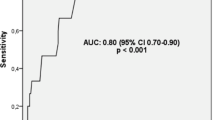
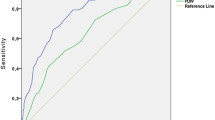
Abstract
Red blood cell distribution width (RDW) is a marker of variability in red blood cell size, and is routinely reported as part of a patient’s complete blood count. RDW has been shown to be associated with the prediction, severity and prognosis of pulmonary embolism (PE) in recent studies. The underlying biomolecular mechanism of the relationship of RDW to PE is largely unknown, but is thought to be due to the relationship of RDW with acute inflammatory markers and variations in blood viscosity. This review substantiates that a high RDW level, defined using either an arbitrary number or according to receiver operator curve statistics, is associated with a higher risk of acute PE, increased severity (massive vs. submassive) of PE and increased mortality in patients with PE. Nevertheless, the comparison of current studies is limited due to the definition of high RDW (each study uses a different RDW cutoff level), the broad range of exclusion criteria and the inclusion of differing modalities used to diagnose a PE (computed tomography angiogram, ventilation-perfusion study, or clinical diagnosis). Despite the above limitations, these studies provide a promising future clinical use for RDW as a marker of PE.
Similar content being viewed by others
Notes
The pulmonary embolism severity index (PESI) score is a clinical tool that predicts 30-day mortality in those with a PE based on 11 criteria—age, sex, history of cancer, history of heart failure, history of chronic lung disease, heart rate ≥ 110 beats/min systolic blood pressure < 100 mmHg, respiratory rate ≥ 30, temperature < 96.8 °F/36 °C, altered mental status and oxygen saturation < 90%; the algorithm categorizes patients into five groups corresponding to various prognoses, with the highest-scoring groups having the poorest outcome (30-day mortality up to 24.5%) [36]. The simplified PESI (sPESI) is derived from its predecessor and predicts 30-day mortality, but only uses six criteria—age > 80 years, history of cancer, history of chronic cardiopulmonary disease, heart rate ≥ 110 beats/min, systolic blood pressure 9 < 100 mmHg and arterial oxygen saturation < 90% measured at the time of PE diagnosis; each positive variable is assigned 1 point and the patient is then classified into a low-risk (0 points and 1.1% 30-day mortality rate) or high-risk (≥ 1 point[s] and 8.9% 30-day mortality rate) group[59].
The European Society of Cardiology (ESC) also uses a scoring system (including the sPESI score) to classify PE patients into risk groups based on 30-day mortality risk: low (1 point), intermediate (2 points), intermediate-high (3 points) and high (4 points)[38, 60]. For the ESC classification system, one point is allotted to each of the following risk variables: shock/hypotension (systolic blood pressure < 90 mmHg, or a systolic pressure drop by ≥ 50 mmHg for > 15 min), sPESI ≥ 1, RV dysfunction, and troponin T ≥ 14 pg/mL and/or NT-proBNP > 600 pg/mL [38, 60]. The low risk group has a < 1% 30-day mortality, whereas the high risk group has a > 15% 30-day mortality [38].
The Jen scoring system, published in 2018, also predicts 30-day mortality using high-risk variables to define a binary prediction classification (low vs. high risk). There are eight variables, and they are weighted as follows: age in years, HF (+ 20), lung disease (+ 25), respiratory rate > 30 bpm (+ 40), altered mental status (+ 10), IV required (+ 50), ALT > 75 IU/L (+ 40), and hemoptysis (+ 60). Thirty-day mortality risks were 2.1% and 23% for low (score ≤ 100) and high (> 100) risk patients, respectively (p < 0.001) [61].
References
Stein PD, Beemath A, Olson RE (2005) Trends in the incidence of pulmonary embolism and deep venous thrombosis in hospitalized patients. Am J Cardiol 95(12):1525–1526
Dalen JE, Alpert JS (1975) Natural history of pulmonary embolism. Prog Cardiovasc Dis 17(4):259–270
Naess IA, Christiansen SC, Romundstad P et al (2007) Incidence and mortality of venous thrombosis: a population-based study. J Thromb Haemost 5(4):692–699
Watson T, Shantsila E, Lip GY (2009) Mechanisms of thrombogenesis in atrial fibrillation: Virchow’s triad revisited. Lancet 373(9658):155–166
Simel DL, DeLong ER, Feussner JR, Weinberg JB, Crawford J (1988) Erythrocyte anisocytosis: visual inspection of blood films versus automated analysis of red blood cell distribution width. Arch Intern Med 148(4):822–824
Marsh WL, Bishop JW, Darcy TP (1987) Evaluation of red cell volume distribution width (RDW). Hematol Pathol 1(2):117–123
Patel KV, Mohanty JG, Kanapuru B et al (2013) Association of the red cell distribution width with red blood cell deformability. Adv Exp Med Biol 765:211–216
Lippi GT, Targher G, Montagnana M et al (2009) Relation between red blood cell distribution width and inflammatory biomarkers in a large cohort of unselected outpatients. Arch Pathol Lab Med 133(4):628–632
Sincer I, Zorlu A, Yilmaz M et al (2012) Relationship between red cell distribution width and right ventricular dysfunction in patients with chronic obstructive pulmonary disease. Heart Lung 41(3):238–243
Oh J, Kang SM, Hong N et al (2009) Relation between red cell distribution width with echocardiographic parameters in patients with acute heart failure. J Card Fail 15(6):517–522
Tonelli M, Sacks F, Arnold M et al (2008) Relation between red blood cell distribution width and cardiovascular event rate in people with coronary disease. Circulation 117(2):163–168
Felker GM, Allen LA, Pocock SJ et al (2007) Red cell distribution width as a novel prognostic marker in heart failure: data from the CHARM Program and the Duke Databank. J Am Coll Cardiol 50(1):40–47
Zorlu A, Bektasoglu G, Guven FM et al (2012) Usefulness of admission red cell distribution width as a predictor of early mortality in patients with acute pulmonary embolism. Am J Cardiol 109(1):128–134
Gong YL, Long X, Jin J et al (2017) Elevation of red cell distribution width during hospitalization predicts mortality in patients with sepsis. Zhonghua Wei Zhong Bing Ji Jiu Yi Xue 29(6):481–485
Otero TMN, Canales C, Yeh DD et al (2016) Elevated red cell distribution width at initiation of critical care is associated with mortality in surgical intensive care unit patients. J Crit Care 34:7–11
Makhoul BF, Khourieh A, Kaplan M et al (2013) Relation between changes in red cell distribution width and clinical outcomes in acute decompensated heart failure. Int J Cardiol 167(4):1412–1416
Kim CH, Park JT, Kim EJ et al (2013) An increase in red blood cell distribution width from baseline predicts mortality in patients with severe sepsis or septic shock. Crit Care 17(6):R282
Zoller B, Melander O, Svensson P, Engstrom G (2014) Red cell distribution width and risk for venous thromboembolism: a population-based cohort study. Thromb Res 133(3):334–339
Yu FT, Armstrong JK, Tripette J, Meiselman HJ, Cloutier G (2011) A local increase in red blood cell aggregation can trigger deep vein thrombosis: evidence based on quantitative cellular ultrasound imaging. J Thromb Haemost 9(3):481–488
Gersh KC, Nagaswami C, Weisel JW (2009) Fibrin network structure and clot mechanical properties are altered by incorporation of erythrocytes. Thromb Haemost 102(6):1169–1175
Celik A, Ozcan IT, Gundes A et al (2015) Usefulness of admission hematologic parameters as diagnostic tools in acute pulmonary embolism. Kaohsiung J Med Sci 31(3):145–149
Akgedik R, Karamanli H, Kurt AB, Gunaydin ZY (2018) Usefulness of admission red blood cell distribution width as a predictor of severity of acute pulmonary embolism. Clin Respir J 12(2):786–794
Gunay E, Sarinic Ulasli S, Kacar E et al (2014) Can platelet indices predict obstruction level of pulmonary vascular bed in patients with acute pulmonary embolism? Clin Respir J 8(1):33–40
Inuzuka R, Abe J (2015) Red blood cell distribution width as a link between ineffective erythropoiesis and chronic inflammation in heart failure. Circ J 79(5):974–975
Allen LA, Felker GM, Mehra MR et al (2010) Validation and potential mechanisms of red cell distribution width as a prognostic marker in heart failure. J Card Fail 16(3):230–238
Cha MJ, Lee HS, Kim HM et al (2017) Association between red cell distribution width and thromboembolic events in patients with atrial fibrillation. Eur J Intern Med 46:41–46
Salvagno GL, Sanchis-Gomar F, Picanza A, Lippi G (2015) Red blood cell distribution width: a simple parameter with multiple clinical applications. Crit Rev Clin Lab Sci 52(2):86–105
Thayer RE, Huang S, Levinson RT et al (2019) Unbiased phenome-wide association studies of red cell distribution width identifies key associations with pulmonary hypertension. Ann Am Thorac Soc. https://doi.org/10.1513/AnnalsATS.201809-594OC
Yardan T, Meric M, Kati C, Celenk Y, Atici AG (2016) Mean platelet volume and mean platelet volume/platelet count ratio in risk stratification of pulmonary embolism. Medicina (Kaunas) 52(2):110–115
Wells PS, Ginsberg JS, Anderson DR et al (1998) Use of a clinical model for safe management of patients with suspected pulmonary embolism. Ann Intern Med 129(12):997–1005
Wicki JP, Perrier A, Perneger TV, Bounameaux H, Junod AF (2000) Prediction adverse outcome in patients with acute pulmonary embolism: a risk score. Thromb Haemost 84:548–552
Calisir C, Yavas US, Ozkan IR et al (2009) Performance of the Wells and revised Geneva scores for predicting pulmonary embolism. Eur J Emerg Med 16(1):49–52
Girardi AM, Bettiol RS, Garcia TS (2018) Wells and Geneva scores are not reliable predictors of pulmonary embolism in critically ill patients: a retrospective study. J Intensive Care Med. https://doi.org/10.1177/0885066618816280
Singh B, Mommer SK, Erwin PJ, Mascarenhas SS, Parsaik AK (2013) Pulmonary embolism rule-out criteria (PERC) in pulmonary embolism–revisited: a systematic review and meta-analysis. Emerg Med J 30(9):701–706
Kucher N, Rossi E, DeRosa M, Goldhaber SZ (2006) Massive pulmonary embolism. Circulation 113(4):577–582
Aujesky D, Obrosky DS, Stone RA et al (2005) Derivation and validation of a prognostic model for pulmonary embolism. Am J Respir Crit Care Med 172(8):1041–1046
Secemsky E, Chang Y, Jain CC et al (2018) Contemporary management and outcomes of patients with massive and submassive pulmonary embolism. Am J Med 131(12):1506–1514
Torbicki A, Perrier A, Konstantinides S et al (2008) Guidelines on the diagnosis and management of acute pulmonary embolism: the task force for the diagnosis and management of acute pulmonary embolism of the European Society of Cardiology (ESC). Eur Heart J 29(18):2276–2315
Jaff MR, McMurtry MS, Archer SL et al (2011) Management of massive and submassive pulmonary embolism, iliofemoral deep vein thrombosis, and chronic thromboembolic pulmonary hypertension: a scientific statement from the American Heart Association. Circulation 123(16):1788–1830
Inonu H, Acu B, Pazarli AC et al (2012) The value of the computed tomographic obstruction index in the identification of massive pulmonary thromboembolism. Diagn Interv Radiol 18(3):255–260
Sunnetcioglu A, Sertogullarindan B, Ozbay B, Asker S, Ekin S (2015) Assessments of the associations of thrombus localization with accompanying disorders, risk factors, D-dimer levels, and the red cell distribution width in pulmonary embolism. Clinics 70(6):441–445
Bucciarelli P, Maino A, Felicetta I et al (2015) Association between red cell distribution width and risk of venous thromboembolism. Thromb Res 136(3):590–594
Rahimtoola A, Bergin JD (2005) Acute pulmonary embolism: an update on diagnosis and management. Curr Probl Cardiol 30(2):61–114
Ozsu S, Abul Y, Gunaydin S, Orem A, Ozlu T (2014) Prognostic value of red cell distribution width in patients with pulmonary embolism. Clin Appl Thromb Hemost 20(4):365–370
Zhou XY, Chen HL, Ni SS (2017) Red cell distribution width in predicting 30-day mortality in patients with pulmonary embolism. J Crit Care 37:197–201
Sen HS, Abakay O, Tanrikulu AC et al (2014) Is a complete blood cell count useful in determining the prognosis of pulmonary embolism? Wien Klin Wochenschr 126(11–12):347–354
Yazici S, Kiris T, Sadik Ceylan U et al (2018) Relation between dynamic change of red cell distribution width and 30-day mortality in patients with acute pulmonary embolism. Clin Respir J 12(3):953–960
Jurin I, Trkulja V, Ajduk M, Letilović T, Hadžibegović I (2019) Red cell distribution width in acute pulmonary embolism patients: a simple aid for improvement of the 30-day mortality risk stratification based on the pulmonary embolism severity index. Heart Lung. https://doi.org/10.1016/j.hrtlng.2019.02.006
Kheirkham-Sabetghadam S, Jenab Y, Ghoreyshi-Hefzabad SM et al (2018) Association between elevated red blood cell distribution width and long-term mortality in acute pulmonary embolism. Turk J Med Sci 48(2):318–323
Yazici S, Siris T, Ceylan US et al (2016) The accuracy of combined use of troponin and red cell distribution width in predicting mortality of patients with acute pulmonary embolism. Wien Klin Wochenschr 128(Suppl 8):596–603
Condliffe R, Kiely DG, Gibbs JS (2008) Improved outcomes in medically and surgically treated chronic thromboembolic pulmonary hypertension. Am J Respir Crit Care Med 177(10):1122–1127
Witkin AS, Channick RN (2015) Chronic thromboembolic pulmonary hypertension: the end result of pulmonary embolism. Curr Cardiol Rep 17(8):63
Abul Y, Ozsu S, Korkmaz A et al (2014) Red cell distribution width: a new predictor for chronic thromboembolic pulmonary hypertension after pulmonary embolism. Chronic Respir Dis 11(2):73–81
Wang W, Liu J, Yang YH et al (2016) Red cell distribution width is increased in chronic thromboembolic pulmonary hypertension. Clin Respir J 10(1):54–60
Forhecz Z, Gombos T, Borgulya G et al (2009) Red cell distribution width in heart failure: prediction of clinical events and relationship with markers of ineffective erythropoiesis, inflammation, renal function, and nutritional state. Am Heart J 158(4):659–666
Montagnana M, Cervellin G, Meschi T, Lippi G (2011) The role of red blood cell distribution width in cardiovascular and thrombotic disorders. Clin Chem Lab Med 50(4):635–641
Kato H, Inshida J, Imagawa S et al (2005) Enhanced erythropoiesis mediated by activation of the renin-angiotensin system via angiotensin II type 1a receptor. FASEB J 19(14):2023–2025
Gossmann J, Burkhardt R, Harder S et al (2001) Angiotensin II infusion increases plasma erythropoietin levels via an angiotensin II type 1 receptor-dependent pathway. Kidney Int 60(1):83–86
Jimenez D, Aujesky D, Moores L et al (2010) Simplification of the pulmonary embolism severity index for prognostication in patients with acute symptomatic pulmonary embolism. Arch Intern Med 170(15):1383–1389
Konstantinides SV, Torbicki A, Agnelli G et al (2014) 2014 ESC guidelines on the diagnosis and management of acute pulmonary embolism. Eur Heart J. https://doi.org/10.1093/eurheartj/ehu283
Jen WY, Jeon YS, Kojodjojo P (2018) A new model for risk stratification of patients with acute pulmonary embolism. Clin Appl Thromb Hemost. https://doi.org/10.1177/1076029618808922
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Disclosures
The authors have no disclosures.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Hammons, L., Filopei, J., Steiger, D. et al. A narrative review of red blood cell distribution width as a marker for pulmonary embolism. J Thromb Thrombolysis 48, 638–647 (2019). https://doi.org/10.1007/s11239-019-01906-w
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-019-01906-w