Abstract
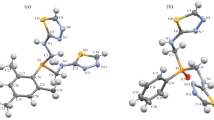
The reaction of 3-phenyl-2-phenylethynyl-1,3,2λ3-oxazaphospholidine with nitrilimines is a multistep process involving formation of the diazaphosphorine ring and cleavage of the oxazaphospholidine ring. The final products are the substituted 4-[N-phenyl-N-(2-chloroethyl)amino]-1,4-dihydro-1,2,4λ5-diazaphosphorines. According to X-ray structural data obtained for 4-[N-phenyl-N-(2-chloroethyl)amino]-1,4-dihydro- 1,5-diphenyl-3-ethoxycarbonyl-1,2,4λ5-diazaphosphorine, the heteroring of these compounds has the conformation of a flattened P-envelope.
Similar content being viewed by others
REFERENCES
Trishin, Yu.G., Gonchar, T.V., and Namestnikov, V.I., Zh. Obshch. Khim., 2004, vol. 74, no.10, p. 1752.
Senyukh, S.V., Namestnikov, V.I., Trishin, Yu.G., and Chistokletov, V.N., Zh. Obshch. Khim., 1990, vol. 60, no.10, p. 1926.
Nifant’ev, E.E. and Vasyanina, L.K., Spektroskopiya YaMR 31 P (31P NMR Spectroscopy), Moscow: Mosk. Gos. Ped. Inst., 1986.
Erofeeva, M.P., Trishin, Yu.G., and Chistokletov, V.N., Zh. Obshch. Khim., 1989, vol. 59, no.9, p. 2146; Namestnikov, V.I., Trishin, Yu.G., Tamm, L.A., and Chistokletov, V.N., Zh. Obshch. Khim., 1990, vol. 60, vol. 3, p. 510; Trishin, Yu.G., Afanasov, A.F., Litvinov, I.A., Naumov, V.A., and Chistokletov, V.N., Zh. Obshch. Khim., 1990, vol. 60, no. 11, p. 2446.
Pudovik, A.N., Pudovik, M.A., Shulyndina, O.S., and Nagaeva, Kh.Kh., Zh. Obshch. Khim., 1970, vol. 40, no.7, p. 1477.
Author information
Authors and Affiliations
Additional information
__________
Translated from Zhurnal Obshchei Khimii, Vol. 75, No. 3, 2005, pp. 406–410.
Original Russian Text Copyright © 2005 by Trishin, Gonchar, Namestnikov, Stash, Zavodnik, Bel’skii
Rights and permissions
About this article
Cite this article
Trishin, Y.G., Gonchar, T.V., Namestnikov, V.I. et al. Synthesis and Molecular Structure of Substituted 4-[N-Phenyl-N-(2-chloroethyl)amino]-1,4-dihydro-1,2,4λ5-diazaphosphorines. Russ J Gen Chem 75, 370–374 (2005). https://doi.org/10.1007/s11176-005-0233-y
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11176-005-0233-y