Abstract
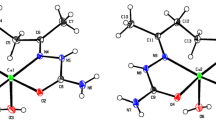
Copper(II) 3-(3′,5′-di-tert-butyl-4′-hydroxybenzyl)acetylacetonate was prepared, and its molecular and crystal structure were determined by single crystal X-ray diffraction. There are two crystallographically independent molecules of the complex in the crystal. In the first molecule, the square planar coordination of Cu(II) is virtually ideal, and in the second molecule it is somewhat distorted. On the whole, the conformations of the independent molecules are similar. The ESR spectrum of the polycrystalline complex suggests magnetic coupling of the Cu atoms and magnetic ordering. In solution in coordination-inert toluene, heating increases the contribution of tetrahedral distortions. In ethanol, the solvent molecule additionally coordinates at the axial positions of the complex.
Similar content being viewed by others
REFERENCES
Milaeva, E.R., Izv. Ross. Akad. Nauk, Ser. Khim., 2001, no. 4, p. 549.
Teoreticheskaya i prikladnaya khimiya β-diketonatov metallov (Theoretical and Applied Chemistry of Metal β-Diketonates), Spitsyn, V.I., Ed., Moscow: Nauka, 1985.
Spek, A.L., Acta Crystallogr., Sect. A, 1990, vol. 46, no.1, p. 34.
Kuska, H.A. and Rogers, M.T., J. Chem. Phys., 1965, vol. 43, no.5, p. 1744.
Zav’yalov, I.A., Milaeva, E.R., and Prokof’ev, A.I., Izv. Ross. Akad. Nauk, Ser. Khim., 1994, no. 1, p. 184.
Altomare, A., Cascarano, G., Giacovazzo, C., and Viterbo, D., Acta Crystallogr., Sect. A, 1991, vol. 47, no.4, p. 744.
Straver, L.H. and Schierbeek, A.J., MolEN. Structure Determination System, vol. 1: Program Description, Nonius B.V., 1994.
Author information
Authors and Affiliations
Additional information
Translated from Zhurnal Obshchei Khimii, Vol. 74, No. 11, 2004, pp. 1775–1781.
Original Russian Text Copyright © 2004 by Pod’‘yachev, Bukharov, Litvinov, Morozov, Gubaidullin, Nugumanova, Mukmeneva.
Rights and permissions
About this article
Cite this article
Pod’‘yachev, S.N., Bukharov, S.V., Litvinov, I.A. et al. Synthesis and structure of copper(II) 3-(3′,5′-Di-tert-butyl-4′-hydroxybenzyl)acetylacetonate. Russ J Gen Chem 74, 1651–1657 (2004). https://doi.org/10.1007/s11176-005-0079-3
Received:
Issue Date:
DOI: https://doi.org/10.1007/s11176-005-0079-3