Abstract
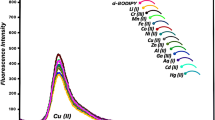
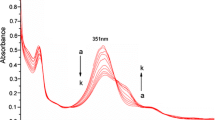
A novel BF2–curcumin-based chemosensor 1, namely monopicolinate of BF2–curcumin complex, was designed, synthesized and applied for the detection of Cu2+ in aqueous buffer solution and living cells. Sensor 1 exhibited sensitive naked-eye color change toward Cu2+ from blue to pink in TBS solution and the detection limit was estimated to be 0.12 µM. The selectivity of sensor 1 for Cu2+ was high over competing metal ions (Ag+, Cu+, Hg2+, Mg2+, Ca2+, Co2+, Zn2+, Mn2+, Ni2+, Fe2+ and Fe3+). Based on the experimental results, the sensing mechanism was proposed for the Cu2+ triggered hydrolysis of 1 to BF2–curcumin which has unique chromogenic and fluorogenic properties. Compared with other chemosensors with a similar mechanism, chemosensor 1 had a comparatively large Stokes shift and the emission wavelength was close to NIR. Moreover, cell imaging investigations indicated that sensor 1 has the potential to be applied for practical Cu2+ detection in biological systems.








Similar content being viewed by others
References
M.C. Linder, M. Hazegh-Azam, Am. J. Clin. Nutr. 63, 797S (1996)
R. Uauy, M. Olivares, M. Gonzalez, Am. J. Clin. Nutr. 67, 952S (1998)
R.A. Festa, D.J. Thiele, Curr. Biol. 21, R877 (2011)
D. Radisky, J. Kaplan, J. Biol. Chem. 274, 4481 (1999)
N.J. Robinson, D.R. Winge, Annu. Rev. Biochem. 79, 537 (2010)
J.L. Burkhead, K.A. Reynolds, S.E. Abdel-Ghany, C.M. Cohu, M. Pilon, New Phytol. 182, 799 (2009)
S. Lutsenko, Curr. Opin. Chem. Biol. 14, 211 (2010)
D.C. Brady, M.S. Crowe, M.L. Turski, G.A. Hobbs, X. Yao, A. Chaikuad, S. Knapp, K. Xiao, S.L. Campbell, D.J. Thiele, C.M. Counter, Nature 509, 492 (2014)
Y.H. Hung, A.I. Bush, R.A. Cherny, J. Biol. Inorg. Chem. 15, 61 (2010)
G. Eskici, P.H. Axelsen, Biochemistry 51, 6289 (2012)
K.M. Davies, J.F.B. Mercer, N. Chen, K.L. Double, Clin. Sci. 130, 565 (2016)
P.C. Bull, G.R. Thomas, J.M. Rommens, J.R. Forbes, D.W. Cox, Nat. Genet. 5, 327 (1993)
E. Gaggelli, H. Kozlowski, D. Valensin, G. Valensin, Chem. Rev. 106, 1995 (2006)
A. Gonzales, M. Firmino, C. Nomura, F. Rocha, P. Oliveira, I. Gaubeur, Anal. Chim. Acta 636, 198 (2009)
M.V. Zoriy, D. Mayer, J.S. Becker, J. Am. Soc. Mass Spectrom. 20, 883 (2009)
Y. Liu, P. Liang, L. Guo, Talanta 68, 25 (2005)
J. Zhuang, L. Zhang, W. Lu, D. Shen, R. Zhu, D. Pan, Int. J. Electrochem. Sci. 6, 4690 (2011)
E.L. Que, D.W. Domaille, C.J. Chang, Chem. Rev. 108, 1517 (2008)
J.A. Cotruvo Jr., A.T. Aron, K.M. Ramos-Torres, C.J. Chang, Chem. Soc. Rev. 44, 4400 (2015)
G.T. Sfrazzetto, C. Satriano, G.A. Tomaselli, E. Rizzarelli, Coord. Chem. Rev. 311, 125 (2016)
A.T. Aron, K.M. Ramos-Torres, J.A. Cotruvo Jr., C.J. Chang, Acc. Chem. Res. 48, 2434 (2015)
Z. Yang, Y. Zhao, S. Chen, Y. Bu, X. Zhu, Y. Du, F. Li, Sens. Actuators B Chem. 235, 414 (2016)
X.T. Chen, A.J. Tong, Dyes Pigments 95, 776 (2012)
Q.Q. Li, M. Peng, N.N. Li, J.G. Qin, Z. Li, Sens. Actuators B Chem. 173, 580 (2012)
C. Zhao, P. Feng, J. Cao, X. Wang, Y. Yang, Y. Zhang, J. Zhang, Y. Zhang, Org. Biomol. Chem. 10, 3104 (2012)
O. Garcia-Beltran, N. Mena, L.C. Friedrich, J.C. Netto-Ferreira, V. Vargas, F.H. Quina, M.T. Nunez, B.K. Cassels, Tetrahedron Lett. 53, 5280 (2012)
D. Li, X. Sun, J. Huang, Q. Wang, Y. Feng, M. Chen, X. Meng, M. Zhu, X. Wang, Dyes Pigments 125, 185 (2016)
L.K. Kumawat, M. Kumar, P. Bhatt, A. Sharma, M. Asif, V.K. Gupta, Sens. Actuators B Chem. 240, 365 (2017)
P. Kaur, S. Kaur, K. Singh, P.R. Sharma, T. Kaur, Dalton Trans. 40, 10818 (2011)
J. Bu, H. Duan, X. Wang, Res. Chem. Intermed. 40, 3119 (2014)
S.O. Tümay, E. Okutan, I.F. Sengul, E. Özcan, H. Kandemir, T. Doruk, M. Çetin, B. Çosut, Polyhedron 117, 161 (2016)
S.K. Sahoo, D. Sharma, A. Moirangthem, A. Kuba, R. Thomas, R. Kumar, A. Kuwar, H.-J. Choi, A. Basu, J. Lumin. 172, 297 (2016)
A. Uslu, S.O. Tümay, A. Şenocak, F. Yuksel, E. Özcan, S. Yeşilot, Dalton Trans. 46, 9140 (2017)
D. Zhu, A. Ren, X. He, Y. Luo, Z. Duan, X. Yan, Y. Xiong, X. Zhong, Sens. Actuators B Chem. 252, 134 (2017)
A. Amalraj, A. Pius, S. Gopi, S. Gopi, J. Tradit. Complement. Med. 7, 205 (2017)
A. Marchiani, C. Rozzo, A. Fadda, G. Delogu, P. Ruzza, Curr. Med. Chem. 21, 204 (2014)
K.I. Priyadarsini, J. Photochem. Photobiol., C 10, 81 (2009)
Z. Pi, J. Wang, B. Jiang, G. Cheng, S. Zhou, Mater. Sci. Eng. C Mater. 46, 565 (2015)
G. Xu, J. Wang, G. Si, M. Wang, B. Wu, S. Zhou, Dyes Pigments 123, 267 (2015)
K. Liu, T.L. Guo, J. Chojnacki, H.G. Lee, X. Wang, S.L. Siedlak, W. Rao, X. Zhu, S. Zhang, ACS Chem. Neurosci. 3, 141 (2012)
K. Park, Y. Seo, M.K. Kim, K. Kim, Y.K. Kim, H. Choo, Y. Chong, Org. Biomol. Chem. 13, 11194 (2015)
X. Zhang, Y. Tian, H. Zhang, A. Kavishwar, M. Lynes, A.L. Brownell, H. Sun, Y.H. Tseng, A. Moore, C. Ran, Sci. Rep. 5, 13116 (2015)
A.D’. Aléo, A. Felouat, F. Fages, Adv. Nat. Sci. Nanosci. Nanotechnol. 6, 015009 (2015)
A. Chaicham, S. Kulchat, G. Tumcharern, T. Tuntulani, B. Tomapatanaget, Tetrahedron 66, 6217 (2010)
G. Xu, J. Wang, G. Si, M. Wang, X. Xue, B. Wu, S. Zhou, Sens. Actuators B Chem. 230, 684 (2016)
H.T. Zhang, R.C. Liu, J. Liu, L. Li, P. Wang, S.Q. Yao, Z.T. Xu, H.Y. Sun, Chem. Sci. 7, 256 (2016)
H. Zhang, L. Feng, Y. Jiang, Y.T. Wong, Y. He, G. Zheng, J. He, Y. Tan, H. Sun, D. Ho, Biosens. Bioelectron. 94, 24 (2017)
L. Tang, P. He, K. Zhong, S. Hou, Y. Bian, Spectrochim. Acta A 169, 246 (2016)
Acknowledgements
We thank the Analytical and Testing Center of Sichuan University for NMR measurements.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Li, Y., Ji, YX., Song, LJ. et al. A novel BF2–curcumin-based fluorescent chemosensor for detection of Cu2+ in aqueous solution and living cells. Res Chem Intermed 44, 5169–5180 (2018). https://doi.org/10.1007/s11164-018-3416-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3416-y