Abstract
Objective
To develop a new questionnaire with good psychometric properties to measure satisfaction with medical care in patients with non-valvular atrial fibrillation.
Method
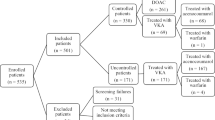
The initial instrument was composed of 37 items, arranged in 6 dimensions: efficacy, ease and convenience, impact on daily activities, satisfaction with medical care, undesired effects of medication, and overall satisfaction. Items and dimensions were extracted from reviewing existing instruments, 3 focus groups with chronic patients, and a panel of 8 experts. Additionally, 3 visual analog scales measuring quality of life, effectiveness, and overall satisfaction were administered. A convenience sample of 119 patients was used for item reduction. Classic psychometric theory and item analysis techniques were used (exploratory factor and confirmatory factor analysis, test–retest, and correlation with visual scales). A validation sample of 230 patients was used to assess convergent validity, and an additional 220 patients sample was used to discriminate between treatment and compliance groups.
Results
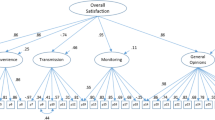
The questionnaire was reduced in length to 25 items, but the impact dimension had split in treatment inconvenience and treatment control. Overall reliability was high (α = 0.861) with acceptable dimensional reliabilities (α = 0.764–0.908). Individual dimensions correlated to varying degrees. Test–retest correlations were high (r = 0.784–0.965), and correlations with visual and already validated scales were substantial. Differences were detected between antivitamin K and new-oral-anticoagulant treatments in several dimensions (p < 0.05). Treatment satisfaction was related with compliance.
Conclusion
This new 25-item questionnaire has good psychometric properties for measuring satisfaction with medical care in patients with this condition. It is capable of detecting differences between different treatments.



Similar content being viewed by others
References
Wolf, P. A., Abbott, R. D., & Kennel, W. B. (1991). Atrial fibrillation as an independent risk factor for stroke: The Framinghan study. Stroke, 22, 983–988.
Albers, G. W., Dalen, J. E., Laupacis, A., Manning, W. J., Petersen, P., & Singer, D. E. (2001). Antitrombotic therapy in atrial fibrillation. Chest, 119(Suppl 1), 194S–206S.
Holbrook, A. M., Pereira, J. A., Labiris, R., McDonald, H., Douketis, J. D., Crowther, M., et al. (2005). Systematic overview of warfarin and its drug and food interaction. Archives of Internal Medicine, 165, 1095–1106.
Katsnelson, M., Sacco, R. L., & Moscucci, M. (2012). Progress for stroke prevention with atrial fibrillation: Emergence of alternative OACs. Stroke, 43, 1179–1185.
Rummel, R. J. (1970). Applied factor analysis. Evanston: Northwestern University Press.
Boomsma, A. (1985). Nonconvergence, improper solutions, and starting values in LISREL maximum likelihood estimation. Psychometrika, 52, 345–370.
Marsh, H. W., & Hau, K.-T. (1999). Confirmatory factor analysis: Strategies for small sample sizes. In R. H. Hoyle (Ed.), Statistical strategies for small sample size (pp. 251–306). Thousand Oaks: Sage.
Wang, J., & Wang, X. (2012). Structural equation modeling. Applications using Mplus. Hoboken: Wiley.
Anderson, J. C., & Gerbing, D. W. (1998). Structural equation modeling in practice: A review and recommended two step approach. Psychological Bulletin, 103, 411–423.
Ding, L., Velicer, W. F., & Harlow, L. L. (1995). Effects of estimation methods, number of indicators per factor, and improper solutions on structural equation modeling fit indices. Structural Equation Modeling, 2, 119–144.
Tabachnick, B. G., & Fidell, L. S. (2001). Using multivariate statistics (4th ed.). Boston: Allyn & Bacon.
Hoogland, J. J., & Boomsma, A. (1998). Robustness studies in covariance structure modeling: An overview and a meta-analysis. Sociological Methods and Research, 26, 329–367.
Kline, R. B. (2005). Principles and practice of structural equation modeling (2nd ed.). New York: Guilford.
Ruiz, M. A., Pardo, A., Rejas, J., Soto, J., Villasante, F., & Aranguren, J. L. (2008). Development and validation of the “Treatment Satisfaction with Medicines Questionnaire” (SATMED-Q)©. Value Health, 11(5), 913–926.
Atkinson, M. J., Sinha, A., Hass, S. L., Colman, S. S., Kumar, R. N., & Rowland, C. R. (2004). Validation of a general measure of treatment satisfaction, the Treatment Satisfaction Questionnaire for Medications (TSQM) using a National Panel Study of Chronic Disease. Health Qual Life Outcomes, 2, 12–25.
Morisky, D. E., Green, L. W., & Levine, D. M. (1986). Concurrent and predictive validity of a self-reported measure of medication adherence. Medical Care, 24, 67–74.
Samsa, G., Matchar, D. B., Dolor, R. J., Wiklund, I., Hedner, E., Wygant, G., et al. (2004). A new instrument for measuring anticoagulation-related quality of life: Development and preliminary validation. Health Qual Life Outcomes, 2, 22.
Cano, S. J., Lamping, D. L., Bamber, L., & Smith, S. (2012). The Anti-Clot Treatment Scale (ACTS) in clinical trials: Cross-cultural validation in venous thromboembolism patients. Health Qual Life Outcomes, 10, 120.
Prins, M. H., Marrel, A., Carita, P., Anderson, D., Bousser, M. G., Crijns, H., et al. (2009). Multinational development of a questionnaire assessing patient satisfaction with anticoagulant treatment: The ‘Perception of Anticoagulant Treatment Questionnaire’ (PACT-Q©). Health Qual Life Outcomes, 7, 9.
Sánchez, R., Yanes, M., Cabrera, A., Ferrer, J. M., Álvarez, R., & Barrera, E. (2004). Adaptación transcultural de un cuestionario para medir la calidad de vida de los pacientes con anticoagulación oral. Atencion Primaria, 34(7), 353–359.
Spertus, J., Dorian, P., Bubien, R., Lewis, S., Godejohn, D., Reynolds, M. R., et al. (2011). Development and validation of the Atrial Fibrillation Effect on QualiTy-of-Life (AFEQT) questionnaire in patients with atrial fibrillation. Circulation Arrhythmia and Electrophysiology, 4, 15–25.
Badia, X., Arribas, F., Ormaetxe, J. M., Peinado, R., & de Los Terreros, M. S. (2007). Development of a questionnaire to measure health-related quality of life (HRQoL) in patients with atrial fibrillation (AF-QoL). Health Qual Life Outcomes, 4(5), 37.
Elewa, H. F, Deremer, C. E., Keller, K., Gujral, J., & Joshua, T. V. (2013). Patient satisfaction with warfarin and willingness to switch to dabigatran: A patient survey. Journal of Thrombosis and Thrombolysis Aug 6. [Epub ahead of print].
Coleman, C. I., Coleman, S. M., Vanderpoel, J., Nelson, W., Colby, J. A., Scholle, J. M., et al. (2013). Patient satisfaction with warfarin- and non-warfarin-containing thromboprophylaxis regimens for atrial fibrillation. Journal of Investigative Medicine, 61(5), 878–881.
Gage, B. F., Cardinalli, A. B., & Owens, D. K. (1996). The effect of stroke and stroke prophylaxis with aspirin or warfarin on quality of life. Archives of Internal Medicine, 156, 1829–1836.
Osterberg, L., & Blaschke, T. (2005). Adherence to medication. New England Journal of Medicine, 353, 487–497.
Barbosa, C. D., Balp, M. M., Kulich, K., et al. (2012). A literature review to explore the link between treatment satisfaction and adherence, compliance, and persistence. Patient Prefer Adherence, 6, 39–48.
Revicki, D. A. (2004). Patient assessment of treatment satisfaction: Methods and practical issues. Gut, 53, iv40–44.
Lindhiem, O., Bennett, C., Trentacosta, C., & McLear, C. (2014). Client preferences affect treatment satisfaction, completion, and clinical outcome: A meta-analyis. Clinical Psychology Review, 34, 506–517.
Acknowledgements
We acknowledge the contribution of the following clinicians for their help with gathering information by interviewing their patients: Ramón Bover, Álvaro Aceña, Ana Pello, Rocío Carda, Óscar González Lorenzo, Angélica Romero, Julia Palfy. This research was funded by an unrestricted grant by Pfizer Spain S.L.U.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
The Helsinki-Tokyo-Venice guidelines for human research were followed. Signed informed consent and permission to use personal records were obtained from all participating patients. The study protocol was approved by the Clinical Research Committees of the Universidad Autónoma de Madrid and the Hospital Clínico de Madrid.
Conflict of interest
This research was funded by an unrestricted grant by Pfizer SLU—Spain. Javier Soto and Marina de Salas-Cansado were employees of Pfizer SLU. All authors declare they do not have any conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Ruiz, M.A., González-Porras, J.R., Aranguren, J.L. et al. Development and validation of a new questionnaire measuring treatment satisfaction in patients with non-valvular atrial fibrillation: SAFUCA® . Qual Life Res 26, 767–778 (2017). https://doi.org/10.1007/s11136-016-1474-4
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11136-016-1474-4