Abstract
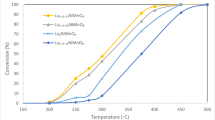
Perovskite-type La0.8Sr0.2CoO3 mixed oxides were prepared by d,l-alanine solution combustion synthesis and used successfully in CH4 combustion as catalysts. These samples were characterized by means of XRD, FTIR, BET, and H2-TPR methods. The effects of stoichiometric ratio (φ) of organic fuel to oxidizer on the structure and catalytic activities of the catalysts were studied. The results indicate that all La0.8Sr0.2CoO3 mixed oxides with different φ have perovskite structures. Their structures and catalytic activities vary along with the change of φ. The catalytic activity of La0.8Sr0.2CoO3 mixed oxide with φ = 1.52 is the best among all the samples, whose T 50 and T 100 (the temperatures of methane conversions reaching 50 and 100%, respectively) are respectively 470 °C and 550 °C, which can be explained in terms of the smaller of average crystal size, higher specific surface area, bigger lattice distortion, lower activation energy, and higher mobility of chemically adsorbed oxygen on the surface and vacancy of the catalysts.





Similar content being viewed by others
References
Narjés, H.B., Pierre, D., Habib, B.: Physicochemical and catalytic properties in methane combustion of La1−xCaxMnO3±y (0 ≤ x ≤ 1; −0.04 ≤ y ≤ 0.24) perovskite-type oxide. Appl. Catal. A. 282, 173–180 (2005)
Uenishi, M., Tanaka, H., Taniguchi, M., Tan, I.: Photo-induced suppression of ferroelectric transition in oxygen-isotope-exchanged SrTiO3. Appl. Catal. A. 296, 114–119 (2005)
Dai, H.X., He, H., Li, P.H., Gao, L.Z., Au, C.: The relationship of structural defect–redox property–catalytic performance of perovskites and their related compounds for CO and NOx removal. Catal. Today 90, 231–214 (2004)
Civera, A.: Combustion synthesis of perovskite-type catalysis for nature gas combustion. Catal. Today 83, 199–211 (2003)
Belessi, V.C., Bakas, T.V., Costa, C.N.: Synergistic effects of crystal phases and mixed valences in La–Sr–Ce–Fe–O mixed oxidic/perovskitic solids on their catalytic activity for the NO+CO reaction. Appl. Catal.B. 28, 13–28 (2000)
Kwang-Sup, S., Cui, H.X., Kim, S.D.: Catalytic combustion of CH4 and CO on La1−xMxMnO3 perovskites. Catal. Today 47, 155–160 (1999)
Kenichirou, U., Yasuyoshi, S., Yoshio, M.: Catalytic combustion of CH4 and CO on La1−xMxMnO3 perovskites. Thermochim. Acta. 431, 117–122 (2005)
Berger, D., Fruth, V., Jitaru, I., Schoonman, J.: Synthesis and characterisation of La1-xSrxCoO3 with large surface area. Mater. Lett. 58, 2418–2422 (2004)
Wachowski, L.: Influence of the method of preparation on the porous structure of perovskite oxides. Surf. Coat. Tech. 29, 303–311 (1986)
Arai, H., Yamada, T., Eguchi, K.: Catalytic combustion of methane over various peroskite-type oxides. Appl. Catal. 26, 265–271 (1986)
Alifanti, M., Kirchnerova, J., Delmonb, B.: Methane and propane combustion over lanthanum transition- metalperovskites: role of oxygen mobility. Appl. Catal. A. 262, 167–176 (2004)
Lal, B., Raghunandan, M.K., Gupta, M., Singh, R.N.: Characterization of precursors and reactivity of LaNi1-xCoxO3 for the partial oxidation of methane. Int. J. Hydrogen Energy 30, 723–730 (2005)
Ladavos, A.K., Pomonis, P.J.: Methane combustion on La–Sr–Ce–Fe–O mixed oxides: bifunctional synergistic action of SrFeO3 and CeOx phases. J. Chem. Soc. 88, 2557–2565 (1992)
Saracco, G., Geobaldo, F., Baldi, G.: Methane combustion on Mg-doped LaMnO3 perovskie catalysts[J]. Appl. Catal. B 20, 277–288 (1999)
Klvana, D., Vaillancourt, J., Kirchnerova, J., Chauki, J.: Surface properties and catalytic performance of La1−xSrxFeO3 perovskite-type oxides for methane combustion. Appl. Catal. A 109, 181–197 (1994)
Acknowledgments
The work described above was fully supported by a grant from the Nanchang University and Nanchang Hangkong University.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, Y., Luo, L. & Liu, W. Effect of stoichiometric ratio of organic fuel to oxidizer on performance of La0.8Sr0.2CoO3 catalysts for CH4 combustion. React Kinet Catal Lett 97, 59–67 (2009). https://doi.org/10.1007/s11144-009-0019-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11144-009-0019-z