Abstract
Aims
Coastal sand dunes have a well-established abiotic gradient from beach to land and a corresponding spatial gradient of plant species representing succession in time. Here, we relate the distribution of plant-feeding nematodes with dominant plant species in the field to host specialization and impacts on plant species under controlled greenhouse conditions.
Methods
We assessed plant-feeding nematodes in soil and roots of six plant species that dominate the vegetation at successional positions along the gradient. In controlled conditions, we determined performance of all plant-feeding nematodes on each plant species and their effects on plant biomass.
Results
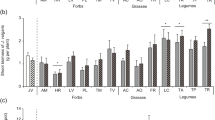
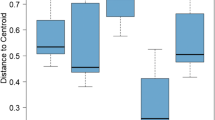
Specialist feeding type nematodes were confined to plant species in either foredunes or landward dunes. Generalist feeding type nematodes were found in highest numbers in the landward dunes. Most tested nematode species decreased root, but not shoot or rhizome biomass.
Conclusions
Host plant suitability determined occurrence of some plant-feeding nematodes in dunes, but abiotic and biotic soil conditions may play a role as well. Generalist feeding type nematodes were able to reproduce on all plant species. Feeding specialists, which are more protected by plant roots, might prefer host plants in the foredunes for the same reason as their host plants: to escape from natural enemies.


Similar content being viewed by others
Abbreviations
- Pf/Pi:
-
(ratio of final to initial population size)
References
Bardgett RD, Wardle DA (2010) Aboveground-belowground linkages: biotic interactions, ecosystem processes, and global change. Oxford University Press, Oxford
Bezemer TM, Fountain MT, Barea JM, Christensen S, Dekker SC, Duyts H, van Hal R, Harvey JA, Hedlund K, Maraun M, Mikola J, Mladenov AG, Robin C, de Ruiter PC, Scheu S, Setälä H, Šmilauer P, van der Putten WH (2010) Divergent composition but similar function of soil food webs of individual plants: plant species and community effects. Ecology 91:3027–3036
Boag B, Neilson R (1996) Distribution and ecology of Rotylenchus and Pararotylenchus (Nematoda: Hoplolaimidae) in Great Britain. Nematologica 42:96–108
Bongers T (1988) De nematoden van Nederland. Pirola, Schoorl, The Netherlands
Brinkman EP, van Veen JA, van der Putten WH (2004) Endoparasitic nematodes reduce multiplication of ectoparasitic nematodes, but do not prevent growth reduction of Ammophila arenaria (L.) Link (marram grass). Appl Soil Ecol 27:65–75
Brinkman EP, Duyts H, van der Putten WH (2005) Consequences of variation in species diversity in a community of root-feeding herbivores for nematode dynamics and host plant biomass. Oikos 110:417–427
Brinkman EP, Duyts H, van der Putten WH (2008) Interactions between root-feeding nematodes depend on plant species identity. Soil Biol Biochem 40:2186–2193. doi:10.1016/j.soilbio.2008.01.023
Clapp JP, van der Stoel CD, van der Putten WH (2000) Rapid identification of cyst (Heterodera spp., Globodera spp.) and root-knot (Meloidogyne spp.) nematodes on the basis of ITS2 sequence variation detected by PCR-single-strand conformational polymorphism (PCR-SSCP) in cultures and field samples. Mol Ecol 9:1223–1232
Costa SR, Kerry BR, Bardgett RD, Davies KG (2012) Interactions between nematodes and their microbial enemies in coastal sand dunes. Oecologia 170:1053–1066. doi:10.1007/s00442-012-2359-z
Davis LT, Bell NL, Watson RN, Rohan TC (2004) Host range assessment of Helicotylenchus pseudorobustus (Tylenchida : Hoplolaimidae) on pasture species. J Nematol 36:487–492
De Boer W, Klein Gunnewiek PJA, Woldendorp JW (1998) Suppression of hyphal growth of soil-borne fungi by dune soils from vigorous and declining stands of Ammophila arenaria. New Phytol 138:107–116
de la Peña E, Moens M, van Aelst A, Karssen G (2006a) Description of Pratylenchus dunensis sp n. (Nematoda : Pratylenchidae), a root-lesion nematode associated with the dune grass Ammophila arenaria (L.) Link. Nematology 8:79–88. doi:10.1163/156854106776179917
de la Peña E, Rodríguez Echeverría S, van der Putten WH, Freitas H, Moens M (2006b) Mechanism of control of root-feeding nematodes by mycorrhizal fungi in the dune grass Ammophila arenaria. New Phytol 169:829–840
de la Peña E, Vandegehuchte M, Bonte D, Moens M (2008) Analysis of the specificity of three root-feeders towards grasses in coastal dunes. Plant Soil 310:113–120. doi:10.1007/s11104-008-9636-y
Doing H (1985) Coastal fore-dune zonation and succession in various parts of the world. Vegetatio 61:65–75. doi:10.1007/bf00039811
Erb M, Lu J (2013) Soil abiotic factors influence interactions between belowground herbivores and plant roots. J Exp Bot 64:1295–1303. doi:10.1093/jxb/ert007
Hol WHG, de la Peña E, Moens M, Cook R (2007) Interaction between a fungal endophyte and root herbivores of Ammophila arenaria. Basic Appl Ecol 8:500–509. doi:10.1016/j.baae.2006.09.013
Hope-Simpson JF, Jefferies RL (1966) Observations relating to vigour and debility in marram grass (Ammophila arenaria (L.) Link). J Ecol 54:271–275
Huiskes AHL (1979) Biological flora of the British isles: Ammophila arenaria (L.) Link (Psamma arenaria (L.) Roem. et Schult.: Calamagrostis arenaria (L.) Roth). J Ecol 67:363–382
Jones JT, Haegeman A, Danchin EGJ, Gaur HS, Helder J, Jones MGK, Kikuchi T, Manzanilla-Lopez R, Palomares-Rius JE, Wesemael WML, Perry RN (2013) Top 10 plant-parasitic nematodes in molecular plant pathology. Mol Plant Pathol 14:946–961. doi:10.1111/mpp.12057
Karssen G, Van Aelst A, Cook R (1998a) Redescription of the root-knot nematode Meloidogyne maritima Jepson, 1987 (Nematoda : Heteroderidae), a parasite of Ammophila arenaria (L.) Link. Nematologica 44:241–253
Karssen G, Van Aelst A, van der Putten WH (1998b) Meloidogyne duytsi n. sp. (Nematoda : Heteroderidae), a root- knot nematode from Dutch coastal foredunes. Fundam Appl Nematol 21:299–306
Karssen G, Waeyenberge L, Moens M (2000) Pratylenchus brzeskii sp nov (Nematoda : Pratylenchidae), a root-lesion nematode from European coastal dunes. Ann Zool 50:255–261
Little LR, Maun MA (1997) Relationships among plant-parasitic nematodes, mycorrhizal fungi and the dominant vegetation of a sand dune system. Ecoscience 4:67–74
Loof PAA (1963) A new species of Telotylenchus (Nematoda: Tylenchida). Nematologica 9:76–80
Loof PAA (1984) Hemicycliophora species from Iran (Nematoda: Criconematoidea). Nematologica 30:22–41
Mateille T, Tavoillot J, Martiny B, Fargette M, Chapuis E, Baudouin M, Dmowska E, Bouamer S (2011) Plant-associated nematode communities in West-palearctic coastal foredunes may relate to climate and sediment origins. Appl Soil Ecol 49:81–93. doi:10.1016/j.apsoil.2011.06.012
Neher DA (2010) Ecology of plant and free-living nematodes in natural and agricultural soil. In: VanAlfen NK, Bruening G, Leach JE (eds) Annual Review of Phytopathology, Vol 48, vol 48. Annual Review of Phytopathology. pp 371–394. doi:10.1146/annurev-phyto-073009-114439
Nico AI, Rapoport HF, Jiménez-Díaz RM, Castillo P (2002) Incidence and population density of plant-parasitic nematodes associated with olive planting stocks at nurseries in Southern Spain. Plant Dis 86:1075–1079
Nkem JN, Virginia RA, Barrett JE, Wall DH, Li G (2006) Salt tolerance and survival thresholds for two species of Antarctic soil nematodes. Polar Biol 29:643–651. doi:10.1007/s00300-005-0101-6
Oostenbrink M (1960) Estimating nematode populations by some selected methods. In: Sasser JN, Jenkins WR (eds) Nematology. The University of North Carolina Press, Chapel Hill, pp 85–102
Oremus PAI, Otten H (1981) Factors affecting growth and nodulation of Hippophaë rhamnoides L. ssp. rhamnoides in soils from two successional stages of dune formation. Plant Soil 63:317–331
Perry RN, Moens M (2006) Plant nematology. CABI Publishing, Wallingford
Piśkiewicz AM, Duyts H, van der Putten WH (2008) Multiple species-specific controls of root-feeding nematodes in natural soils. Soil Biol Biochem 40:2729–2735. doi:10.1016/j.soilbio.2008.07.006
Rohlf FJ, Sokal RR (1981) Statistical tables, 2nd edn. W.H. Freeman and Company, San Fransisco
Rooij-van D, der Goes PCEM (1995) The role of plant-parasitic nematodes and soil-borne fungi in the decline of Ammophila arenaria (L.) Link. New Phytol 129:661–669
Rooij-van D, der Goes PCEM, van der Putten WH, van Dijk C (1995) Analysis of nematodes and soil-borne fungi from Ammophila arenaria (Marram Grass) in Dutch coastal foredunes by multivariate techniques. Eur J Plant Pathol 101:149–162
Seliskar DM, Huettel RN (1993) Nematode involvement in the dieout of Ammophila breviligulata (Poaceae) on the mid-Atlantic coastal dunes of the United States. J Coast Res 9:97–103
Silva RA, Oliveira CMG, Inomoto MM (2008) Fauna of plant-parasitic nematodes in natural and cultivated areas of the Amazon forest, Mato Grosso State, Brazil. Trop Plant Pathol 33:204–211
Sokal RR, Rohlf FJ (1995) Biometry: the principles and practice of statistics in biological research, 3rd edn. W.H. Freeman and Company, New York
Van der Laan D, van Tongeren OFR, van der Putten WH, Veenbaas G (1997) Vegetation development in coastal foredunes in relation to methods of establishing marram grass (Ammophila arenaria). J Coastal Conserv 3:179–190
Van der Putten WH, van Dijk C, Troelstra SR (1988) Biotic soil factors affecting the growth and development of Ammophila arenaria. Oecologia 76:313–320
Van der Putten WH, van Dijk C, Peters BAM (1993) Plant-specific soil-borne diseases contribute to succession in foredune vegetation. Nature 362:53–56
Van der Putten WH, Cook R, Costa S, Davies KG, Fargette M, Freitas H, Hol WHG, Kerry BR, Maher N, Mateille T, Moens M, de la Peña E, Piśkiewicz AM, Raeymaekers ADW, Rodríguez-Echeverría S, van der Wurff AWG (2006) Nematode interactions in nature: models for sustainable control of nematode pests of crop plants? Adv Agron 89:227–260
Van der Stoel CD, van der Putten WH (2006) Pathogenicity and host range of Heterodera arenaria in coastal foredunes. Nematology 8:255–263
Van der Stoel CD, van der Putten WH, Duyts H (2002) Development of a negative plant-soil feedback in the expansion zone of the clonal grass Ammophila arenaria following root formation and nematode colonization. J Ecol 90:978–988
Van der Stoel CD, Duyts H, van der Putten WH (2006) Population dynamics of a host-specific root-feeding cyst nematode and resource quantity in the root zone of a clonal grass. Oikos 112:651–659
Verschoor BC, de Goede RGM, de Hoop JW, de Vries FW (2001a) Seasonal dynamics and vertical distribution of plant-feeding nematode communities in grasslands. Pedobiologia 45:213–233. doi:10.1078/0031-4056-00081
Verschoor BC, de Goede RGM, de Vries FW, Brussaard L (2001b) Changes in the composition of the plant-feeding nematode community in grasslands after cessation of fertiliser application. Appl Soil Ecol 17:1–17. doi:10.1016/s0929-1393(00)00135-9
Wall JW, Skene KR, Neilson R (2002) Nematode community and trophic structure along a sand dune succession. Biol Fertil Soils 35:293–301
Wilson JB, Sykes MT (1999) Is zonation on coastal sand dunes determined primarily by sand burial or by salt spray? A test in New Zealand dunes. Ecol Lett 2:233–236
Zoon FC, Troelstra SR, Maas PWT (1993) Ecology of the plant-feeding nematode fauna associated with sea buckthorn (Hippophaë rhamnoides L. ssp. rhamnoides) in different stages of dune succession. Fundam Appl Nematol 16:247–258
Acknowledgments
We thank Sven-Erik Burger and André Kamp for assistance with the greenhouse experiments. The former Water and Civil Board ‘De Brielse Dijkring’ kindly permitted to sample their terrains. This is NIOO-KNAW publication 5821.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Hans Lambers.
Rights and permissions
About this article
Cite this article
Brinkman, E.P., Duyts, H., Karssen, G. et al. Plant-feeding nematodes in coastal sand dunes: occurrence, host specificity and effects on plant growth. Plant Soil 397, 17–30 (2015). https://doi.org/10.1007/s11104-015-2447-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-015-2447-z