Abstract
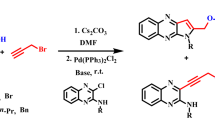
A reaction of 3-(α-chlorobenzyl)quinoxalin-2(1 H)-one with α-picoline gives 2-(3-phenyl-indolizin-2-yl)benzimidazole as a result of a ring-contracting rearrangement involving the fragment C(2)—C(3)—C(Cl)Ph of the quinoxaline system and the fragment N=CH—Me of the α-picoline system. Possible pathways of this reaction are discussed.
Similar content being viewed by others
References
N. G. Brink, F. W. Holly, C. H. Shunk, E. W. Peel, J. J. Cahill, K. Folkers, J. Am. Chem. Soc., 1950, 72, 1866; D. E. Wolf, W. H. Jones, J. Valiant, K. Folkers, J. Am. Chem. Soc. 1950, 72, 2820.
S. Salluja, R. Zou, J. C. Drach, L. B. Townsend, J. Med. Chem., 1996, 39, 881.
H. Zarrinmayeh, D. M. Zimmerman, B. E. Cantrell, D. A. Schober, R. F. Bruns, S. L. Gackenheimer, P. L. Oznstein, P. A. Hipskind, T. C. Britton, D. R. Gehlert, Bioorg. Med. Chem. Lett., 1999, 9, 647.
I. Antonini, F. Claudi, G. Cristalli, P. Franchetti, M. Grifantini, S. Martelli, J. Med. Chem., 1988, 31, 260.
E. B. Skibo, W. G. Schulz, J. Med. Chem., 1993, 36, 3050.
R. Zhou, E. B. Skibo, J. Med. Chem., 1996, 39, 4321.
W. A. Craigo, B. W. LeSueur, E. B. Skibo, J. Med. Chem., 1999, 42, 3324.
R. Cedillo-Rivera, O. Muñoz, J. Med. Microbiol., 1992, 37.
B. Chavez, R. Cedillo-Rivera, A. Martinez-Palomo, J. Protozool., 1992, 39, 510.
S. D. Fears, O’Jare, J. Antimicrob. Agents Chemother., 1998, 32, 144.
A. A. Spasov, I. N. Yoshitsa, L. I. Bugaeva, V. A. Anisimova, Pharm. Chem. J., 1999, 33, 232.
P. J. Taggart, L. R. Cooke, P. C. Mercer, M. W. Shaw, Crop Prot., 1998, 17, 727.
E. S. Lane, J. Chem. Soc., 1955, 1079.
C. T. Brain, S. A. Brunton, Tetrahedron Lett., 2002, 43, 1893.
H. Wagner, J. Org. Chem., 1944, 9, 31.
V. A. Mamedov, I. A. Nuretdinov, F. G. Sibgatullina, Izv. Akad. Nauk SSSR, Ser. Khim., 1989, 1412 [Bull. Acad. Sci. USSR, Div. Chem. Sci. (Engl. Transl.), 1989, 38, 1292].
A. A. Kalinin, O. G. Isaikina, V. A. Mamedov, Khim. Geterotsikl. Soedin., 2004, 1741 [Chem. Heterocycl. Compd. (Engl. Transl.), 2004, 40].
V. A. Mamedov, A. A. Kalinin, A. T. Gubaidullin, I. A. Litvinov, Izv. Akad. Nauk, Ser. Khim., 2007, 2386 [Russ. Chem. Bull., Int. Ed., 2007, 56, 2471].
V. A. Mamedov, D. F. Saifina, I. Kh. Rizvanov, A. T. Gubaidullin, Tetrahedron Lett., 2008, 49, 4647.
G. M. Sheldrick, SADABS, Program for Empirical X- ray Absorption Correction, Bruker-Nonius, 1990–2004.
G. M. Sheldrick, SHELXTL v.6.12, Structure Determination Software Suite, Bruker AXS, Madison (Wisconsin, USA), 2000.
L. J. Farrugia, J. Appl. Crystallogr., 1999, 32, 837.
APEX2 (Version 2.1), SAINTPlus. Data Reduction and Correction Program (Version 7.31 A), Bruker Advanced X- ray Solutions, Bruker AXS, Madison (Wisconsin, USA), 2006.
A. L. Spek, Acta Crystallogr., Sect. A, 1990, 46, 34.
Author information
Authors and Affiliations
Corresponding author
Additional information
Published in Russian in Izvestiya Akademii Nauk. Seriya Khimicheskaya, No. 9, pp. 1924–1928, September, 2009.
Rights and permissions
About this article
Cite this article
Mamedov, V.A., Saifina, D.F., Gubaidullin, A.T. et al. A novel rearrangement in the system 3-[aryl(chloro)methyl]quinoxalin-2(1H)-one— α-picoline as a simple and efficient route to indolizin-2-ylbenzimidazoles. Russ Chem Bull 58, 1986–1990 (2009). https://doi.org/10.1007/s11172-009-0271-4
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11172-009-0271-4