induced root system types in cuttings of some broad leaved tree species. Acta Hort 226:601–605
Tang H, Ren Z, Reustle G, Krczal G (2002) Plant regeneration from leaves of sweet and sour cherry cultivars. Sci Hortic 93:235–244
Vandana CD, Shanti KN, Shantha SL (2017) GC-MS analysis of callus and leaf extracts in vitro propagated plants of Justicia wynaadensis (Nees) T. Anderson. Int J Pharm Res 9:535–543
Verpoorte R, Contin A, Memelink J (2002) Biotechnology for the production of plant secondary metabolites. Phytochem Rev 1:13–25
Werbrouck SPO, Strnad M, Van Onckelen HA, Debergh PC (1996) Meta-topolin, an alternative to benzyladenine in tissue culture. Physiol Plant 98:291–297 380. Plant Cell Tissue Organ Culture (2014) 118:373–381123
Wojtania A (2010) Effect of meta-topolin on in vitro propagation of Pelargonium x hortorum and Pelargonium x hederaefolium cultivars. Acta Soc Bot Pol 79:101–106
Yang T, Davies PJ (1999) Promotion of stem elongation by indole-3-butyric acid in intact plants of Pisum sativum L. Plant Growth Regul 27:157–160
Zhang C, Qi M, Shao Q, Zhou S, Fu R (2007) Analysis of the volatile compounds in Ligusticum chuanxiong Hort. using HS-SPME-GC-MS. J Pharm Biomed Anal 44:464–470
Acknowledgements
The authors extend their appreciation to the International Scientific Partnership Program (ISPP), at King Saud University for funding this research work through ISPP#0082. The award of UGC- BSR Faculty Fellowship (2017) (F.18-1/2011(BSR) to MA by the University Grants Commission, New Delhi, is duly acknowledged. SAH is thankful to the University Grants Commission, New Delhi for the award of SRF (MANF-2013-14-MUS-JAM-22128).
Author information
Authors and Affiliations
Contributions
SAH performed the experiments, collected data and conducted statistical analysis. NA helped in genetic fidelity experiment and MA designed, supervised and provided valuable suggestions throughout the study. The manuscript was edited and reviewed by AAA.
Corresponding author
Ethics declarations
Conflicts of interest
The authors declare there are no conflicts of interest.
Additional information
Communicated by Sergio J. Ochatt.
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hussain, S.A., Ahmad, N., Anis, M. et al. Influence of meta-topolin on in vitro organogenesis in Tecoma stans L., assessment of genetic fidelity and phytochemical profiling of wild and regenerated plants. Plant Cell Tiss Organ Cult 138, 339–351 (2019). https://doi.org/10.1007/s11240-019-01631-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11240-019-01631-5