Abstract
Background
We aimed to determine the utility of a patient-reported outcome (PRO) as it relates to patient performed testing (PPT) for measuring functional status in multiple myeloma patients after autologous hematopoietic stem cell transplantation (auto-HCT).
Methods
Symptom interference on walking (a PRO) was measured by the MD Anderson Symptom Inventory (MDASI). PPT was assessed via 6-min walk test (6MWT). Mixed effects modeling was used to examine (1) the longitudinal relationship between the MDASI score and 6MWT distance and (2) the MDASI scores between patients who did or did not complete the 6WMT. Receiver operating characteristic (ROC) curve analysis was performed to quantify the construct validity of the PRO by differentiating performance status.
Results
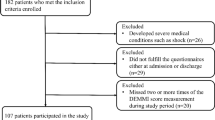
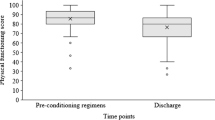
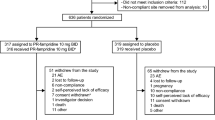
Seventy-nine patients were included. Mean 6MWT distance significantly correlated with MDASI-walking interference score (PRO) over the first month of auto-HCT (est = 6.09, p = 0.006). There was a significantly higher completion rate for MDASI versus 6MWT at each time point (p < 0.01). Patients who completed the 6MWT reported less interference on walking during the study period (est = 1.61, p < 0.0001). Finally, the PRO demonstrated significant construct validity for measuring functioning status with MDASI-walking against ECOG-PS as the anchor (AUC = 0.77, 95% CI 0.60–0.94, p = 0.003).
Conclusion
The PRO of MDASI-walking interference is a valid physical functioning measure, correlating with an objective functional measure (6MWT) in MM patients undergoing auto-HCT. As patients with poorer functional status during therapy are less likely to complete PPT, this PRO may offer a more practical quantitative measure of functioning in patients.



Similar content being viewed by others
References
Deshpande, P. R., Rajan, S., Sudeepthi, B. L., & Abdul Nazir, C. P. (2011). Patient-reported outcomes: A new era in clinical research. Perspectives in Clinical Research, 2(4), 137–144.
Kotronoulas, G., Kearney, N., Maguire, R., Harrow, A., Di Domenico, D., Croy, S., & MacGillivray, S. (2014). What is the value of the routine use of patient-reported outcome measures toward improvement of patient outcomes, processes of care, and health service outcomes in cancer care? A systematic review of controlled trials. Journal of Clinical Oncology, 32(14), 1480–1501.
Barney, B. J., Wang, X. S., Lu, C., Liao, Z., Johnson, V. E., Cleeland, C. S., & Mendoza, T. R. (2013). Prognostic value of patient-reported symptom interference in patients with late-stage lung cancer. Quality of Life Research, 22(8), 2143–2150.
Penson, R. T., Huang, H. Q., Wenzel, L. B., Monk, B. J., Stockman, S., Long, H. J., 3rd, Ramondetta, L. M., Landrum, L. M., Oaknin, A., Reid, T. J., Leitao, M. M., Method, M., Michael, H., & Tewari, K. S. (2015). Bevacizumab for advanced cervical cancer: patient-reported outcomes of a randomised, phase 3 trial (NRG Oncology-Gynecologic Oncology Group protocol 240). The Lancet Oncology, 16(3), 301–311.
Mesa, R. A., Gotlib, J., Gupta, V., Catalano, J. V., Deininger, M. W., Shields, A. L., Miller, C. B., Silver, R. T., Talpaz, M., Winton, E. F., Harvey, J. H., Hare, T., Erickson-Viitanen, S., Sun, W., Sandor, V., Levy, R. S., Kantarjian, H. M., & Verstovsek, S. (2013). Effect of ruxolitinib therapy on myelofibrosis-related symptoms and other patient-reported outcomes in COMFORT-I: a randomized, double-blind, placebo-controlled trial. Journal of Clinical Oncology, 31(10), 1285–1292.
Tefferi, A., Hudgens, S., Mesa, R., Gale, R. P., Verstovsek, S., Passamonti, F., Cervantes, F., Rivera, C., Tencer, T., & Khan, Z. M. (2014). Use of the Functional Assessment of Cancer Therapy—Anemia in persons with myeloproliferative neoplasm-associated myelofibrosis and anemia. Clinical Therapeutics, 36(4), 560–566.
Wang, X. S., Shi, Q., Shah, N. D., Heijnen, C. J., Cohen, E. N., Reuben, J. M., Orlowski, R. Z., Qazilbash, M. H., Johnson, V. E., Williams, L. A., Mendoza, T. R., & Cleeland, C. S. (2014). Inflammatory markers and development of symptom burden in patients with multiple myeloma during autologous stem cell transplantation. Clinical Cancer Research, 20(5), 1366–1374.
Lee, S. J. (2004). Patient-reported outcomes in multiple myeloma. Journal of the National Comprehensive Cancer Network, 2(4), 379–383.
King, T. A., King, M. T., & White, K. J. (2017). Patient reported outcomes in optimizing myeloma patients’ health-related quality of life. Seminars in Oncology Nursing, 33(3), 299–315.
Wang, X. S., Shi, Q., Williams, L. A., Shah, N. D., Mendoza, T. R., Cohen, E. N., Reuben, J. M., Cleeland, C. S., & Orlowski, R. Z. (2015). Longitudinal analysis of patient-reported symptoms post-autologous stem cell transplant and their relationship to inflammation in patients with multiple myeloma. Leukemia & Lymphoma, 56(5), 1335–1341.
Richardson, P. G., Barlogie, B., Berenson, J., Singhal, S., Jagannath, S., Irwin, D., Rajkumar, S. V., Srkalovic, G., Alsina, M., Alexanian, R., Siegel, D., Orlowski, R. Z., Kuter, D., Limentani, S. A., Lee, S., Hideshima, T., Esseltine, D. L., Kauffman, M., Adams, J., Schenkein, D. P., & Anderson, K. C. (2003). A phase 2 study of bortezomib in relapsed, refractory myeloma. New England Journal of Medicine, 348(26), 2609–2617.
Cleeland, C. S., Mendoza, T. R., Wang, X. S., Chou, C., Harle, M. T., Morrissey, M., & Engstrom, M. C. (2000). Assessing symptom distress in cancer patients: the M.D. Anderson Symptom Inventory. Cancer, 89(7), 1634–1646.
Shi, Q., Wang, X. S., Vaporciyan, A. A., Rice, D. C., Popat, K. U., & Cleeland, C. S. (2016). Patient-reported symptom interference as a measure of postsurgery functional recovery in lung cancer. Journal of Pain and Symptom Management, 52, 822–831
Jones, D., Zhao, F., Fisch, M. J., Wagner, L. I., Patrick-Miller, L. J., Cleeland, C. S., & Mendoza, T. R. (2014). The validity and utility of the MD Anderson Symptom Inventory in patients with prostate cancer: evidence from the Symptom Outcomes and Practice Patterns (SOAPP) data from the Eastern Cooperative Oncology Group. Clinical Genitourinary Cancer, 12(1), 41–49.
Jones, D., Vichaya, E. G., Wang, X. S., Williams, L. A., Shah, N. D., Thomas, S. K., Johnson, V. E., Champlin, R. E., Cleeland, C. S., & Mendoza, T. R. (2013). Validation of the M. D. Anderson Symptom Inventory multiple myeloma module. Journal of Hematology & Oncology, 6, 13.
Overcash, J. (2015). Assessing the functional status of older cancer patients in an ambulatory care visit. Healthcare, 3(3), 846–859.
Miyamoto, S., Nagaya, N., Satoh, T., Kyotani, S., Sakamaki, F., Fujita, M., Nakanishi, N., & Miyatake, K. (2000). Clinical correlates and prognostic significance of six-minute walk test in patients with primary pulmonary hypertension. Comparison with cardiopulmonary exercise testing. American Journal of Respiratory and Critical Care Medicine, 161(2 Pt 1), 487–492.
Ko, V., Naylor, J. M., Harris, I. A., Crosbie, J., & Yeo, A. E. (2013). The six-minute walk test is an excellent predictor of functional ambulation after total knee arthroplasty. BMC Musculoskeletal Disorders, 14, 145.
Jones, L. W., Devlin, S. M., Maloy, M. A., Wood, W. A., Tuohy, S., Espiritu, N., Aquino, J., Kendig, T., Michalski, M. G., Gyurkocza, B., Schaffer, W. L., Ali, B., Giralt, S., & Jakubowski, A. A. (2015). Prognostic importance of pretransplant functional capacity after allogeneic hematopoietic cell transplantation. The Oncologist, 20(11), 1290–1297.
Shah, N., Shi, Q., Williams, L. A., Mendoza, T. R., Wang, X. S., Reuben, J. M., Dougherty, P. M., Bashir, Q., Qazilbash, M. H., Champlin, R. E., Cleeland, C. S., & Giralt, S. A. (2016). Higher stem cell dose infusion after intensive chemotherapy does not improve symptom burden in older patients with multiple myeloma and amyloidosis. Biology of Blood and Marrow Transplantation, 22(2), 226–231.
Castel, L. D., Abernethy, A. P., Li, Y., Depuy, V., Saville, B. R., & Hartmann, K. E. (2007). Hazards for pain severity and pain interference with daily living, with exploration of brief pain inventory cutpoints, among women with metastatic breast cancer. Journal of Pain and Symptom Management, 34(4), 380–392.
Oken, M. M., Creech, R. H., Tormey, D. C., Horton, J., Davis, T. E., McFadden, E. T., & Carbone, P. P. (1982). Toxicity and response criteria of the Eastern Cooperative Oncology Group. American Journal of Clinical Oncology, 5(6), 649–655.
Terwee, C. B., Bot, S. D., de Boer, M. R., van der Windt, D. A., Knol, D. L., Dekker, J., Bouter, L. M., & de Vet, H. C. (2007). Quality criteria were proposed for measurement properties of health status questionnaires. Journal of Clinical Epidemiology, 60(1), 34–42.
Shi, Q., Mendoza, T. R., Wang, X. S., & Cleeland, C. S. (2016). Using a symptom-specific instrument to measure patient-reported daily functioning in patients with cancer. European Journal of Cancer, 67, 83–90.
Wood, W. A., Le-Rademacher, J., Syrjala, K. L., Jim, H., Jacobsen, P. B., Knight, J. M., Abidi, M. H., Wingard, J. R., Majhail, N. S., Geller, N. L., Rizzo, J. D., Fei, M., Wu, J., Horowitz, M. M., & Lee, S. J. (2016). Patient-reported physical functioning predicts the success of hematopoietic cell transplantation (BMT CTN 0902). Cancer, 122(1), 91–98.
Sorror, M. L., Maris, M. B., Storb, R., Baron, F., Sandmaier, B. M., Maloney, D. G., & Storer, B. (2005). Hematopoietic cell transplantation (HCT)-specific comorbidity index: a new tool for risk assessment before allogeneic HCT. Blood, 106(8), 2912–2919.
Jones, L. W., Hornsby, W. E., Goetzinger, A., Forbes, L. M., Sherrard, E. L., Quist, M., Lane, A. T., West, M., Eves, N. D., Gradison, M., Coan, A., Herndon, J. E., & Abernethy, A. P. (2012). Prognostic significance of functional capacity and exercise behavior in patients with metastatic non-small cell lung cancer. Lung Cancer, 76(2), 248–252.
Baumann, F. T., Kraut, L., Schule, K., Bloch, W., & Fauser, A. A. (2010). A controlled randomized study examining the effects of exercise therapy on patients undergoing haematopoietic stem cell transplantation. Bone Marrow Transplantation, 45(2), 355–362.
Dimeo, F., Fetscher, S., Lange, W., Mertelsmann, R., & Keul, J. (1997). Effects of aerobic exercise on the physical performance and incidence of treatment-related complications after high-dose chemotherapy. Blood, 90(9), 3390–3394.
Izano, M., Satariano, W. A., Hiatt, R. A., & Braithwaite, D. (2013). The impact of functional limitations on long-term outcomes among African-American and white women with breast cancer: a cohort study. British Medical Journal Open, 3(10), e003232.
Acknowledgements
This work was supported in part by the National Cancer Institute of the National Institutes of Health through The University of Texas MD Anderson Cancer Center’s Cancer Center Support Grant, P30 CA016672 (PI: RA DePinho) and program project P01 CA124787 (PI: Charles S. Cleeland).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The MD Anderson Symptom Inventory (MDASI) is copyrighted and licensed by The University of Texas MD Anderson Cancer Center and by Charles S. Cleeland. The authors declare no other conflicts of interest in this work.
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards. This study was approved by the M.D. Anderson Institutional Review Board.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Electronic supplementary material
Below is the link to the electronic supplementary material.
11136_2017_1759_MOESM1_ESM.pdf
Supplementary Figure 1. Example of MDASI daily symptom interference on module. Question 25 (red arrow) asked patients to rate interference with “walking” on a 0-10 scale (PDF 15 KB)
11136_2017_1759_MOESM2_ESM.tif
Supplementary Figure 2. Kaplan-Meier analysis of overall survival, comparing patients who were able to perform the 6MWT versus those who were not at 5 days after auto-HCT. There was no significant difference in median overall survival between these groups (TIF 24917 KB)
Rights and permissions
About this article
Cite this article
Shah, N., Shi, Q., Giralt, S. et al. Utility of a patient-reported outcome in measuring functional impairment during autologous stem cell transplant in patients with multiple myeloma. Qual Life Res 27, 979–985 (2018). https://doi.org/10.1007/s11136-017-1759-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-017-1759-2