Abstract
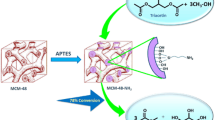
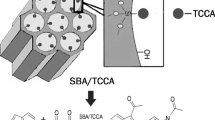
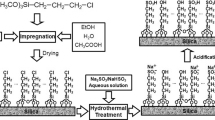
Covalently linked sulfonic acid (–SO3H)-modified ordered mesoporous silicas MCM-48, MCM-41, and SBA-15 were synthesized, characterized and their catalytic activities were evaluated in the transesterification reaction of triacetin with methanol. Acid modified materials were prepared by oxidative transformation of immobilized functionalized unit, 3-mercaptopropyltriethoxysilane (MPTES) as a precursor. The mesophase and porosity of the catalysts were determined by means of X-ray diffraction and N2 adsorption techniques. No degradation of structure was observed in the preparation process. The acid concentrations were calculated using TG–DTA and NH3–TPD analysis. The acid modified materials were found to be active catalysts for the transesterification of triacetin with methanol. Especially, three-dimensional-MCM-48-SO3H showed better catalytic activity compared to its two-dimensional counterparts MCM-41 and SBA-15.










Similar content being viewed by others
References
Zhao D, Feng J, Huo Q, Melosh N, Fredrickson GH, Chmelka BF, Stucky GD (1998) Science 279:548–552
Stein A, Melde BJ, Schrodein RC (2000) Adv Mater 12:1403–1419
Van Rhijn WM, De Vos DE, Bossaert WD, Jacobs PA (1998) Chem Commun 3:317–318
Ide Y, Iwata M, Yagenji Y, Tsunoji N, Sohmiya M, Komaguchi K, Sano T, Sugahara Y (2016) J Mater Chem A 4:15829–15835
Ziarani GM, Badiei A, Mousavi S, Lashgari N, Shahbazi A (2012) Chin J Catal 33:1832–1839
Tsai CT, Pan YC, Ting CC, Vetrivel S, Chiang AST, Fey GTK, Kao HM (2009) Chem Commun 33:5018–5020
Mondal J, Nandi M, Modak A, Bhaumik A (2012) J Mol Catal A 254:363–364
Wu B, Tong Z, Yuan X (2012) J Porous Mater 19:641–647
Canilho N, Jacoby J, Pasc A, Carteret C, Dupire F, Stébé MJ, Blin JL (2013) Colloids Surf B 112:139–145
Shieh FK, Hsiao CT, Wu JW, Sue YC, Bao YL, Liu YH, Wan L, Hsu MH, Deka JR, Kao HM (2013) J Hazard Mater 260:1083–1091
Harmer MA, Sun Q (2001) Appl Catal A 221:45–62
Timofeeva MN, Panchenko VN, Hasan Z, Khan NA, Mel’gunov MS, Abel AA, Matrosova M, Volchod KP, Jhung SH (2014) Appl Catal A 469:427–433
Khan NA, Mishra DK, Ahmed I, Yoon JW, Hwang JS, Jhung SH (2013) Appl Catal A 452:34–38
Timofeeva MN (2003) Appl Catal A 256:19–35
Goestena MG, Juan-Alcañiz J, Ramos-Fernandez EV, Gupta KBSS, Stavitski E, Bekkum HV, Gascon J, Kapteijn F (2011) J Catal 281:177–187
Akiyama G, Matsuda R, Sato H, Takata M, Kitagawa S (2011) Adv Mater 23:3294–3297
Hasan Z, Jhung SH (2014) Eur J Inorg Chem 21:3420–3426
Kureshy RI, Ahmad I, Pathak K, Khan NH, Abdi SHR, Jasra RV (2009) Catal Commun 10:572–575
Zhang G, Zhang X, Lv J, Liu H, Qiu J, Yeung KL (2012) Catal Today 193:221–225
Meziani MJ, Zajac J, Jones DJ, Patyka S, Roziere J, Auroux A (2000) Langmuir 16:2262–2268
Brunel D, Blanc AC, Galarneau A, Fajula F (2002) Catal Today 73:139–152
Dias AS, Pillinger M, Valente AA (2005) J Catal 229:414–423
Malero JA, Stucky GD, Grieken R, Morales G (2002) J Mater Chem 12:1664–1670
Bandyopadhyay M, Shiju NR, Brown DR (2010) Catal Commun 11:660–664
Diaz I, Mohino F, Perez-Pariente J, Sastre E, Wright P, Zhou W (2001) Stud Surf Sci Catal 135:1248–1253
Margolese D, Melero JA, Christiansen SC, Chmelka BF, Stucky GD (2000) Chem Mater 12:2448–2459
Bossaert WD, De Vos DE, Van Rhijn WM, Bullen J, Grobet PJ, Jacobs PA (1999) J Catal 182:156–164
Bender M (1999) Bioresour Technol 70:81–87
Diasakou M, Louloudi A, Papayannakos N (1998) Fuel 77:1297–1302
Ogoshi T, Miyawaki Y (1985) J Am Oil Chem Soc 62:331–335
Suppes GJ, Bockwinkel K, Lucas S, Botts JB, Mason MH, Heppert JA (2001) J Am Oil Chem Soc 78:139–145
Kildiran G, Yucel SO, Turkay S (1996) J Am Oil Chem Soc 73:225–232
Nam LTH, Vinh TQ, Loan NTT, Van Tho DS, Yang X, Su B (2011) Fuel 90:1069–1075
Hara M (2009) Chem Sus Chem 2:109–135
Sharma YC, Singh B (2010) Biofuels, Bioprod Biorefin 5:69–92
Serio MD, Tesser R, Pengmei L, Santacesaria E (2008) Energy Fuels 22:207–217
Diaz I, Mohino F, Perez-Pariente J, Sastre E (2003) Appl Catal A 242:161–169
Alvaro M, Corma A, Das D, Fornes V, Garcia H (2005) J Catal 231:48–55
Mbaraka IK, Radu DR (2003) Y Lin VS, Shanks BH. J Catal 219:329–336
Sayari A, Hamoudi S (2001) Chem Mater 13:3151
Lee AF, Bennett JA, Manayil JC, Wilson K (2014) Chem Soc Rev 43:7887–7916
Shagufta Ahmad I, Dhar R (2017) Catal. Surv. Asia 21:53–69
Bandyopadhyay M, Tsunoji N, Sano T (2017) Catal Lett 147:1040–1050
Gies H, Grabowski S, Bandyopadhyay M, Grunert W, Tkachenko OP, Klementiev KV, Birkner A (2003) Micropor Mesopor Mater 60:31–42
Lesaint C, Lebeau B, Marichal C, Patarin J (2005) Micropor Mesopor Mater 83:76–84
Wang X, Tseng YH, Chan JCC (2007) J Phy Chem C 111:2156–2164
Siril PF, Davison AD, Randhawa JK, Brown DR (2007) J Mol Catal A 267:72–78
Yoshitake H, Yokoi T, Tatsumi T (2002) Chem Mater 14:4603–4610
Li Y, Zhou G, Li C, Qin D, Qiao W, Chu B (2009) Colloids Surf A 341:79–85
Fredman B, Pryde EH, Mounts TL (1984) J Am Oil Chem Soc 61:1638–1643
Silveira JQ, Vargas MD, Ronconi CM (2011) J Mater Chem 21:6034–6039
Molnar A (2011) Chem Rev 111:2251–2320
Molnar A, Papp A (2017) Coord Chem Rev 349:1–65
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Bandyopadhyay, M., Tsunoji, N., Bandyopadhyay, R. et al. Comparison of sulfonic acid loaded mesoporous silica in transesterification of triacetin. Reac Kinet Mech Cat 126, 167–179 (2019). https://doi.org/10.1007/s11144-018-1447-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-018-1447-4