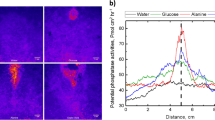
Abstract
Aims
Biological soil crusts (biocrusts) are microbial communities commonly found in the upper layer of arid soils. These microorganisms release exopolysaccharides (EPS), which form the exopolymeric matrix (EPM), allowing them to bond soil particles together and survive long periods of dryness. The aim of this work is to develop methods for measuring metabolite retention by biocrust EPM and EPS.
Methods
We report new methods for the investigation of metabolite sorption on biocrusts compared to the underlying unconsolidated subcrust fraction. A 13C–labeled bacterial lysate metabolite mixture was incubated with biocrust, subcrust and biocrust-extracted EPS. Non-sorbed metabolites were extracted and analyzed by liquid chromatography/mass spectrometry.
Results
This simple and rapid approach enabled the comparison of metabolite sorption on the biocrust EPM or EPS versus mineral sorption on the underlying soils. Our results suggest that the biocrust (and its extracted EPS) sorb more metabolites, especially amino acids and organic acids, than the underlying subcrust.
Conclusions
This study demonstrates a useful method to highlight the essential role of biocrust (especially the EPM), which acts as a passive nutrient filter, sequestering metabolites released by microbes during wetting events. This may facilitate recovery of the community upon wetting and further enhance biocrust survival and nutrient retention.




Similar content being viewed by others
Abbreviations
- EPM:
-
exopolymeric matrix
- EPS:
-
exopolysaccharides
- LC/MS:
-
liquid chromatograph-mass spectrometry
- XRPD:
-
X-ray powder diffraction
References
Al-Thani RF (2015) Cyanomatrix and cyanofilm. J Res Dev 3:10000123. https://doi.org/10.4172/jrd.1000123
Baran R, Brodie EL, Mayberry-Lewis J et al (2015) Exometabolite niche partitioning among sympatric soil bacteria. Nat Commun 6:1–9. https://doi.org/10.1038/ncomms9289
Belnap J (2006) The potential roles of biological soil crusts in dryland hydrologic cycles. Hydrol Process 20:3159–3178. https://doi.org/10.1002/hyp.6325
Belnap J, Gardner JS (1993) Soil microstructure in soils of the Colorado plateau: the role of the cyanobacterium microcoleus vaginatus. Great Basin Nat 53:40–47. https://doi.org/10.2307/41712756?ref=search-gateway:2ec8b0369dbbaffac44bb84c3f340d96
Belnap J, Phillips SL, Miller ME (2004) Response of desert biological soil crusts to alterations in precipitation frequency. Oecologia 141:306–316. https://doi.org/10.1007/s00442-003-1438-6
Beraldi-Campesi H, Hartnett HE, Anbar A et al (2009) Effect of biological soil crusts on soil elemental concentrations: implications for biogeochemistry and as traceable biosignatures of ancient life on land. Geobiology 7:348–359. https://doi.org/10.1111/j.1472-4669.2009.00204.x
Bowker MA, Belnap J, Bala Chaudhary V, Johnson NC (2008) Revisiting classic water erosion models in drylands: the strong impact of biological soil crusts. Soil Biol Biochem 40:2309–2316. https://doi.org/10.1016/j.soilbio.2008.05.008
Braissant O, Decho AW, Dupraz C et al (2007) Exopolymeric substances of sulfate-reducing bacteria: interactions with calcium at alkaline pH and implication for formation of carbonate minerals. Geobiology 5:401–411. https://doi.org/10.1111/j.1472-4669.2007.00117.x
Brenowitz S, Castenholz RW (1997) Long-term effects of UV and visible irradiance on natural populations of a scytonemin-containing cyanobacterium (Calothrix sp.) FEMS Microbiol Ecol 24:343–352
Chen L, Rossi F, Deng S et al (2014) Macromolecular and chemical features of the excreted extracellular polysaccharides in induced biological soil crusts of different ages. Soil Biol Biochem 78:1–9. https://doi.org/10.1016/j.soilbio.2014.07.004
Cleaves HJ II, Crapster-Pregont E, Jonsson CM et al (2011) The adsorption of short single-stranded DNA oligomers to mineral surfaces. Chemosphere 83:1560–1567. https://doi.org/10.1016/j.chemosphere.2011.01.023
Colica G, Li H, Rossi F et al (2014) Microbial secreted exopolysaccharides affect the hydrological behavior of induced biological soil crusts in desert sandy soils. Soil Biol Biochem 68:62–70. https://doi.org/10.1016/j.soilbio.2013.09.017
Couradeau E, Karaoz U, Lim HC et al (2016) Bacteria increase arid-land soil surface temperature through the production of sunscreens. Nat Commun 7:1–7. https://doi.org/10.1038/ncomms10373
De Philippis R, Colica G, Micheletti E (2011) Exopolysaccharide-producing cyanobacteria in heavy metal removal from water: molecular basis and practical applicability of the biosorption process. Appl Microbiol Biotechnol 92:697–708. https://doi.org/10.1007/s00253-011-3601-z
De Philippis R, Vincenzini M (1998) Exocellular polysaccharides from cyanobacteria and their possible applications. FEMS Microbiol Rev 22:151–175. https://doi.org/10.1016/S0168-6445(98)00012-6
Elbert W, Weber B, Burrows S et al (2012) Contribution of cryptogamic covers to the global cycles of carbon and nitrogen. Nat Geosci 5:459–462. https://doi.org/10.1038/ngeo1486
Feuillie C, Daniel I, Michot LJ, Pedreira-Segade U (2013) Adsorption of nucleotides onto Fe–Mg–Al rich swelling clays. Geochim Cosmochim Acta 120:97–108. https://doi.org/10.1016/j.gca.2013.06.021
Fischer T, Veste M, Wiehe W, Lange P (2010) Water repellency and pore clogging at early successional stages of microbiotic crusts on inland dunes, Brandenburg, NE Germany. Catena 80:47–52. https://doi.org/10.1016/j.catena.2009.08.009
Flemming H-C, Wingender J (2010) The biofilm matrix. Nat Rev Microbiol 8:623–633. https://doi.org/10.1038/nrmicro2415
Garcia-Pichel F (2003) Desert environments: biological soil crusts. In: Bitton G (ed) Encyclopedia of environmental microbiology, vol 6. Wiley-Interscience, New York
Garcia-Pichel F, Belnap J (1996) Microenvironments and microscale productivity of cyanobacterial desert crusts. J Phycol 32:774–782
Garcia-Pichel F, Castenholz RW (1991) Characterization and biological implications of scytonemin, a cyanobacterial sheath pigment. J Phycol 27:395–409. https://doi.org/10.1111/j.0022-3646.1991.00395.x
Garcia-Pichel F, Johnson SL, Youngkin D, Belnap J (2003) Small-scale vertical distribution of bacterial biomass and diversity in biological soil crusts from arid lands in the Colorado plateau. Microb Ecol 46:312–321. https://doi.org/10.1007/s00248-003-1004-0
Garcia-Pichel F, Pringault O (2001) Cyanobacteria track water in desert soils. Nature 413:380–381. https://doi.org/10.1038/35096640
Garcia-Pichel F, Wojciechowski MF (2009) The evolution of a capacity to build supra-cellular ropes enabled filamentous cyanobacteria to colonize highly erodible substrates. PLoS One 4:e7801–e7806. https://doi.org/10.1371/journal.pone.0007801
Gundlapally SR, Garcia-Pichel F (2006) The community and phylogenetic diversity of biological soil crusts in the Colorado plateau studied by molecular fingerprinting and intensive cultivation. Microb Ecol 52:345–357. https://doi.org/10.1007/s00248-006-9011-6
Johnson SL, Kuske CR, Carney TD et al (2012) Increased temperature and altered summer precipitation have differential effects on biological soil crusts in a dryland ecosystem. Glob Chang Biol 18:2583–2593. https://doi.org/10.1111/j.1365-2486.2012.02709.x
Kleber M, Sollins P, Sutton R (2007) A conceptual model of organo-mineral interactions in soils: self-assembly of organic molecular fragments into zonal structures on mineral surfaces. Biogeochemistry 85:9–24. https://doi.org/10.1007/s10533-007-9103-5
Lutterotti L, Matthies S, Wenk HR (1999) MAUD: a friendly Java program for material analysis using diffraction. IUCr Newsletter of the CPD 21:14–15
Maestre FT, Escolar C, de Guevara ML et al (2013) Changes in biocrust cover drive carbon cycle responses to climate change in drylands. Glob Chang Biol 19:3835–3847. https://doi.org/10.1111/gcb.12306
Mager DM, Thomas AD (2011) Extracellular polysaccharides from cyanobacterial soil crusts: a review of their role in dryland soil processes. J Arid Environ 75:91–97. https://doi.org/10.1016/j.jaridenv.2010.10.001
Morbach S, Krämer R (2002) Body shaping under water stress: osmosensing and osmoregulation of solute transport in bacteria. Chembiochem 3:384–397. https://doi.org/10.1002/1439-7633(20020503)3:5<384::AID-CBIC384>3.0.CO;2-H
Mugnai G, Rossi F, Felde VJMNL et al (2017) Development of the polysaccharidic matrix in biocrusts induced by a cyanobacterium inoculated in sand microcosms. Biol Fertil Soils. https://doi.org/10.1007/s00374-017-1234-9
Murik O, Oren N, Shotland Y et al (2016) What distinguishes cyanobacteria able to revive after desiccation from those that cannot: the genome aspect. Environ Microbiol 19:535–550. https://doi.org/10.1111/1462-2920.13486
Pereira S, Zille A, Micheletti E et al (2009) Complexity of cyanobacterial exopolysaccharides: composition, structures, inducing factors and putative genes involved in their biosynthesis and assembly. FEMS Microbiol Rev 33:917–941. https://doi.org/10.1111/j.1574-6976.2009.00183.x
Rajeev L, da Rocha UN, Klitgord N et al (2013) Dynamic cyanobacterial response to hydration and dehydration in a desert biological soil crust. ISME J 7:2178–2191. https://doi.org/10.1038/ismej.2013.83
Reed SC, Maestre FT, Ochoa-Hueso R et al (2016) Biocrusts in the context of global climate change. In: Weber B, Büdel B, Belnap J (eds) Biological soil crusts: an organizing principle in drylands. Springer International Publishing, Switzerland, pp 451–476
Reynolds R, Belnap J, Reheis M et al (2001) Aeolian dust in Colorado plateau soils: nutrient inputs and recent change in source. Proc Natl Acad Sci U S A 98:7123–7127. https://doi.org/10.1073/pnas.121094298
Rossi F, De Philippis R (2015) Role of cyanobacterial exopolysaccharides in phototrophic biofilms and in complex microbial mats. Life 5:1218–1238. https://doi.org/10.3390/life5021218
Rossi F, Mugnai G, De Philippis R (2017) Complex role of the polymeric matrix in biological soil crusts. Plant Soil. https://doi.org/10.1007/s11104-017-3441-4
Rossi F, Potrafka RM, Pichel FG, De Philippis R (2012) The role of the exopolysaccharides in enhancing hydraulic conductivity of biological soil crusts. Soil Biol Biochem 46:33–40. https://doi.org/10.1016/j.soilbio.2011.10.016
Sasse J, Martinoia E, Northen T (2017) Feed your friends: do plant exudates shape the root microbiome? Trends Plant Sci:1–17. https://doi.org/10.1016/j.tplants.2017.09.003
Schimel J, Balser TC, Wallenstein M (2007) Microbial stress-response physiology and its implications for ecosystem function. Ecology 88:1386–1394. https://doi.org/10.1890/06-0219
Seviour RJ, Stasinopoulos SJ, Auer DPF, Gibbs PA (1992) Production of pullulan and other exopolysaccharides by filamentous fungi. Crit Rev Biotechnol 12:279–298. https://doi.org/10.3109/07388559209069196
Steven B, Gallegos-Graves LV, Belnap J, Kuske CR (2013) Dryland soil microbial communities display spatial biogeographic patterns associated with soil depth and soil parent material. FEMS Microbiol Ecol 86:101–113. https://doi.org/10.1111/1574-6941.12143
Stuart RK, Mayali X, Lee JZ et al (2016) Cyanobacterial reuse of extracellular organic carbon in microbial mats. ISME J 10:1240–1251. https://doi.org/10.1038/ismej.2015.180
Sun WQ, Leopold AC (1997) Cytoplasmic vitrification and survival of anhydrobiotic organisms. Comp Biochem Physiol 117:327–333. https://doi.org/10.1016/S0300-9629(96)00271-X
Swenson TL, Bowen BP, Nico PS, Northen TR (2015a) Competitive sorption of microbial metabolites on an iron oxide mineral. Soil Biol Biochem 90:34–41. https://doi.org/10.1016/j.soilbio.2015.07.022
Swenson TL, Jenkins S, Bowen BP, Northen TR (2015b) Untargeted soil metabolomics methods for analysis of extractable organic matter. Soil Biol Biochem 80:189–198. https://doi.org/10.1016/j.soilbio.2014.10.007
Wolfaardt GM, Lawrence JR, Korber DR (1999) Function of EPS. In: Wingender DJ, Neu D, Flemming P-C (eds) Microbial extracellular polymeric Substances. Springer, Berlin, pp 171–200
Yao Y, Sun T, Wang T et al (2015) Analysis of metabolomics datasets with high-performance computing and metabolite atlases. Meta 5:431–442. https://doi.org/10.3390/metabo5030431
Snyder RL (1993) Analytical profile fitting of X-ray powder diffraction profiles in Rietveld analysis. In: Young RA (ed) The Rietveld method. Oxford University Press, Oxford
Yu J, Kidron GJ, Pen-Mouratov S et al (2012) Do development stages of biological soil crusts determine activity and functional diversity in a sand-dune ecosystem? Soil Biol Biochem 51:66–72. https://doi.org/10.1016/j.soilbio.2012.04.007
Zheng Y, Xu M, Zhao J et al (2010) Effects of inoculated Microcoleus vaginatus on the structure and function of biological soil crusts of desert. Biol Fertil Soils 47:473–480. https://doi.org/10.1007/s00374-010-0521-5
Ziegler R, Egle K (1965) Zur quantitative Analyse der Chloropasten-pigmente. 1. Kritische Uberprufung des spectralphotometrischen Chlorophylls- Bestinungung. Beitra ge zur Biol der Pflanz 41:11–37
Acknowledgements
This work was funded by the Office of Science Early Career Research Program, Office of Biological and Environmental Research, of the U. S. Department of Energy under contract number DE-AC02-05CH11231. We thank Marco Voltolini (LBNL) for his assistance with all of the XRPD experiments and data analysis.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Responsible Editor: Fernando T. Maestre.
Electronic supplementary material
Supplementary Figure 1
(PDF 85 kb)
Supplementary Figure 2
(PDF 69 kb)
Supplementary Table 1
(XLSX 58 kb)
Supplementary Table 2
(XLSX 44 kb)
Supplementary Table 3
(DOCX 56 kb)
Supplementary Table 4
(XLSX 56 kb)
Rights and permissions
About this article
Cite this article
Swenson, T.L., Couradeau, E., Bowen, B.P. et al. A novel method to evaluate nutrient retention by biological soil crust exopolymeric matrix. Plant Soil 429, 53–64 (2018). https://doi.org/10.1007/s11104-017-3537-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-017-3537-x