Abstract
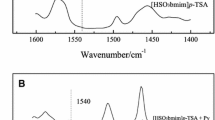
The catalytic performance of acidic ionic liquids (ILs) was evaluated for the hydrolysis of cyclohexyl acetate to cyclohexanol. Cyclohexyl acetate conversion increased with increasing acid strength of the ILs, but the five SO3H-functionalized ILs with different acidity had similarly high activity. All of the ILs exhibited high selectivity to cyclohexanol. This can be attributed to the high polarity of the ILs, which inhibits the formation of the transition state for the side reaction, pyrolysis of cyclohexyl acetate to cyclohexene, and then increases cyclohexanol selectivity. Under optimized conditions, cyclohexyl acetate conversion was 80.2 with 96.6 % cyclohexanol selectivity over [HSO3bmim]HSO4 (bmim = butyl-3-methyl-imidazolium). Moreover, [HSO3bmim]HSO4 also showed high reusability.









Similar content being viewed by others
References
Li J, Yang L, Li F, Xue W, Wang Y (2015) Reac Kinet Mech Cat 114:173–183
Lou S, Xiao C, Sun G, Kou Y (2013) Chin J Catal 34:251–256
Nagahara H, Ono M, Konishi M, Fukuoka Y (1997) Appl Surf Sci 121(122):448–451
Ishida H (1997) Catal Surv Jpn 1:241–246
Imam RA, Freund H, Guit RPM, Fellay C, Meier RJ, Sundmacher K (2013) Org Process Res Dev 17:343–358
Steyer F, Sundmacher K (2007) Ind Eng Chem Res 46:1099–1104
Katariya A, Freund H, Sundmacher K (2009) Ind Eng Chem Res 48:9534–9545
Du W, Xue W, Li F, Wang Y (2012) J Hebei Univ Technol 41:34–39
Yang L (2013) Hydration of Cyclohexene Catalyzed by Brönsted Acidic Ionic Liquid. Master’s Thesis. Hebei University of Technology, China
Chakrabarti A, Sharma MM (1992) React Polym 18:107–115
Kimura M, Nakato T, Okuhara T (1997) Appl Catal A 165:227–240
Hara M, Yoshida T, Takagaki A, Takata T, Kondo JN, Hayashi S, Domen K (2004) Angew Chem Int Ed 43:2955–2958
Yang Q, Kapoor MP, Shirokura N, Ohashi M, Inagaki S, Kondo JN, Domen K (2005) J Mater Chem 15:666–673
Jin J, Li F, Yang L, Zhang D, Xue W, Wang Y (2014) Acta Pet Sin (Pet Process Sect) 30:169–174
Cole AC, Jensen JL, Ntai I, Tran KLT, Weaver KJ, Forbes DC, Davis JH Jr (2002) J Am Chem Soc 124:5962–5963
Gu Y, Shi F, Deng Y (2003) Catal Commun 4:597–601
Kore R, Srivastava R (2011) Catal Commun 12:1420–1424
Liu H, Zeng F, Deng L, Liao B, Pang H, Guo Q (2013) Green Chem 15:81–84
Simirskaya NI, Ignat’ev NV, Schulte M, Zlotin SG (2012) Mendeleev Commun 22:317–319
Luo H, Xue K, Fan W, Li C, Nan G, Li Z (2014) Ind Eng Chem Res 53:11653–11658
Dong C, Geng Y, An H, Zhao X, Wang Y (2013) CIESC J 64:2086–2091
Xu D, Wu J, Luo S, Zhang J, Wu J, Du X, Xu Z (2009) Green Chem 11:1239–1246
Thomazeau C, Olivier-Bourbigou H, Magna L, Luts S, Gilbert B (2003) J Am Chem Soc 125:5264–5265
Acknowledgments
This work was financially supported by National Natural Science Foundation of China (21236001, 21176056), Programme for 100 Excellent Talents in University of Hebei Province (II) (BR2-208) and Natural Science Foundation of Hebei Province (B2015202228).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Yang, F., Xue, W., Zhang, D. et al. Hydrolysis of cyclohexyl acetate to cyclohexanol with high selectivity over SO3H-functionalized ionic liquids. Reac Kinet Mech Cat 117, 329–339 (2016). https://doi.org/10.1007/s11144-015-0909-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-015-0909-1