Abstract
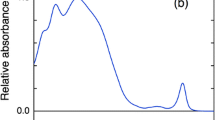
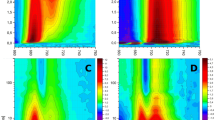
The major light-harvesting complex of Amphidinium (A.) carterae, chlorophyll-a–chlorophyll-c 2–peridinin–protein complex (acpPC), was studied using ultrafast pump-probe spectroscopy at low temperature (60 K). An efficient peridinin–chlorophyll-a energy transfer was observed. The stimulated emission signal monitored in the near-infrared spectral region was stronger when redder part of peridinin pool was excited, indicating that these peridinins have the S1/ICT (intramolecular charge-transfer) state with significant charge-transfer character. This may lead to enhanced energy transfer efficiency from “red” peridinins to chlorophyll-a. Contrary to the water-soluble antenna of A. carterae, peridinin–chlorophyll-a protein, the energy transfer rates in acpPC were slower under low-temperature conditions. This fact underscores the influence of the protein environment on the excited-state dynamics of pigments and/or the specificity of organization of the two pigment–protein complexes.





Similar content being viewed by others
References
Bautista JA, Hiller RG, Sharples FP, Gosztola D, Wasielewski MR, Frank HA (1999) Singlet and triplet energy transfer in the peridinin–chlorophyll a–protein from Amphidinium carterae. J Phys Chem A 103:2267–2273. doi:10.1021/jp983943f
Billsten HH, Zigmantas D, Sundström V, Polívka T (2002) Dynamics of vibrational relaxation in the S1 state of carotenoids having 11 conjugated C=C bonds. Chem Phys Lett 355:465–470
Boldt L, Yellowlees D, Leggat W (2012) Hyperdiversity of genes encoding integral light-harvesting proteins in the dinoflagellate Symbiodinium sp. PLoS One 7:e47456. doi:10.1371/journal.pone.0047456
Büchel C (2003) Fucoxanthin–chlorophyll–proteins in diatoms: 18 and 19 kDa subunits assemble into different oligomeric states. Biochemistry 42:13027–13034. doi:10.1021/bi0349468
Di Valentin M, Salvadori E, Agostini G, Biasibetti F, Ceola S, Hiller R, Giacometti GM, Carbonera D (2010) Triplet-triplet energy transfer in the major intrinsic light-harvesting complex of Amphidinium carterae as revealed by ODMR and EPR spectroscopies. Biochim Biophys Acta 1797:1759–1767. doi:10.1016/j.bbabio.2010.06.011
Di Valentin M, Büchel C, Giacometti GM, Carbonera D (2012) Chlorophyll triplet quenching by fucoxanthin in the fucoxanthin–chlorophyll–protein from the diatom Cyclotella meneghiniana. Biochem Biophys Res Commun 427:637–641. doi:10.1016/j.bbrc.2012.09.113
Durchan M, Fuciman M, Šlouf V, Keşan G, Polívka T (2012) Excited-state dynamics of monomeric and aggregated carotenoid 8′-apo-β-carotenal. J Phys Chem A 116:12330–12338. doi:10.1021/jp310140k
Falkowski PG, Katz ME, Knoll AH, Quigg A, Raven JA, Schofield O, Taylor FJR (2004) The evolution of modern eukaryotic phytoplankton. Science 305:354–360. doi:10.1126/science.1095964
Field CB, Behrenfeld MJ, Randerson JT, Falkowski P (1998) Primary production of the biosphere: integrating terrestrial and oceanic components. Science 281:237–240. doi:10.1126/science.281.5374.237
Frank HA, Bautista JA, Josue J, Pendon Z, Hiller RG, Sharples FP, Gosztola D, Wasielewski MR (2000) Effect of the solvent environment on the spectroscopic properties and dynamics of the lowest excited states of carotenoids. J Phys Chem B 104:4569–4577. doi:10.1021/jp000079u
Gildenhoff N, Amarie S, Gundermann K, Beer A, Büchel C, Wachtveitl J (2010a) Oligomerization and pigmentation dependent excitation energy transfer in fucoxanthin–chlorophyll–proteins. Biochim Biophys Acta 1797:543–549. doi:10.1016/j.bbabio.2010.01.024
Gildenhoff N, Herz J, Gundermann K, Büchel C, Wachtveitl J (2010b) The excitation energy transfer in the trimeric fucoxanthin–chlorophyll–protein from Cyclotella meneghiniana analyzed by polarized transient absorption spectroscopy. Chem Phys 373:104–109. doi:10.1016/j.chemphys.2010.02.012
Gradinaru CC, van Stokkum IHM, Pascal AA, van Grondelle R, van Amerongen H (2000) Identifying the pathways of energy transfer between carotenoids and chlorophylls in LHCII and CP29. A multicolor, femtosecond pump-probe study. J Phys Chem B 104:9330–9342. doi:10.1021/jp001752i
Hiller RG, Wrench PM, Gooley AP, Shoebridge G, Breton J (1993) The major intrinsic light-harvesting protein of Amphidinium-characterization and relation to other light-harvesting proteins. Photochem Photobiol 57:125–131. doi:10.1111/j.1751-1097.1993.tb02267.x
Hofmann E, Wrench PM, Sharples FP, Hiller RG, Welte W, Diederichs K (1996) Structural basis of light harvesting by carotenoids: peridinin–chlorophyll–protein from Amphidinium carterae. Science 272:1788–1791. doi:10.1126/science.272.5269.1788
Ilagan RP, Christensen RL, Chapp TW, Gibson GN, Pascher T, Polívka T, Frank HA (2005) Femtosecond time-resolved absorption spectroscopy of astaxanthin in solution and in α-crustacyanin. J Phys Chem A 109:3120–3127. doi:10.1021/jp0444161
Ilagan RP, Koscielecki JF, Hiller RG, Sharples FP, Gibson GN, Birge RR, Frank HA (2006) Femtosecond time-resolved absorption spectroscopy of main-form and high-salt peridinin–chlorophyll a–proteins at low temperatures. Biochemistry 45:14052–14063. doi:10.1021/bi061217u
Jiang J, Zhang H, Kang Y, Bina D, Lo CS, Blankenship RE (2012) Characterization of the peridinin–chlorophyll a–protein complex in the dinoflagellate Symbiodinium. Biochim Biophys Acta 1817:983–989. doi:10.1016/j.bbabio.2012.03.027
Kosumi D et al (2011) Ultrafast excited state dynamics of fucoxanthin: excitation energy dependent intramolecular charge transfer dynamics. Phys Chem Chem Phys 13:10762–10770. doi:10.1039/c0cp02568b
Lee JJ, Olea R, Cevasco M, Pochon X, Correia M, Shpigel M, Pawlowski J (2003) A marine dinoflagellate, Amphidinium eilatiensis n. sp., from the benthos of a mariculture sedimentation pond in Eilat, Israel. J Eukaryot Microbiol 50:439–448. doi:10.1111/j.1550-7408.2003.tb00270.x
Macpherson AN, Hiller RG (2003) Light-harvesting systems in chlorophyll c-containing algae. In: Green BR, Parson WW (eds) Light-harvesting antennas in photosynthesis. Kluwer Academic Publishers, Dordrecht, pp 323–352
Miloslavina Y, Grouneva I, Lambrev PH, Lepetit B, Goss R, Wilhelm C, Holzwarth AR (2009) Ultrafast fluorescence study on the location and mechanism of non-photochemical quenching in diatoms. Biochim Biophys Acta 1787:1189–1197. doi:10.1016/j.bbabio.2009.05.012
Niedzwiedzki DM, Enriquez MM, Lafountain AM, Frank HA (2010) Ultrafast time-resolved absorption spectroscopy of geometric isomers of xanthophylls. Chem Phys 373:80–89. doi:10.1016/j.chemphys.2010.01.019
Niedzwiedzki DM, Jiang J, Lo CS, Blankenship RE (2013) Spectroscopic properties of the chlorophyll a–chlorophyll c 2–peridinin–protein-complex (acpPC) from the coral symbiotic dinoflagellate Symbiodinium. Photosynth Res. doi:10.1007/s11120-013-9794-5
Norris BJ, Miller DJ (1994) Nucleotide-sequence of a cDNA clone encoding the precursor of the peridinin–chlorophyll a-binding protein from the dinoflagellate Symbiodinium sp. Plant Mol Biol 24:673–677. doi:10.1007/BF00023563
Papagiannakis E, van Stokkum IHM, Fey H, Büchel C, van Grondelle R (2005) Spectroscopic characterization of the excitation energy transfer in the fucoxanthin–chlorophyll–protein of diatoms. Photosynth Res 86:241–250. doi:10.1007/s11120-005-1003-8
Papagiannakis E, Vengris M, Larsen DS, van Stokkum IHM, Hiller RG, van Grondelle R (2006) Use of ultrafast dispersed pump-dump-probe and pump-repump-probe spectroscopies to explore the light-induced dynamics of peridinin in solution. J Phys Chem B 110:512–521. doi:10.1021/jp053094d
Polívka T, Frank HA (2010) Molecular factors controlling photosynthetic light harvesting by carotenoids. Acc Chem Res 43:1125–1134. doi:10.1021/ar100030m
Polívka T, Pascher T, Sundström V, Hiller RG (2005) Photosynth Res 86:217–227. doi:10.1007/s11120-005-1447-x
Polívka T, van Stokkum IHM, Zigmantas D, van Grondelle R, Sundström V, Hiller RG (2006) Energy transfer in the major intrinsic light-harvesting complex from Amphidinium carterae. Biochemistry 45:8516–8526. doi:10.1021/bi060265b
Polívka T, Hiller RG, Frank HA (2007) Spectroscopy of the peridinin–chlorophyll-a–protein: insight into light-harvesting strategy of marine algae. Arch Biochem Biophys 458:111–120. doi:10.1016/j.abb.2006.10.006
Premvardhan L, Robert B, Beer A, Büchel C (2010) Pigment organization in fucoxanthin–chlorophyll a/c 2–proteins (FCP) based on resonance Raman spectroscopy and sequence analysis. Biochim Biophys Acta 1797:1647–1656. doi:10.1016/j.bbabio.2010.05.002
Schulte T, Niedzwiedzki DM, Birge RR, Hiller RG, Polívka T, Hofmann E, Frank HA (2009) Identification of a single peridinin sensing Chl-a excitation in reconstituted PCP by crystallography and spectroscopy. Proc Natl Acad Sci USA 106:20764–20769. doi:10.1073/pnas.0908938106
Stomp M, Huisman J, Stal LJ, Matthijs HCP (2007) Colorful niches of phototrophic microorganisms shaped by vibrations of the water molecule. ISME J 1:271–282. doi:10.1038/ismej.2007.59
Szabó M, Premvardhan L, Lepetit B, Goss R, Wilhelm C, Garab G (2010) Functional heterogeneity of the fucoxanthins and fucoxanthin–chlorophyll–proteins in diatom cells revealed by their electrochromic response and fluorescence and linear dichroism spectra. Chem Phys 373:110–114. doi:10.1016/j.chemphys.2010.03.001
Trautman JK, Shreve AP, Owens TG, Albrecht AC (1990) Femtosecond dynamics of carotenoid-to-chlorophyll energy transfer in thylakoid membrane preparations from Phaeodactylum tricornutum and Nannochloropsis sp. Chem Phys Lett 166:369–374. doi:10.1016/0009-2614(90)85045-E
van Amerongen H, van Grondelle R (2001) Understanding the energy transfer function of LHCII, the major light-harvesting complex of green plants. J Phys Chem B 105:604–617. doi:10.1021/jp0028406
Wagner NL, Greco JA, Enriquez MM, Frank HA, Birge RR (2013) The nature of the intramolecular charge transfer state in peridinin. Biophys J 104:1314–1325. doi:10.1016/j.bpj.2013.01.045
Zigmantas D, Hiller RG, Sundström V, Polívka T (2002) Carotenoid to chlorophyll energy transfer in the peridinin–chlorophyll-a–protein complex involves an intramolecular charge transfer state. Proc Natl Acad Sci USA 99:16760–16765. doi:10.1073/pnas.262537599
Zigmantas D, Hiller RG, Yartsev A, Sundström V, Polívka T (2003) Dynamics of excited states of the carotenoid peridinin in polar solvents: dependence on excitation wavelength, viscosity, and temperature. J Phys Chem B 107:5339–5348. doi:10.1021/jp0272318
Zigmantas D, Hiller RG, Sharples FP, Frank HA, Sundström V, Polívka T (2004) Effect of a conjugated carbonyl group on the photophysical properties of carotenoids. Phys Chem Chem Phys 6:3009–3016. doi:10.1039/b315786e
Acknowledgments
The authors thank Miriam Enriquez for help with measuring the absorption spectra at 77 K. The work in Czech Republic was supported by the Czech Science Foundation (P501/12/G055 and P205/11/1164), and project KONTAKT ME09037 from the Ministry of Education of the Czech Republic. Work in the laboratory of HAF was supported by grants from the National Science Foundation (MCB-1243565) and the University of Connecticut Research Foundation.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Šlouf, V., Fuciman, M., Johanning, S. et al. Low-temperature time-resolved spectroscopic study of the major light-harvesting complex of Amphidinium carterae . Photosynth Res 117, 257–265 (2013). https://doi.org/10.1007/s11120-013-9900-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11120-013-9900-8