Abstract
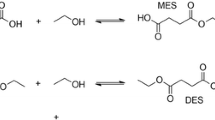
Modified palygorskite with 3-aminopropyltriethoxysilane (KH550) was used as the carrier to immobilize a 12-heteropolyacid of molybdenum and tungsten. The prepared catalyst was characterized by FT-IR, XRD, XPS, BET and TG. The kinetic behavior of heterogeneous esterification of acetic acid with n-amyl alcohol over the catalyst was investigated. The nonideality of the liquid phase was considered by using activities instead of molar fractions. The activity coefficients have been calculated by using the group contribution method UNIFAC. The experimental results showed that the reaction is controlled by chemical steps rather than external and internal mass transfer steps. Experimental data fitted well to the pseudo-homogeneous (PH) model. The temperature dependency of the equilibrium constant and the reaction rate constant were found. It indicated that the heat of reaction is 7.33 kJ mol−1. The new rate equation gave a good fit to the data and was able to describe the behavior of the system.









Similar content being viewed by others
References
Ali SH, Tarakmah A, Merchant SQ et al (2007) Chem Eng Sci 62:3197–3217
Weissermel K, Arpe H-J (1997) Industrial organic chemistry, 3rd edn. VCH, New York, p 289
Venkateswarlu K, Sinha R, Rao RJ (1976) J Chem Petro 3–10
Altiokka MR, Hosgün HL (2007) Ind Eng Chem Res 46:1058–1062
Lilja J, Aumo J, Salmi T et al (2002) Appl Catal A: Gen 228:253–267
Zhang LX, Jin QZ, Shan L et al (2010) Appl Clay Sci 47:229–234
Lilja J, Murzin DY, Salmi T et al (2002) J Mol Catal A: Chem 182–183:555–563
Ju IB, Lim H-W, Lim W et al (2011) J Chem Eng 168:293–302
Altiokka MR, itak AC (2003) Appl Catal A: Gen 239:141–148
Tanabe K, Misono M, Hattori H (1989) New solid acid bases. Kodansha/Elsevier Science, Tokyo/Amsterdam
Ganapati DY, Pranav HM (1994) Ind Eng Chem Res 33:2198–2208
Xu JP, Chuang KT (1996) Can J Chem Eng 74:493–500
Liu W-T, Tan C-S (2001) Ind Eng Chem Res 40:3281–3286
Lee MJ, Wu HT, Lin HM (2000) Ind Eng Chem Res 39:4094–4099
Lee MJ, Wu HT, Kang CH et al (1999) J Chin Inst Chem Eng 30:117–122
Misono M, Mizuno N, Katamura K et al (1982) Bull Chem Soc Jpn 55:400–406
Kekre SY, Gopala RM (1969) Ind Chem Eng 4:115
Yadav GD, Kirthivasan N (1997) Appl Catal A: Gen 154:29–53
Madhusudhan PR, Wolfson A, Kababya S et al (2005) J Catal 232:210–225
Zhang LX, Jin QZ, Huang JH et al (2010) Appl Surf Sci 256:5911–5917
Baronett G, Thomas H, Querin CA (2001) Appl Catal A: Gen 217:131–141
Ruan YH, Liu YF, Liu ZC (2004) J Chin Catal 25:948–954
Tarlani A, Abedini M, Khabaz M et al (2005) J Colloid Interf Sci 292:486–492
Madhusudhan RP, Wolfson A, Kababya S et al (2005) J Catal 232:210–225
Pizzio LR, Caceres CV, Balanco MN (1998) Appl Catal A: Gen 167:283–287
Chakrabarti A, Sharma MM (1993) React Polym 20:1–45
Ahmed OS, Dutta DK (2003) Thermochim Acta 395:209–216
Zhang HB, Zhang K, Yuan ZY et al (1998) J Nat Gas Chem 7:336–345
Lu JL, Li XH, Yang SJ (2002) Appl Chem Ind 31:8–9
Dong JH, Yang SJ (2003) Chem Propellant Polym Mater 1:18–19
Sun MZ, Qi YT, Yuan XD et al (2003) Speciality Petrochem 1:4–7
Jin L, Zhu L (2011) Chem Bioeng 28:51–53
Yu W, Hidajat K, Ray AK (2004) Appl Catal A: Gen 260:191–205
Darge O, Thyrion FC (1993) J Chem Technol Biotechnol 58:351–355
Qu YX, Peng SJ, Wang S et al (2009) J Chin Chem Eng 17:773–780
Seo Y, Hong WH (2000) J Chem Eng Jpn 33:128–133
Teo HTR, Saha B (2004) J Catal 228:174–182
Mostafa V, Gholamreza V-N, Morteza A (2012) Can J Chem Eng 3:37–44
Aage F, Russell LJ, John MP (1975) J AIChE 21:1086–1099
Emine S, Ferhan SA (2010) Reac Kinet Mech Cat 99:125–134
Moffat JB (2001) Metal-oxygen clusters, surface and catalytic properties of heteropoly oxometalates. Kluwer, New York
ElifÖdes A, Mehmet RA (2011) Appl Catal A: Gen 396:14–19
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Li, G., Mu, R., Fan, Z. et al. Kinetic study of the esterification of acetic acid and n-amyl alcohol catalyzed by H3PW6Mo6O40 immobilized on silylated palygorskite. Reac Kinet Mech Cat 110, 163–175 (2013). https://doi.org/10.1007/s11144-013-0595-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-013-0595-9