Abstract
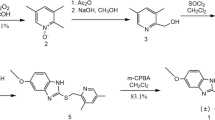
An impurity was isolated from crude synthesized roflumilast and characterized by 1H NMR, 13C NMR, and HR-MS, which confirmed the structure as N-(3,5-dichloropyrid-4-yl)-4-difluoromethoxy-3-hydroxybenzamide. To further verify the structure, this compound was synthesized from 4-difluoromethoxy-3-hydroxybenzaldehyde. Comparison of the 1H NMR, 13C NMR, HR-MS, and the HPLC spectrum of the impurity and the authentic sample indicated that this impurity was N-(3,5-dichloropyrid-4-yl)-4-difluoromethoxy-3-hydroxybenzamide. This demonstrated the significance of monitoring the reaction process of roflumilast.

Similar content being viewed by others
References
M.A. Giembycz, S.K. Field, Drug Des. Dev. Ther. 21(4), 147–158 (2010)
R. Beume, A. Hatelmann, EP2366393, 2006
J. Maus, H. Kastrup, A. Bauhofer, P. Cnota, I. Szelenyi, WO2007071313, 2007
M. Kobayashi, S. Kubo, M. Iwata et al., Int. Immunopharmacol. 11, 732–739 (2011)
A. Hermann, F. Dieter, G. Beate, et al., WO9501338, 1994
L.A. Sorbera, P.A. Leeson, Castañer. J. Drugs Future 25, 1261–1264 (2000)
Y.G. Zhong, G.H. Chen, A. Li, S.Y. Li, Chinese J. Pharm. 42(12), 884–886 (2011)
C.D. Cook, H.R. Jones, H. Kabir, J.D. Lythgoe et al., Org. Process Res. Dev. 2, 157–168 (1998)
B. Kohl, B. Mueller, W. Palosch, WO2004080967, 2004
Y. Lin, S. Liu, L.F. Sima, L.G. Chen, Res. Chem. Intermed. doi:10.1007/s11164-012-0742-3
G.W. Smith, WO2005041864, 2005
P. Venkata, B. Sarala, M. Nagarajan, et al., WO2006117653, 2006
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Lin, Y., Huang, P., Qi, H. et al. Isolation, synthesis and structure confirmation of the impurity in crude roflumilast product. Res Chem Intermed 39, 3111–3115 (2013). https://doi.org/10.1007/s11164-012-0823-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11164-012-0823-3