Abstract
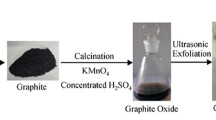
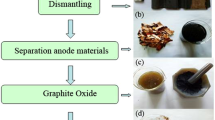
The wide use of lithium ion batteries (LIBs) has created much waste, which has become a global issue. It is vital to recycle waste LIBs considering their environmental risks and resource characteristics. Anode graphite from spent LIBs still possess a complete layer structure and contain some oxygen-containing groups between layers, which can be reused to prepare high value-added products. Given the intrinsic defect structure of anode graphite, copper foils in LIB anode electrodes, and excellent properties of graphene, graphene oxide–copper composite material was prepared in this work. Anode graphite was firstly purified to remove organic impurities by calcination and remove lithium. Purified graphite was used to prepare graphene oxide–copper composite material after oxidation to graphite oxide, ultrasonic exfoliation to graphene oxide (GO), and Cu2+ adsorption. Compared with natural graphite, preparing graphite oxide using anode graphite consumed 40% less concentrated H2SO4 and 28.6% less KMnO4. Cu2+ was well adsorbed by 1.0 mg L−1 stable GO suspension at pH 5.3 for 120 min. Graphene oxide–copper composite material could be successfully obtained after 6 h absorption, 3 h bonding between GO and Cu2+ with 3/100 of GO/CuSO4 mass ratio. Compared to CuO, graphene oxide–copper composite material had better catalytic photodegradation performance on methylene blue, and the electric field further improved the photodegradation efficiency of the composite material.










Similar content being viewed by others
References
L.H. Saw, K. Somasundaram, Y. Ye, A.A.O. Tay, J. Power Sources 249, 231 (2014)
X. Wang, G. Gaustad, C.W. Babbitt, Waste Manag. 51, 204 (2016)
H. Zou, E. Gratz, D. Apelian, Y. Wang, Green Chem. 15(5), 1183 (2013)
X. Zhang, S.C. Han, P.G. Xiao, C.L. Fan, W.H. Zhang, Carbon 100, 600 (2016)
N.C. Gallego, C.I. Contescu, H.M. Meyer, J.Y. Howe, R.A. Meisner, E.A. Payzant, M.J. Lance, S.Y. Yoon, M. Denlinger, D.L. Wood, Carbon 72, 393 (2014)
D.A. Ferreira, L.M.Z. Prados, D. Majuste, M.B. Mansur, J. Power Sources 187(1), 238 (2009)
X. Zeng, J. Li, N. Singh, Crit. Rev. Environ. Sci. Technol. 44(10), 1129 (2014)
L.P. He, S.Y. Sun, Y.Y. Mu, X.F. Song, J.G. Yu, Acs Sustain. Chem. Eng. 5(1), 714 (2017)
J. Guan, Y.G. Li, Y.G. Guo, R.J. Su, G.L. Gao, H.X. Song, H. Yuan, B. Liang, Z.H. Guo, Acs Sustain. Chem. Eng. 5(1), 1026 (2017)
X.X. Zhang, Q. Xue, L. Li, E. Fan, F. Wu, R.J. Chen, Acs Sustain. Chem. Eng. 4(12), 7041 (2016)
X.P. Chen, C.B. Luo, J.X. Zhang, J.R. Kong, T. Zhou, Acs Sustain. Chem. Eng. 3(12), 3104 (2015)
Z. Sun, H. Cao, Y. Xiao, J. Sietsma, W. Jin, H. Agterhuis, Y. Yang, Acs Sustain. Chem. Eng. 5(1), 21 (2017)
J. Rao, P. Zhang, S. He, Z. Li, H. Ma, Z. Shen, S. Miao, Scientia Sinica Technologica. 47(1), 13 (2017)
H.Y. Du Yilun, X. Lei, F. Zhang, China Min. Mag. 24, 28 (2015)
T. Gao, Q. Chen, W. Yu, L. Shen, Resour. Sci. 37(5), 1059 (2015)
B. Moradi, G.G. Botte, J. Appl. Electrochem. 46(2), 123 (2016)
Y. Guo, F. Li, H. Zhu, G. Li, J. Huang, W. He, Waste Manag. 51, 227 (2016)
K.V. Ragavan, N.K. Rastogi, Sens. Actuat. B Chem. 229, 570 (2016)
J. Zheng, W. Zhang, Z. Lin, C. Wei, W. Yang, P. Dong, Y. Yan, S. Hu, J. Mater. Chem. B 4(7), 1247 (2016)
X. Zhang, Y. Hu, D. Zhu, A. Xie, Y. Shen, Ceram. Int. 42(1), 1833 (2016)
H. Yu, B. Zhang, C. Bulin, R. Li, R. Xing, Sci. Rep. 6, 36143 (2016)
R.Y. Wang, Z.W. Wu, Z.F. Qin, C.M. Chen, H.Q. Zhu, J.B. Wu, G. Chen, W.B. Fan, J.G. Wang, Catal. Sci. Technol. 6(4), 993 (2016)
N.S. Yusof, B. Babgi, Y. Alghamdi, M. Aksu, J. Madhavan, M. Ashokkumar, Ultrason. Sonochem. 29, 568 (2016)
S. Stankovich, D.A. Dikin, R.D. Piner, K.A. Kohlhaas, A. Kleinhammes, Y. Jia, Y. Wu, S.T. Nguyen, R.S. Ruoff, Carbon 45(7), 1558 (2007)
T. Soltani, B.K. Lee, J. Colloid Interface Sci. 481, 168 (2016)
R. Rahimi, M. Moshari, M. Rabbani, A. Azad, Rsc Adv. 6(47), 41156 (2016)
H. Qiao, Study on the Structural Transformation and Electrical Properties of Products Formed by the Oxidation-Reduction of Graphite (in Chinese)[D] (Southwest Univerisity, 2012)
D. Li, M.B. Muller, S. Gilje, R.B. Kaner, G.G. Wallace, Nat. Nanotechnol. 3(2), 101 (2008)
Acknowledgements
This work is supported by the Key Research Project of Science and Technology Commission of Shanghai Municipality (12dz2294000). The authors would like to thank the anonymous reviewers for valuable comments.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Zhang, W., Liu, Z., Xu, C. et al. Preparing graphene oxide–copper composite material from spent lithium ion batteries and catalytic performance analysis. Res Chem Intermed 44, 5075–5089 (2018). https://doi.org/10.1007/s11164-018-3410-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-018-3410-4