Abstract
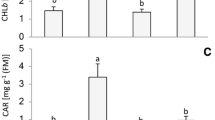
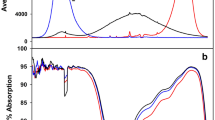
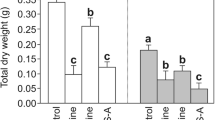
The antioxidant balance, photochemical activity of photosystem II (PSII), and photosynthetic pigment content, as well as the expression of genes involved in the light signalling of callus lines of Eutrema salsugineum plants (earlier Thellungiella salsuginea) under different spectral light compositions were studied. Growth of callus in red light (RL, maximum 660 nm), in contrast to blue light (BL, maximum 450 nm), resulted in a lower H2O2 content and thiobarbituric acid reactive substances (TBARS). The BL increased the activities of key antioxidant enzymes in comparison with the white light (WL) and RL and demonstrated the minimum level of PSII photochemical activity. The activities of catalase (CAT) and peroxidase (POD) had the highest values in BL, which, along with the increased H2O2 and TBARS content, indicate a higher level of oxidative stress in the cells. The expression levels of the main chloroplast protein genes of PSII (PSBA and PSBD), the NADPH-dependent oxidase gene of the plasma membrane (RbohD), the protochlorophyllide oxidoreductase genes (POR B, C) involved in the biosynthesis of chlorophyll, and the key photoreceptor signalling genes (CIB1, CRY2, PhyB, PhyA, and PIF3) were determined. Possible mechanisms of light quality effects on the physiological parameters of callus cells are discussed.





Similar content being viewed by others
Abbreviations
- APX:
-
Ascorbate peroxidase
- SOD:
-
Superoxide dismutase
- POD:
-
Peroxidase
- CAT:
-
Catalase
- TBARS:
-
Thiobarbituric acid reactive substances
- BL:
-
Blue light from light-emitting diodes 450 nm
- WL:
-
White light from light-emitting diodes 450 nm + 660 nm
- RL:
-
Red light from light-emitting diodes 660 nm
- WCFL:
-
Light from white compact fluorescent lamps
- CRY:
-
Cryptochrome
- PHY:
-
Phytochrome
- PSII:
-
Photosystem II
- ROS:
-
Reactive oxygen species
- ETC:
-
Electron transport chain
- Chla :
-
Chlorophyll a
- Chlb :
-
Chlorophyll b
- Car:
-
Carotenoids
References
Ahmad JV, Smirnova O, Cashmore AR (1998) The CRY1 blue light photoreceptor of Arabidopsis interacts with phytochrome A in vitro. Mol Cell 1:939–948. doi:10.1016/S1097-2765(00)80094-5
Allakhverdiev SI (2011) Recent progress in the studies of structure and function of photosystem II. J Photoch Photobio B 104: 1–8. doi:10.1016/j.jphotobiol.2011.03.010
Allakhverdiev SI, Murata N (2004). Environmental stress inhibits the synthesis de novo of proteins involved in the photodamage–repair cycle of photosystem II in Synechocystis sp. PCC 6803. Biochimica et Biophysica Acta (BBA)-Bioenergetics. 1657:23–32. doi:10.1016/j.bbabio.2004.03.003
Allakhverdiev SI, Klimov VV, Carpentier R (1997) Evidence for the involvement of cyclic electron transport in the protection of photosystem II against photoinhibition: influence of a new phenolic compound. Biochem 36:4149–4154. doi:10.1021/bi962170n
Allakhverdiev SI, Los DA, Mohanty P, Nishiyama Y, Murata N (2007) Glycinebetaine alleviates the inhibitory effect of moderate heat stress on the repair of photosystem II during photoinhibition. Biochimica et Biophysica Acta (BBA)-Bioenergetics 1767(12):1363–1371. doi:10.1016/j.bbabio.2007.10.005
Asada K (1999) The water–water cycle in chloroplasts: scavenging of active oxygens and dissipation of excess photons. Annu Rev Plant Physiol Plant Mol Biol 50:601–639. doi:10.1146/annurev.arplant.50.1.601
Bae G, Choi G (2008) Decoding of light signals by plant phytochromes and their interacting proteins. Annu Rev Plant Biol 59:281–311. doi:10.1146/annurev.arplant.59.032607.092859
Beauchamp C, Fridovich I (1971) Superoxide dismutase: improved assays and an assay applicable to acrylamide gels. Anal Biochem 44:276–287
Beel B, Prager K, Spexard M et al (2012) A flavin binding cryptochrome photoreceptor responds to both blue and red light in Chlamydomonas reinhardtii. Plant Cell 24:2992–3008. doi:10.1105/tpc.112.098947
Bliznikas Z, Zukauskas A, Samuoliene G, Viršile A, Brazaityte A, Jankauskiene J, Novičkovas A (2012) The effect of supplementary pre-harvest LED lighting on the antioxidant and nutritional properties of green vegetables. Acta Hortic 939:85–91
Casal JJ (2000) Phytochromes, cryptochromes, phototropin, photoreceptor interactions in plants. Photochem Photobiol 71:1–11
Chance B, Maehly AC (1955) Assay of catalases and peroxidases. Method Enzymol 2:764–775. doi:10.1016/S0076-6879(55)02300-8
Cluis CP, Mouchel CF, Hardtke CS (2004) The Arabidopsis transcription factor HY5 integrates light and hormone signaling pathways. Plant J 38:332–347. doi:10.1111/j.1365-313X.2004.02052.x
Consentino L, Lambert S, Martino C, Jourdan N, Bouchet PE, Witczak J, Ahmad M (2015) Blue-light dependent reactive oxygen species formation by Arabidopsis cryptochrome may define a novel evolutionarily conserved signaling mechanism. New Phytol 206:1450–1462. doi:10.1111/nph.13341
Cormier MJ, Prichard PM (1968) An investigation of the mechanism of the luminescent peroxidation of luminol by stopped flow techniques. J Biol Chem 243:4706–4714
Dai Y, Shen Z, Liu Y, Wang L, Hannaway D, Lu H (2009) Effects of shade treatments on the photosynthetic capacity, chlorophyll fluorescence, and chlorophyll content of Tetrastigma hemsleyanum Diels et Gilg. Environ Exp Bot 65:177–182. doi:10.1016/j.envexpbot.2008.12.008
Desikan R, Soheila AH, Hancock JT, Neill SJ (2001) Regulation of the Arabidopsis transcriptome by oxidative stress. Plant Physiol 127(1):159–172. doi:10.1104/pp.127.1.159
Foyer CH, Noctor G (2005) Oxidant and antioxidant signalling in plants: a re-evaluation of the concept of oxidative stress in a physiological context. Plant Cell Env 28:1056–1071. doi:10.1111/j.1365-3040.2005.01327.x
Franklin KA, Whitelam GC (2004) Light signals, phytochromes and crosstalk with other environmental cues. J Exp Bot 55:271–276. doi:10.1093/jxb/erh026
Fraser DP, Hayes S, Franklin KA (2016) Photoreceptor crosstalk in shade avoidance. Curr Opin Plant Biol 33:1–7. doi:10.1016/j.pbi.2016.03.008
Havaux M, Niyogi KK (1999) The violaxanthin cycle protects plants from photooxidative damage by more than one mechanism. Proc Natl Acad Sci USA 96(15):8762–8767. doi:10.1073/pnas.96.15.8762
Heath RL, Packer L (1968) Photoperoxidation in isolated chloroplasts: I. Kinetics and stoichiometry of fatty acid peroxidation. Arch Biochem Biophys 125:189–198. doi:10.1016/0003-9861(68)90654-1
Imaizumi T, Kadota A, Hasebe M, Wada M (2002) Cryptochrome light signals control development to suppress auxin sensitivity in the moss Physcomitrella patens. Plant Cell 14:373–386. doi:10.1105/tpc.010388
Jeong J, Kim K, Kim ME, Kim HG, Heo GS, Park OK, Oh E (2016) Phytochrome and ethylene signaling integration in arabidopsis occurs via the transcriptional regulation of genes co-targeted by PIFs and EIN3. Front Plant Sci 7:1055. doi:10.3389/fpls.2016.01055
Jin H, Li M, Duan S, Fu M, Dong X, Liu B, Wang HB (2016) Optimization of light harvesting pigment improves photosynthetic efficiency. Plant Physiol. doi:10.1104/pp.16.00698
Johkan M, Shoji K, Goto F, Hashida SN, Yoshihara T (2010) Blue light-emitting diode light irradiation of seedlings improves seedling quality and growth after transplanting in red leaf lettuce. HortSci 45:1809–1814
Jourdan N, Martino CF, El-Esawi M, Witczak J, Bouchet P-E, d’Harlingue A, Ahmad M (2015) Blue-light dependent ROS formation by Arabidopsis cryptochrome-2 may contribute toward its signaling role. Plant Signal Behav 10:e1042647. doi:10.1111/nph.13341
Kalaji HM, Schansker G, Ladle RJ, Goltsev V, Bosa K, Allakhverdiev SI, Elsheery NI (2014a) Photosynth Res 122:121–158. doi:10.1007/s11120-014-0024-6
Kalaji HM, Jajoo A, Oukarroum A, Brestic M, Zivcak M, Samborska I, Ahmad P (2014b) The use of chlorophyll fluorescence kinetics analysis to study the performance of photosynthetic machinery in plants. Emerg Technol Manag Crop Stress Toler 2:347–385. doi:10.1016/B978-0-12-800875-1.00009-0
Kendrick RE, Kronenberg GH (eds) (2012). Photomorphogenesis in plants. Springer, Dordrecht. doi:10.1007/978-94-011-1884-2
Kleine T, Kindgren P, Benedict C, Hendrickson L (2007) Strand A Genome-wide gene expression analysis reveals a critical role for CRYPTOCHROME1 in the response of Arabidopsis to high irradiance. Plant Physiol 127:1391–1406. doi:10.1104/pp.107.098293
Kosobryukhov AA, Lyubimov VYu, Kreslavski VD. (2015) Adaptive mechanisms of photosynthetic apparatus to UV radiation. In: Stress response in plants, Tripathi BN, Müller M (eds). Springer, Dordrecht, Chap. 3, pp 59–78. doi:10.1007/978-3-319-13368-3_3
Kreslavski VD, Carpentier R, Klimov VV, Allakhverdiev SI (2009) Transduction mechanisms of photoreceptor signals in plant cells. J Photochem Photobiol C 10:63–80. doi:10.1016/j.jphotochemrev.2009.04.001
Kreslavski VD, Shirshikova GN, Lyubimov VY, Shmarev AN, Boutanaev AM, Kosobryukhov AA, Allakhverdiev SI (2013a) Effect of preillumination with red light on photosynthetic parameters and oxidant-/antioxidant balance in Arabidopsis thaliana in response to UV-A. J Photochem Photobiol B 127:229–236. doi:10.1016/j.jphotobiol.2013.08.008
Kreslavski VD, Lyubimov VY, Shirshikova GN, Shmarev AN, Kosobryukhov AA, Schmitt FJ, Allakhverdiev SI (2013b) Preillumination of lettuce seedlings with red light enhances the resistance of photosynthetic apparatus to UV-A. J Photochem Photobiol B 122:1–6. doi:10.1016/j.jphotobiol.2013.02.016
Lankin AV, Kreslavski VD, Khudyakova AY, Zharmukhamedov SK, Allakhverdiev SI (2014) Effect of naphthalene on photosystem 2 photochemical activity of pea plants. Biochem Moscow 79:1216–1225. doi:10.1134/S0006297914110091
Lau OS, Deng XW (2012) The photomorphogenic repressors COP1 and DET1: 20 years later. Trends Plant Sci 17:584–593. doi:10.1016/j.tplants.2012.05.004
Lefsrud MG, Kopsell DA, Sams CE (2008) Irradiance from distinct wavelength light-emitting diodes affect secondary metabolites in kale. HortSci 43:2243–2244
Li Q, Kubota C (2009) Effects of supplemental light quality on growth and phytochemicals of baby leaf lettuce. Env Exp Bot 67:59–64. doi:10.1016/j.envexpbot.2009.06.011
Lichtenthaler HK (1987) Chlorophylls and carotenoids: pigments of photosynthetic biomembranes. Method Enzymol 148:350–382. doi:10.1016/0076-6879(87)48036-1
Liu H, Yu X, Li K, Klejnot J, Yang H, Lisiero D, Lin C (2008) Photoexcited CRY2 interacts with CIB1 to regulate transcription and floral initiation in Arabidopsis. Science 322:1535–1539. doi:10.1126/science.1163927
Liu H, Liu B, Zhao C, Pepper M, Lin C (2011) The action mechanisms of plant cryptochromes. Trends Plant Sci 16:684–691. doi:10.1016/j.tplants.2011.09.002
Massa GD, Kim HH, Wheeler RM, Mitchell CA (2008) Plant productivity in response to LED lighting. HortSci 43:1951–1956
Meinhard M, Grill E (2001) Hydrogen peroxide is a regulator of ABI1, a protein phosphatase 2C from Arabidopsis. Febs Lett 508:443–446. doi:10.1016/S0014-5793(01)03106-4
Miyake С (2010) Alternative electron flows (water–water cycle and cyclic electron flow around psi) in photosynthesis: molecular mechanisms and physiological functions. Plant Cell Physiol 51:1951–1963. 10.1093/pcp/pcq173
Mohanty P, Allakhverdiev SI, Murata N (2007) Application of low temperatures during photoinhibition allows characterization of individual steps in photodamage and the repair of photosystem II. Photosynth Res 94:217–224. 10.1007/s11120-007-9184-y
Nakano Y, Asada K (1981) Hydrogen peroxide is scavenged by ascorbate-specific peroxidase in spinach chloroplasts. Plant Cell Physiol 22:867–880. doi:10.1093/oxfordjournals.pcp.a076232
Neill S, Desikan R, Hancock J (2002) Hydrogen peroxide signalling. Curr Opin Plant Biol 5:388–395. 10.1016/S1369-5266(02)00282-0
Nishiyama Y, Allakhverdiev SI, Murata N (2006) A new paradigm for the action of reactive oxygen species in the photoinhibition of photosystem II. Biochim Biophys Acta. 10.1016/j.bbabio.2006.05.013
Ohnishi N, Allakhverdiev SI, Takahashi S, Higashi S, Watanabe M, Nishiyama Y, Murata N (2005) Two-step mechanism of photodamage to photosystem II: step 1 occurs at the oxygen-evolving complex and step 2 occurs at the photochemical reaction center. BioChemistry 44(23):8494–8499. doi:10.1021/bi047518q
Pathak V, Prasad A, Pospısil P.(2017). Formation of singlet oxygen by decomposition of protein hydroperoxide in photosystem II. PLoS One 12(7):e0181732. doi:10.1371/journal.pone.0181732
Penuelas J, Baret F, Filella I (1995) Semi-empirical indices to assess carotenoids/chlorophyll a ratio from leaf spectral reflectance. Photosynthetica 31(2):221–230
Powles SB (1984) Photoinhibition of photosynthesis induced with visible light. Annu Rev Plant Physiol 35:15–44. doi:10.1146/annurev.pp.35.060184.000311
Radyukina NL, Ivanov YV, Kartashov AV, Shevyakova NI, Rakitin VY, Khryanin VN, Kuznetsov VV (2007) Inducible and constitutive mechanisms of salt stress resistance in Geum urbanum L. Russ J Plant Physiol 54:612–618. doi:10.1134/S102144370705007X
Radyukina NL, Mapelli S, Ivanov YV, Kartashov AV, Brambilla I, Kuznetsov VV (2009) Homeostasis of polyamines and antioxidant systems in roots and leaves of Plantago major under salt stress. Russ J Plant Physiol 56(3):323–331
Radyukina NL, Ivanov YV, Kartashov AV, Pashkovskiy PP, Shevyakova NI, Kuznetsov VV (2011) Regulation of gene expression governing proline metabolism in Thellungiella salsuginea by NaCl and paraquat. Russ J Plant Physiol 58:643–652. doi:10.1134/S102144371104011X
Ridge I, Osborne DJ (1971) Role of peroxidase when hydroxyproline-rich protein in plant cell walls is increased by ethylene. Nature 229:205–208. 10.1038/newbio229205a0 doi
Sagi M, Fluhr R (2006) Production of reactive oxygen species by plant NADPH oxidases. Plant Physiol 141:336–340. doi:10.1104/pp.106.078089
Samuolienė G, Brazaitytė A, Sirtautas R, Novičkovas A, Duchovskis P (2011) Supplementary red-LED lighting affects phytochemicals and nitrate of baby leaf lettuce. JFAE 9:271–274
Samuolienė G, Sirtautas R, Brazaitytė A, Duchovskis P (2012) LED lighting and seasonality effects antioxidant properties of baby leaf lettuce. Food Chem 134:1494–1499. doi:10.1016/j.foodchem.2012.03.061
Sarvikas P, Hakala M, Patsikka E, Tyystjarvi T, Tyystjarvi E (2006) Action spectrum of photoinhibition in leaves of wild type and npq1-2 and npq4-1 mutants of Arabidopsis thaliana. Plant Cell Physiol 47:391–400. doi:10.1093/pcp/pcj006
Schenk RU, Hildebrandt AC (1972) Medium and techniques for induction and growth of monocotyledonous and dicotyledonous plant cell cultures. Can J Bot 50:199–204. doi:10.1139/b72-026
Schmitt FJ, Renger G, Friedrich T, Kreslavski VD, Zharmukhamedov SK, Los DA, Kuznetsov VV, Allakhverdiev SI. 2014. Reactive oxygen species: reevaluation of generation, monitoring and role in stress-signalling in phototrophic organisms. Biochim Biophys Acta 1837:835–848. doi:10.1016/j.bbabio.2014.02.005
Sellaro R, Crepy M, Trupkin SA, Karayekov E, Buchovsky AS, Rossi C, Casal JJ (2010) Cryptochrome as a sensor of the blue/green ratio of natural radiation in Arabidopsis. Plant Physiol 154:401–409. doi:10.1104/pp.110.160820
Soshinkova TN, Radyukina NL, Korolkova DV, Nosov AV (2013) Proline and functioning of the antioxidant system in Thellungiella salsuginea plants and cultured cells subjected to oxidative stress. Russ J Plant Physiol 60:41–54. doi:10.1134/S1021443713010093
Strasser RJ, Tsimilli-Michael M, Qiang S, Goltsev V (2010) Simultaneous in vivo recording of prompt and delayed fluorescence and 820-nm reflection changes during drying and after rehydration of the resurrection plant Haberlea rhodopensis. Biochim Biophys Acta 1797:1313–1326. doi:10.1016/j.bbabio.2010.03.008
Stutte GW (2009) Light-emitting diodes for manipulating the phytochrome apparatus. HortSci 44:231–234
Tefler A (2002) What is b-carotene doing in the photosystem II reaction centre? Phil Trans R Soc Lond B 357:1431–1440
Wen F, Xing D, Zhang L (2008) Hydrogen peroxide is involved in high blue light-induced chloroplast avoidance movements in Arabidopsis. J Exp Bot 59:2891–2901. doi:10.1093/jxb/ern147
Wu HJ, Zhang Z, Wang JY, Oh DH, Dassanayake M, Liu B, Huang Q, Sun HX, Xia R, Wu Y, Wang YN (2012) Insights into salt tolerance from the genome of Thellungiella salsuginea. Proc Natl Acad Sci USA 109:12219–12224. doi:10.1073/pnas.1209954109
Yamamoto YY, Matsui M, Ang LH, Deng XW (1998) Role of a COP1 interactive protein in mediating light-regulated gene expression in Arabidopsis. Plant Cell 10:1083–1094. doi:10.1105/tpc.10.7.1083
Yurina NP, Mokerova DV, Odintsova MS (2013) Light-inducible stress plastid proteins of phototrophs. Russ J Plant Physiol 60:577–588. doi:10.1134/S1021443713050154
Zhao X, Tan HJ, Liu YB, Li XR, Chen GX (2009) Effect of salt stress on growth and osmotic regulation in Thellungiella and Arabidopsis callus. PCTOC 98:97–103. doi:10.1007/s11240-009-9542-x
Acknowledgements
This work was supported by the Russian Foundation for Basic Research (RFBR) on 15-04-01199a and by the Program of the Presidium of the Russian Academy of Sciences “Molecular and Cellular Biology” (MCB RAS).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Pashkovskiy, P.P., Soshinkova, T.N., Korolkova, D.V. et al. The effect of light quality on the pro-/antioxidant balance, activity of photosystem II, and expression of light-dependent genes in Eutrema salsugineum callus cells. Photosynth Res 136, 199–214 (2018). https://doi.org/10.1007/s11120-017-0459-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-017-0459-7