Abstract
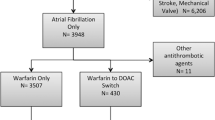
Real-world evidence focusing on medication switching patterns amongst direct oral anticoagulant (DOACs) has not been well studied. The objective of this study is to evaluate patterns of prescription switching in non-valvular atrial fibrillation (NVAF) patients initiated on a DOAC and previously naïve to anticoagulation (AC) therapy. Data was obtained from Truven Health MarketScan® Commercial and Medicare Supplemental database (2009–2013). AC naïve (those without prior anticoagulant use) NVAF patients initiated on a DOAC, with 6 months of continuous health plan enrollment before and after treatment initiation and maintained on continuous therapy for a minimum of 6 months were included. Of 34,022 AC naïve NVAF patients initiating a DOAC, 6613 (19.4%) patients switched from an index DOAC prescription to an alternate anticoagulant and 27,409 (80.6%) remained on the DOAC [age: 68.5 ± 11.7 vs. 67.1 ± 12.7 years, p < 0.001; males: 3781 (57.2%) vs. 17,160 (62.6%), p < 0.001]. Amongst those that switched medication, 3196 (48.3%) did so within the first 6 months of therapy. Overall, 2945 (44.5%) patients switched to warfarin, 2912 (44.0%) switched to another DOAC and 756 (11.4%) switched to an injectable anticoagulant. The highest proportion of patients switched from dabigatran to warfarin (N = 2320; 42.5%) or rivaroxaban (N = 2252; 41.3%). The median time to switch from the index DOAC to another DOAC was 309.5 days versus 118.0 days (p < 0.001) to switch to warfarin. In NVAF patients newly initiated on DOAC therapy, one in five patients switch to an alternate anticoagulant and one of every two patients do so within the first 6 months of therapy. Switching from an initial DOAC prescription to traditional anticoagulants occurs as frequently as switching to an alternate DOAC.

Similar content being viewed by others
References
Connolly SJ, Ezekowitz MD, Yusuf S et al (2009) Dabigatran versus warfarin in patients with atrial fibrillation. N Engl J Med 361(12):1139–1151
Patel MR, Mahaffey KW, Garg J et al (2011) Rivaroxaban versus warfarin in nonvalvular atrial fibrillation. N Engl J Med 365:883–891
Granger CB, Alexander JH, McMurray JJ et al (2011) Apixaban versus warfarin in patients with atrial fibrillation. N Engl J Med 365(11):981–992
Giugliano RP, Ruff CT, Braunwald E et al (2013) Edoxaban versus warfarin in patients with atrial fibrillation. N Engl J Med 369:2093–2104
Spyropoulos AC, Goldenberg NA, Kessler CM, Kittelson J, Schulman S, Turpie AG, Cutler NR, Hiatt WR, Halperin JL, The Antithrombotic Trials Leadership and Steering Group (2012) comparative effectiveness and safety of the novel oral anticoagulants: do the pivotal clinical trials point to a new paradigm? J Thromb Haemost 10(12):2621–2624
Barnes GD, Ageno W, Ansell J, Kaatz S, Subcommittee on the Control of Anticoagulation of the International Society on Thrombosis and Haemostasis (2015) Recommendation on the nomenclature for oral anticoagulants: communication from the SSC of the ISTH. J Thromb Haemost 13:1154–1156
Elewa H, Alhaddad A, Al-Rawi S et al (2017) Trends in oral anticoagulant use in Qatar: a 5-year experience. J Thromb Thrombolysis 43(3):411–416
Barnes GD, Lucas E, Alexander GC et al (2015) National trends in ambulatory oral anticoagulant use. Am J Med 128:1300–1305
Weitz JI, Semchuk W, Turpie AG, Fisher WD, Kong C, Ciaccia A, Cairns JA (2015) Trends in prescribing oral anticoagulants in Canada, 2008–2014. Clin Ther 37(11):2506–2514.e4
Desai NR, Krumme AA, Schneeweiss S, Shrank WH, Brill G, Pezalla EJ, Spettell CM, Brennan TA, Matlin OS, Avorn J, Choudhry NK (2014) Patterns of initiation of oral anticoagulants in patients with atrial fibrillation-quality and cost implications. Am J Med 127(11):1075–1082.e1
Olesen JB, Sørensen R, Hansen ML, Lamberts M, Weeke P et al (2015) Non-vitamin K antagonist oral anticoagulation agents in anticoagulant naïve atrial fibrillation patients: Danish nationwide descriptive data 2011–2013. Europace 17(2):187–193
Huisman MV, Rothman KJ, Paquette M, Teutsch C, Diener HC et al (2015) Antithrombotic treatment patterns in patients with newly diagnosed nonvalvular atrial fibrillation: the GLORIA-AF Registry, Phase II. Am J Med 128(12):1306–1313
Schiavoni M, Margaglione M, Coluccia A (2017) Use of dabigatran and rivaroxaban in non-valvular atrial fibrillation: one-year follow-up experience in an Italian centre. Blood Transfus 31:1–6
Beyer-Westendorf J, Gelbricht V, Förster K, Ebertz F, Röllig D et al (2014) Safety of switching from vitamin K antagonists to dabigatran or rivaroxaban in daily care–results from the Dresden NOAC registry. Br J Clin Pharmacol 78(4)908–917
Clemens A, van Ryn J, Sennewald R, Yamamura N, Stangier J et al (2012) Switching from enoxaparin to dabigatran etexilate: pharmacokinetics, pharmacodynamics, and safety profile. Eur J Clin Pharmacol 68(5):607–616
Bouillon K, Bertrand M, Maura G, Blotière PO, Ricordeau P et al (2015) Risk of bleeding and arterial thromboembolism in patients with non-valvular atrial fibrillation either maintained on a vitamin K antagonist or switched to a non-vitamin K-antagonist oral anticoagulant: a retrospective, matched-cohort study. Lancet Haematol 2(4):e150–e159
Auyeung V, Patel JP, Abdou JK, Vadher B, Bonner L et al (2016) Anticoagulated patient’s perception of their illness, their beliefs about the anticoagulant therapy prescribed and the relationship with adherence: impact of novel oral anticoagulant therapy—study protocol for The Switching Study: a prospective cohort study. BMC Hematol 16(1):22
Attaya S, Bornstein T, Ronquillo N, Volgman R, Braun LT et al (2012) Study of warfarin patients investigating attitudes toward therapy change (SWITCH Survey). Am J Ther 19:432–435
Hale ZD, Kong X, Haymart B, Gu X, Kline-Rogers E et al (2017) Prescribing trends of atrial fibrillation patients who switched from warfarin to a direct oral anticoagulant. J Thromb Thrombolysis 43(2):283–288
Hellfritzsch M, Husted SE, Grove EL, Rasmussen L, Poulsen BK et al (2017) Treatment changes among users of non-vitamin K antagonist oral anticoagulants in atrial fibrillation. Basic Clin Pharmacol Toxicol 120(2):187–194
Truven Health Analytics Health research data for the real world: the MarketScan Databases. Available at http://truvenhealth.com/portals/0/assets/PH_11238_0612_TEMP_MarketScan_WP_FINAL.pdf
Truven Health Analytics Truven health analytics links clinical data with claims to enhance oncology outcomes research. Available at http://truvenhealth.com/news-and-events/press-releases/detail/prid/33/Truven-Health-Analytics-Links-Clinical-Data-with-Claims-to-Enhance-Oncology-Outcomes-Research
Charlson ME, Pompei P, Ales KL, MacKenzie C (1987) A new method of classifying prognostic comorbidity in longitudinal studies: development and validation. J Chronic Dis 40:373–383
Adelborg K, Grove EL, Sundbøll J et al (2016) Sixteen-year nationwide trends in antithrombotic drug use in Denmark and its correlation with landmark studies. Heart 102:1883–1889
Grymonpre R, Cheang M, Fraser M, Metge C, Sitar DS (2006) Validity of a prescription claims database to estimate medication adherence in older person. Med Care 44(5):471–477
Acknowledgements
Department of Pharmacy Systems, Outcomes and Policy (PSOP) and the Center for Pharmacoepidemiology and Pharmacoeconomic Research (CPR) at University of Illinois at Chicago for providing material support to the research. Dr. Galanter is supported by grant U19HS021093 from the Agency for Healthcare Research and Quality. Dr. Nutescu is supported by the National Heart, Lung, And Blood Institute Award Number K23HL112908 and the National Institute on Minority Health and Health Disparities Award Number U54MD010723. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health or the Agency for Healthcare Research and Quality.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Rights and permissions
About this article
Cite this article
Manzoor, B.S., Walton, S.M., Sharp, L.K. et al. High number of newly initiated direct oral anticoagulant users switch to alternate anticoagulant therapy. J Thromb Thrombolysis 44, 435–441 (2017). https://doi.org/10.1007/s11239-017-1565-2
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11239-017-1565-2