Abstract
Aims
Zinc distribution at the tissue level is studied almost exclusively in lab-grown plants. It is essential to establish to what extent the patterns observed in lab-grown plants are corresponding with those in nature. To this end, we compared Zn localization in Noccaea caerulescens growing in its natural environment, a zinc/lead mine tailing, with that in hydroponically grown plants of the same origin.
Methods
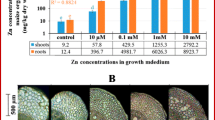
Zinc concentrations in plants and soil were determined by flame AAS and Zn localization in leaf tissues was studied using Zn indicators Zincon and Zinpyr-1.
Results
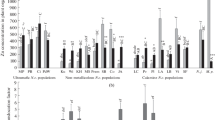
The mean Zn concentration in plants at the mine tailings was around 15,000 mg/kg DW, which corresponded well with the Zn concentration in the leaves of plants grown at 1600 μM Zn in the nutrient solution. The Zn distribution patterns in leaves of plants sampled from the mine and plants grown in hydroponics were identical. Zn-dependent staining was the most intensive in water-storage epidermal cells, guard cells and vascular bundles, and less intensive in subsidiary and mesophyll cells.
Conclusions
Zinc distribution in hydroponically grown plants is representative for plants in nature. Preferential Zn sequestration in leaves, particularly in water-storage epidermal cells, restricts metal accumulation in mesophyll and contributes to Zn hypertolerance.







Similar content being viewed by others
References
Assunção AGL, Schat H (2003) Thlaspi caerulescens, an attractive model species to study heavy metal hyperaccumulation in plants. New Phytol 159:351–360
Assunção AGL, da Costa Martins P, de Folter S, Voojis R, Schat H, Aarts MGM (2001) Elevated expression of metal transporter genes in three accessions of the metal hyperaccumulator Thlaspi caerulescens. Plant Cell Environ 24:217–226
Assunção AGL, Bookum WM, Nelissen HJM, Vooijs R, Schat H, Ernst WHO (2003) Differential metal-specific tolerance and accumulation patterns among Thlaspi caerulescens populations originating from different soil types. New Phytol 159:411–419
Assunção AGL, Bleeker P, Bookum WM, Vooijs R, Schat H (2008) Intraspecific variation of metal preference patterns for hyperaccumulation in Thlaspi caerulescens: evidence from binary metal exposures. Plant Soil 303:289–299
Becher M, Talke IN, Krall L, Krämer U (2004) Cross-species microarray transcript profiling reveals high constitutive expression of metal homeostasis genes in shoots of the zinc hyperaccumulator Arabidopsis halleri. Plant J 37:251–268
Burdette SC, Walkup GK, Spingler B, Tsien RY, Lippard SJ (2001) Fluorescent sensors for Zn(2+) based on a fluorescein platform: synthesis, properties and intracellular distribution. J Am Chem Soc 123:7831–7841
Craciun AR, Meyer C-L, Chen J, Roosens N, Groodt RD, Hilson P, Verbruggen N (2012) Variation in HMA4 gene copy number and expression among Noccaea caerulescens populations presenting different levels of Cd tolerance and accumulation. J Exp Bot 63:4179–4189
Deng T, Tang Y, van der Ent A, Sterckeman T, Echevarria G, Morel JL, Qiu RL (2016) Nickel translocation via the phloem in the hyperaccumulator Noccaea caerulescens (Brassicaceae). Plant Soil 404:35–45
Dinh NT, Vu DT, Mulligan D, Nguyen AV (2014) Accumulation and distribution of zinc in the leaves and roots of the hyperaccumulator Noccaea caerulescens. Environ Exp Bot 110:85–95
Dräger DB, Desbrosses-Fonrouge AG, Krach C, Chardonnens AN, Meyer RC, Saumitou-Laprade P, Krämer U (2004) Two genes encoding Arabidopsis halleri MTP1 metal transport proteins co-segregate with zinc tolerance and account for high MTP1 transcript levels. Plant J 39:425–439
Frerot H (2011) A challenge for hyperaccumulating plant models: 'cycling' as fast as Arabidopsis thaliana. New Phytol 189:357–359
Frey B, Keller C, Zierold K, Schulin R (2000) Distribution of Zn in functionally different leaf epidermal cells of the hyperaccumulator Thlaspi caerulescens. Plant Cell Environ 23:675–687
Gustin JL, Loureiro ME, Kim D, Na G, Tikhonova M, Salt DE (2009) MTP1-dependent Zn sequestration into shoot vacuoles suggests dual roles in Zn tolerance and accumulation in Zn-hyperaccumulating plants. Plant J 57:1116–1127
Halimaa P, Lin Y-F, Ahonen VH, Blane D, Clemens S, Gyenesei A, Haikio E, Karenlampi SO, Laiho A, Aarts MGM, Pursiheimo J-P, Schat H, Schmidt H, Tuomainen MH, Tervahauta AI (2014) Gene expression differences between Noccaea caerulescens ecotypes help indentifying candidate genes for metal phytoremediation. Environ Sci Technol 48:3344–3353
Hammond JP, Bowen HC, White PJ, Mills V, Pyke KA, Baker AJ, Whiting SN, May ST, Broadley MR (2006) A comparison of the Thlaspi caerulescens and Thlaspi arvense shoot transcriptomes. New Phytol 170:239–260
Hanikenne M, Talke IN, Haydon MJ, Lanz C, Nolte A, Motte P, Kroymann J, Weigel D, Krämer U (2008) Evolution of metal hyperaccumulation required cis-regulatory changes and triplication of HMA4. Nature 453:391–395
Iqbal M, Nawaz I, Hassan Z, Hakvoort HWJ, Bliek M, Aarts MAM, Schat H (2013) Expression of HMA4 cDNAs of the zinc hyperaccumulator Noccaea caerulescens from endogenous NcHMA4 promoters does not complement the zinc-deficiency phenotype of the Arabidopsis thaliana hma2hma4 double mutant. Front Plant Sci 4:article 404
Isaure MP, Huguet S, Meyer CL, Castillo-Michel H, Testemale D, Vantelon D, Saumitou-Laprade P, Verbruggen N, Sarret G (2015) Evidence of various mechanisms of Cd sequestration in the hyperaccumulator Arabidopsis halleri, the non-accumulator Arabidopsis lyrata, and their progenies by combined synchrotron-based techniques. J Exp Bot 66:3201–3214
Klein MA, Sekimoto H, Milner MJ, Kochian LV (2008) Investigation of heavy metal hyperaccumulation at the cellular level: development and characterization of Thlaspi caerulescens suspension cell lines. Plant Physiol 147:2006–2016
Koren’kov V, Park S, Cheng NH, Sreevidya C, Lachmansingh J, Morris J, Hirschi K, Wagner GJ (2007) Enhanced Cd2+-selective root-tonoplast-transport in tobaccos expressing Arabidopsis cation exchangers. Planta 225:403–411
Kozhevnikova AD, Erlikh NT, Zhukovskaya NV, Obroucheva NV, Ivanov VB, Seregin IV (2014a) Nickel and zinc accumulation, distribution and effects on ruderal plants Lepidium ruderale and Capsella bursa-pastoris. Acta Physiol Plant 36:3291–3305
Kozhevnikova AD, Seregin IV, Erlikh NT, Shevyreva TA, Andreev IM, Verweij R, Schat H (2014b) Histidine-mediated xylem loading of zinc is a species-wide character in Noccaea caerulescens. New Phytol 203:508–519
Krämer U (2010) Metal hyperaccumulation in plants. Annu Rev Plant Biol 61:517–534
Küpper H, Kochian LV (2010) Transcriptional regulation of metal transport genes and mineral nutrition during acclimatization to cadmium and zinc in the Cd/Zn hyperaccumulator Thlaspi caerulescens (Ganges population). New Phytol 185:114–129
Küpper H, Zhao FJ, McGrath SP (1999) Cellular compartmentation of zinc in leaves of the hyperaccumulator Thlaspi caerulescens. Plant Physiol 119:305–311
Küpper H, Lombi E, Zhao FJ, McGrath SP (2000) Cellular compartmentation of cadmium and zinc in relation to other elements in the hyperaccumulator Arabidopsis halleri. Planta 212:75–84
Küpper H, Lombi E, Zhao FJ, Wieshammer G, McGrath SP (2001) Cellular compartmentation of nickel in the hyperaccumulators Alyssum lesbiacum, Alyssum bertolonii and Thlaspi goesingense. J Exp Bot 52:2291–3000
Küpper H, Parameswaran A, Leitenmaier B, Trtílek M, Šetlík I (2007) Cadmium-induced inhibition of photosynthesis and long-term acclimation to cadmium stress in the hyperaccumulator Thlaspi caerulescens. New Phytol 175:655–674
Küpper H, Mijovilovich A, Götz B, Küpper FC, Meyer-Klaucke W (2009) Complexation and toxicity of copper in higher plants (I): characterisation of copper accumulation, speciation and toxicity in Crassula helmsii as a new copper hyperaccumulator. Plant Physiol 151:702–714
Leitenmaier B, Küpper H (2011) Cadmium uptake and sequestration kinetics in individual leaf cell protoplasts of the Cd/Zn hyperaccumulator Thlaspi caerulescens. Plant Cell Environ 34:208–219
Leitenmaier B, Küpper H (2013) Compartmentation and complexation of metals in hyperaccumulator plants. Front Plant Sci 4:1–13
Liu M-Q, Yanai J, Jiang R, Zhang F, McGrath S, Zhao F (2008) Does cadmium play a physiological role in the hyperaccumulator Thlaspi caerulescens? Chemosphere 71:1276–1283
Lu L, Tian S, Zhang J, Yang X, Labavitch JM, Webb SM, Latimer M, Brown PH (2013) Efficient xylem transport and phloem remobilization of Zn in the hyperaccumulator plant species Sedum alfredii. New Phytol 198:721–731
Ma JF, Ueno D, Zhao FJ, McGrath SP (2005) Subcellular localisation of Cd and Zn in the leaves of a Cd-hyperaccumulating ecotype of Thlaspi caerulescens. Planta 220:731–736
Marques L, Cossegal M, Bodin S, Czernic P, Lebrun M (2004) Heavy metal specificity of cellular tolerance in two hyperaccumulating plants, Arabidopsis halleri and Thlaspi caerulescens. New Phytol 164:289–295
Metcalfe CR, Chalk L (1950) Anatomy of the dicotyledons. Leaves, stem, and wood in relation to taxanomy with notes on economic uses, vol 1. Clarendon Press, Oxford, pp. 83–91
Meyer CL, Juraniec M, Huguet S, Chaves-Rodriguez E, Salis P, Isaure MP, Goormaghtigh E, Verbruggen N (2015) Intraspecific variability of cadmium tolerance and accumulation, and cadmium-induced cell wall modifications in the metal hyperaccumulator Arabidopsis halleri. J Exp Bot 66:3215–3227
Milner MJ, Kochian LV (2008) Investigating heavy-metal hyperaccumulation using Thlaspi caerulescens as a model system. Ann Bot 102:3–13
Monsant A, Wang Y, Tang C (2010) Nitrate nutrition enhances zinc hyperaccumulation in Noccaea caerulescens (Prayon). Plant Soil 336:391–404
Oomen RJ, Wu J, Lelièvre F, Blanchet S, Richaud P, Barbier-Brygoo H, Aarts MG, Thomine S (2009) Functional characterization of NRAMP3 and NRAMP4 from the metal hyperaccumulator Thlaspi caerulescens. New Phytol 181:637–650
Pauwels M, Roosens N, Frerot H, Saumitou-Laprade P (2008) When population genetics serves genomics: putting adaptation back in a spatial and historical context. Curr Opin Plant Biol 11:129–134
Richau KH, Schat H (2008) Intraspecific variation of nickel and zinc accumulation and tolerance in the hyperaccumulator Thlaspi caerulescens. Plant Soil 314:253–262
Richau KH, Kozhevnikova AD, Seregin IV, Vooijs R, Koevoets PLM, Smith JAC, Ivanov VB, Schat H (2009) Chelation by histidine inhibits the vacuolar sequestration of nickel in roots of the hyperaccumulator, Thlaspi caerulescens. New Phytol 183:106–116
Robinson BH, Leblanc M, Petit D, Brooks RR, Kirkman JH, Gregg PEH (1998) The potential of Thlaspi caerulescens for phytoremediation of contaminated soils. Plant Soil 203:47–56
Roosens N, Verbruggen N, Meerts P, Ximenez-Embun P, Smith JAC (2003) Natural variation in cadmium tolerance and its relationship to metal hyperaccumulation for seven populations of Thlaspi caerulescens from Western Europe. Plant Cell Environ 26:1657–1672
Roosens NH, Willems G, Saumitou-Laprade P (2008) Using Arabidopsis to explore zinc tolerance and hyperaccumulation. Trends Plant Sci 13:208–215
Schneider T, Persson DP, Husted S, Schellenberg M, Gehrig P, Lee Y, Martinoia E, Schjoerring JK, Meyer S (2013) A proteomics approach to investigate the process of Zn hyperaccumulation in Noccaea caerulescens (J & C.Presl) F. K. Meyer. Plant J 73:131–142
Seregin IV, Kozhevnikova AD (2008) Roles of root and shoot tissues in transport and accumulation of cadmium, lead, nickel, and strontium. Russ J Plant Physiol 55:1–22
Seregin IV, Kozhevnikova AD (2011) Histochemical methods for detection of heavy metals and strontium in the tissues of higher plants. Russ J Plant Physiol 58:721–727
Seregin IV, Kozhevnikova AD, Gracheva VV, Bystrova EI, Ivanov VB (2011) Tissue zinc distribution in maize seedling roots and its action on growth. Russ J Plant Physiol 58:109–117
Seregin IV, Erlikh NT, Kozhevnikova AD (2014) Nickel and zinc accumulation capacities and tolerance to these metals in the excluder Thlaspi arvense and the hyperaccumulator Noccaea caerulescens. Russ J Plant Physiol 61:204–214
Seregin IV, Kozhevnikova AD, Zhukovskaya NV, Schat H (2015) Cadmium tolerance and accumulation in excluder Thlaspi arvense and various accessions of hyperaccumulator Noccaea caerulescens. Russ J Plant Physiol 62:837–846
Sinclair SA, Sherson SM, Jarvis R, Camakaris J, Cobbet CS (2007) The use of the zinc-fluorophore Zinpyr-1, in the study of zinc homeostasis in Arabidopsis roots. New Phytol 174:39–45
Sokal RR, Rohlf FJ (1981) Biometry, 2nd edn. W.H. Freeman & Co, San Francisco
Solereder H (1899) Systematische anatomie der dicotyledonen. Verlag von Ferdinand Enke, Stuttgart, pp. 67–77
Tanaka N, Fujiwara T, Tomioka R, Krämer U, Kawachi M, Maeshima M (2015) Characterization of the histidine-rich loop of Arabidopsis vacuolar membrane zinc transporter AtMTP1 as a sensor of zinc level in the cytosol. Plant Cell Physiol 56:510–519
Ueno D, Milner MJ, Yamaji N, Yokosho K, Koyama E, Clemencia Zambrano M, Kaskie M, Ebbs S, Kochian LV, Ma JF (2011) Elevated expression of TcHMA3 plays a key role in the extreme Cd tolerance in a Cd-hyperaccumulating ecotype of Thlaspi caerulescens. Plant J 66:852–862
van der Ent A, Baker AJM, Reeves RD, Pollard AJ, Schat H (2013) Hyperaccumulators of metal and metalloiod trace elements: facts and fiction. Plant Soil 362:319–334
van der Zaal BJ, Neuteboom LW, Pinas JE, Chardonnens AN, Schat H, Verkleij JA, Hooykaas P (1999) Overexpression of a novel Arabidopsis gene related to putative zinc-transporter genes from animals can lead to enhanced zinc resistance and accumulation. Plant Physiol 119:1047–1055
Vazquez MD, Barcelo J, Poschenrieder C, Madico J, Hatton P, Baker AJM, Cope GH (1992) Localization of zinc and cadmium in Thlaspi caerulescens (Brassicaceae), a metallophyte that can hyperaccumulate both metals. J Plant Physiol 140:350–355
Vazquez MD, Poschenrieder C, Barcelo J, Baker AJM, Hatton P, Cope GH (1994) Compartmentation of zinc in roots and leaves of the zinc hyperaccumulator Thlaspi caerulescens. Bot Acta 107:243–250
Verbruggen N, Hermans C, Schat H (2009) Molecular mechanisms of metal hyperaccumulation in plants. New Phytol 181:759–776
Visioli G, Gullì M, Marmiroli N (2014) Noccaea caerulescens populations adapted to grow in metalliferousand non-metalliferous soils: Ni tolerance, accumulation and expression analysis of genes involved in metal homeostasis. Environ Exp Bot 105:10–17
Vogel-Mikus K, Simcic J, Pelicon P, Budnar M, Kump P, Necemer M, Mesjasz-Przybylowicz J, Przybylowicz WJ, Regvar M (2008) Comparison of essential and non-essential element distribution in leaves of the Cd/Zn hyperaccumulator Thlaspi praecox as revealed by micro-PIXE. Plant Cell Environ 31:1484–1496
Acknowledgements
The authors are grateful to Victor Ivanov for critical discussion of the results, to Rudo Verweij, Rob Broekman, Richard van Logtestijn and Riet Vooijs for technical support, Marc Aarts and Patrick Doumas for providing the coordinates of the mines. This work was partially supported by the grants from the Russian Foundation for Basic Research (RFBR, № 15-04-02236) and from the international scientific program GDRI LOCOMET (Transport, localization and complexation of metals in hyperaccumulating plants) funded by The National Center for Scientific Research, as well as by the French Embassy Metchnikov scholarship funded by Campus France.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Juan Barcelo.
Rights and permissions
About this article
Cite this article
Kozhevnikova, A.D., Seregin, I.V., Gosti, F. et al. Zinc accumulation and distribution over tissues in Noccaea сaerulescens in nature and in hydroponics: a comparison. Plant Soil 411, 5–16 (2017). https://doi.org/10.1007/s11104-016-3116-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-016-3116-6