Abstract
Background and aims
Burkholderia phymatum strain GR01 is a recently reported common bean (Phaseolus vulgaris) symbiont isolated from nodules of plants grown in semi-arid soils in Morocco. The osmotolerance of B. phymatum GR01N under free-living and in symbiotic association with P. vulgaris was investigated in this study.
Methods
The osmotolerance of B. phymatum GR01N was checked by growing cells in the presence of varying concentrations of NaCl or sucrose, and the cellular solutes were analyzed in cell extracts by 13C-nuclear magnetic resonance (NMR) spectroscopy. Nodule occupancy was checked in P. vulgaris grown in the presence of 25, 35 or 50 mM NaCl and inoculated with a mixture of B. phymatum GR01N and R. tropici CIAT899R cells. The effect of salt stress on nodule biomass, plant dry weight, plant nitrogen content and leghaemoglobin content of nodules was also analyzed in plants inoculated with either B. phymatum GR01N or R. tropici CIAT899R and grown in the presence of 25 or 35 mM NaCl.
Results
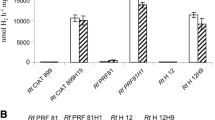
Burkholderia phymatum strain GR01N showed increased tolerance to osmotic stress under free-living conditions as compared to the reference strain R. tropici CIAT899R. Strain GR01N accumulated trehalose, mannitol and alanine in response to saline stress, suggesting their role in the observed osmoloterance. Under conditions of saline stress, P. vulgaris plants nodulated by B. phymatum GR01N showed increased plant dry weight and nitrogen fixation, when compared to those inoculated with R. tropici CIAT899R. Nodule competition assays revealed that B. phymatum GR01N had higher levels of nodule occupancy than R. tropici CIAT899R in P. vulgaris plants grown under saline conditions.
Conclusions
Burkholderia phymatum strain GR01N displays a remarkable osmotolerance under free-living and symbiotic conditions.



Similar content being viewed by others
References
Angus AA, Hirsch AM (2010) Insights into the history of the legumes-betaproteobacterial symbiosis. Mol Ecol 19:28–30
Ardley JK, Parker MA, De Meyer SE, Trengove RD, O'Hara GW, Reeve WG, Yates RJ, Dilworth MJ, Willems A, Howieson JG (2011) Microvirga lupini sp. nov., Microvirga lotononidis sp. nov., and Microvirga zambiensis sp. nov. are Alphaproteobacterial root nodule bacteria that specifically nodulate and fix nitrogen with geographically and taxonomically separate legume hosts. Int J Syst Evol Microbiol. doi:10.1099/ijs.0.035097-0
Aydi S, Sassi S, Abdelley C (2008) Growth, nitrogen fixation and ion distribution in Medicago truncatula subjected to salt stress. Plant Soil 312:59–67
Baker A, Sprent J, Wilson J (1995) Effects of sodium chloride and mycorrhizal infection on the growth and nitrogen fixation of Prosopis juliflora. Symbiosis 19:39–51
Bao Y, Lies DP, Fu H, Roberts GP (1991) An improved Tn7-based system for the single-copy insertion of cloned genes into chromosomes of Gram-negative bacteria. Gene 109:167–168
Behrends V, Bundy JG, Williams HD (2011) Differences in strategies to combat osmotic stress in Burkholderia cenocepacia elucidated by NMR-based metabolic profiling. Lett Appl Microbiol 52:619–625
Ben Romdhane S, Tajini F, Trabelsi M, Aouani ME, Mhamdi R (2007) Competition for nodule formation between introduced strains of Mesorhizobium ciceri and native populations of rhizobia nodulating chickpea (Cicer arietinum) in Tunisia. World J Microbiol Biotechnol 23:1195–1201
Beringer J (1974) R factor transfer in Rhizobium leguminosarum. J Gen Microbiol 84:188–189
Bontemps C, Elliott GN, Simon MF, Dos Reis F, Gross E, Lawton R, Neto NE, Loureiro MF, Faria SM, Sprent JI, James EK, Young JPW (2010) Burkholderia species are ancient symbionts of legumes. Mol Ecol 19:44–52
Bouhmouch I, Souad-Mouhsine B, Brhada F, Aurag J (2005) Influence of host cultivars and Rhizobium species on the growth and symbiotic performance of Phaseolus vulgaris under salt stress. J Plant Physiol 162:1103–1113
Chen WM, Laevens S, Lee TM, Coenye T, de Vos P, Mergeay M, Vandamme P (2001) Ralstonia taiwanensis spp. nov. isolated from root nodules of Mimosa species and sputum of a cystic fibrosis patient. Int J Syst Evol Microbiol 51:1729–1735
Chen WM, James EK, Prescott AR, Kierans M, Sprent JI (2003) Nodulation of Mimosa spp. by the β-proteobacterium Ralstonia taiwanensis. Mol Plant Microbe Interact 16:1051–1061
Chen WM, de Faria SM, Straliotto R, Pitard RM, Simoes-Araujo JL, Chou JH, Chou YJ, Barrios E, Prescott AR, Elliott GN, Sprent JI, Young JPW, James EK (2005a) Proof that Burkholderia strains form effective symbioses with legumes: a study of novel Mimosa nodulating strains from South America. Appl Environ Microbiol 71:7461–7471
Chen WM, James EK, Chou J-H, Sheu S-Y, Yang S-Z, Sprent JI (2005b) Beta-rhizobia from Mimosa pigra, a newly-discovered invasive plant in Taiwan. New Phytol 168:661–675
Cordovilla MP, Ligero F, Lluch C (1996) Growth and nitrogen assimilation in nodules response to nitrate levels in Vicia faba under salt stress. J Exp Bot 47:203–210
da Costa MS, Santos H, Galinski EA (1998) An overview of the role and diversity of compatible solutes in Bacteria and Archaea. Adv Biochem Eng Biotechnol 61:117–153
Delgado MJ, Olivares J, Bedmar EJ (1989) Nitrate reductase activity of free-living and symbiotic uptake hydrogenase positive and uptake hydrogenase-negative strains of Bradyrhizobium japonicum. Arch Microbiol 151:166–170
Delgado MJ, Garrido JM, Ligero F, Lluch C (1993) Nitrogen fixation and carbon metabolismo by nodules and bacteroids of pea plants under chloride stress. Physiol Plant 89:824–829
Delgado MJ, Ligero F, Lluch C (1994) Effect of salt stress on growth and nitrogen fixation by pea, faba bean, common bean and soybean plants. Soil Biol Biochem 26:371–376
Domínguez-Ferreras A, Muñoz S, Olivares J, Soto MJ, Sanjuán J (2009a) Role of potassium uptake systems in Sinorhizobium meliloti osmoadaptation and symbiotic performance. J Bacteriol 191:2133–2143
Domínguez-Ferreras A, Soto MJ, Pérez-Arnedo R, Olivares J, Sanjuán J (2009b) Importance of trehalose biosynthesis for Sinorhizobium meliloti osmotolerance and nodulation of Alfalfa roots. J Bacteriol 191:7490–7499
dos Reis Junior FB, Simon MF, Gross E, Boddey RM, Elliot GN, Neto NE, Loureiro MF, Queiroz LP, Scott MR, Chen W-M, Norén A, Rubio MC, de Faria SM, Bontemps C, Goi SR, Young JPW, Sprent JI, James EK (2010) Nodulation and nitrogen fixation by Mimosa spp. in the Cerrado and Caatinga biomes of Brazil. New Phytol 186:934–946
Dua RP (1992) Differencial response of chickpea (Cicer arietimum) genotypes to salinity. J Agr Sci 119:367–71
Elliott GN, Chen WM, Chou J-H, Wang H-C, Sheu S-Y, Perin L, Reis VM, Moulin L, Simon MF, Bontemps C, Sutherland JM, Bessi R, de Faria SM, Trinick MJ, Prescott AR, Sprent JI, James EK (2007a) Burkholderia phymatum is a highly effective nitrogen-fixing symbiont of Mimosa spp. and fixes nitrogen ex planta. New Phytol 173:168–180
Elliott GN, Chen W-M, Bontemps C, Chou J-H, Young JPW, Sprent JI, James EK (2007b) Nodulation of Cyclopia spp. (Leguminosae, Papilionoideae) by Burkholderia tuberum. Ann Bot 100:1403–1411
Elliott GN, Chou JH, Chen WM, Bloemberg G, Bontemps C, Martínez-Romero E, Velázquez E, Young JPW, Sprent JI, James JK (2009) Burkholderia spp. are the most competitive symbionts of Mimosa, particularly under N-limited conditions. Environ Microbiol 11:762–778
Fernandez-Aunión C, Ben Hamouda T, Iglesias-Guerra F, Argandoña M, Reina-Bueno M, Nieto JJ, Elarbi-Aouani M, Vargas C (2010) Biosynthesis of compatible solutes in rhizobial strains isolated from Phaseolus vulgaris nodules in Tunisian fields. BMC Microbiol 192:3–16
Garau G, Yates RJ, Deiana P, Howieson JG (2009) Novel strains of nodulating Burkholderia have a role in nitrogen fixation with papilionoid herbaceous legumes adapted to acid, infertile soils. Soil Biol Biochem 41:125–134
García-Estepa R, Argandoña M, Reina-Bueno M, Capote N, Iglesias-Guerra F, Nieto JJ, Vargas C (2006) The ectD gene, which is involved in the synthesis of the compatible solute hydroxyectoine, is essential for thermoprotection of the halophilic bacterium Chromohalobacter salexigens. J Bacteriol 88:3774–3784
Garg N, Singla R (2004) Growth, photosynthesis, nodule nitrogen and carbon fixation in the chickpea cultivars under salt stress. Braz J Plant Physiol 16:137–146
Gepts P (1990) Biochemical evidence bearing on the domestication of Phaseolus (Fabaceae) beans. Econ Bot 44:28–38
Gepts P, Bliss FA (1988) Dissemination pathways of common bean (Phaseolus vulgaris, Fabaceae) deduced from phaseolin electrophoretic variability II Europe and Africa. Econ Bot 42:86–104
Graham PH (1981) Some problems of nodulation and symbiotic nitrogen fixation in Phaseolus vulgaris L.: a review. Field Crop Res 4:93–112
Graham PH (2008) Ecology of the root-nodule bacteria of legumes. In: Nitrogen-fixing Leguminous. In: Dilworth MJ, James EK, Sprent JI, Newton WE (eds) Symbiosis Springer. The Netherlands, Dordrecht, pp 23–43
Graham PH, Draeger KJ, Ferrey ML, Conroy MJ, Hammer BE, Martinez E, Aarons SR, Quinto C (1994) Acid pH tolerance in strains of Rhizobium and Bradyrhizobium and initial studies on the basis for acid tolerance of Rhizobium tropici UMR 1899. Can J Microbiol 40:198–207
Gyaneshwar P, Hirsch AM, Moulin L, Chen WM, Elliott GN, Bontemps C, Estrada-de los Santos P, Gross E, Dos Reis Junior FB, Sprent JI, Young JP, James EK (2011) Legume-nodulating betaproteobacteria: diversity, host range, and future prospects. Mol Plant Microbe Interact 24:1276–88
Hardarson G (1993) Methods for enhancing symbiotic nitrogen fixation. Plant Soil 152:1–17
Kessler B, de Lorenzo V, Timmis KN (1992) A general system to integrate lacZ fusions into the chromosome of Gram-negative eubacteria: regulation of the Pm promoter in the TOL plasmid studied with all controlling elements in monocopy. Mol Gen Genet 233:293–301
Kets EP, Galinski EA, de Wit M, de Bont JA, Heipieper HJ (1996) Mannitol, a novel bacterial compatible solute in Pseudomonas putida S12. J Bacteriol 178:6665–1670
Koch B, Jensen LE, Nybroe O (2001) A panel of Tn7-based vectors for insertion of gfp marker gene or for delivery of cloned DNA into Gram-negative bacteria at a neutral chromosomal site. J Microbiol Methods 45:187–195
Lambertsen L, Sternberg C, Molin S (2004) Mini-Tn7 transposons for site-specific tagging of bacteria with fluorescent proteins. Environ Microbiol 6:726–732
LaRue TA, Child JJ (1979) Sensitive fluorometric assay for leghaemoglobin. Anal Biochem 92:11–15
Leonard LT (1943) A simple assembly for use in testing of culture of rhizobia. J Bacteriol 45:523–527
Lewis G, Schrire B, Mackinder B, Lock M (2005) Legumes of the world. Royal Botanic Gardens, Kew
Liesack W, Janssen PH, Rainey FA, Ward-Rainey NL, Stackebrandt E (1997) Microbial diversity in soil: the need for a combined approach using molecular and cultivation techniques. In: Van Elsas JD, Trevors JT, Wellington EMH (eds) Modern Soil Microbiology. Marcel Dekker, New York, pp 375–439
Martínez-Romero E, Segovia L, Mercante FM (1991) Rhizobium tropici, a novel nodulating Phaseolus vulgaris L. bean and Leucaena sp. trees. Int J Syst Bacteriol 41:417–426
Meuelenberg F, Dakora FD (2007) Assessing the biological potential of N2-fixing leguminosae in Botswana for increased crop yields and commercial exploitation. Afr J Biotechnol 6:325–334
Mhadhbi H, Jebara M, Zitoun A, Limam F, Aouani ME (2008) Symbiotic effectiveness and response to mannitol-mediated osmotic stress of various chickpea-rhizobia associations. World J Microbiol Biotechnol 24:1027–1035
Mhadhbi H, Chihaoui S, Mhamdi R, Mnasri B, Jebara M, Mhamdi R (2011) A highly osmotolerant rhizobial strain confers a better tolerance of nitrogen fixation and enhances protective activities to nodules of Phaseolus vulgaris under drought stress. Afr J Biotechnol 10:4555–4563
Miller-Williams M, Loewen PC, Oresnik IJ (2006) Isolation of salt-sensitive mutants of Sinorhizobium meliloti strain Rm1021. Microbiol 152:2049–2059
Mishra RPN, Tisseyre P, Melkonian R, Chaintreuil C, Miché L, Klonowska A, González S, Bena G, Laguerre G, Moulin L (2012) Genetic diversity of Mimosa pudica rhizobial symbionts in soils of French Guiana: investigating the origin and biodiversity of Burkholderia phymatum and other beta-proteobacteria. FEMS Microbiol Ecol 79:487–503
Mnasri B, Mrabet M, Laguerre G, Aouani ME, Mhamdi R (2007) Salt tolerant rhizobia isolated from a Tunisian oasis that are highlyeffective for N2-Fixation with Phaseolus vulgaris constitute a novel biovar (bv. mediterranense) of Sinorhizobium meliloti. Arch Microbiol 187:79–85
Morón B, Soria-Díaz ME, Ault J, Verroios G, Noreen S, Rodríguez-Navarro DN, Gil-Serrano A, Thomas-Oates J, Megías M, Sousa C (2005) Low pH changes the profile of nodulation factors produced by Rhizobium tropici CIAT899. Chemistry Biol 12:1029–1040
Moulin L, Munive A, Dreyfus B, Boivin-Masson C (2001) Nodulation of legumes by members of the β-subclass of Proteobacteria. Nature 411:948–950
Nogales J, Campos R, Ben Abdelkhalek H, Olivares J, Lluch C, Sanjuan J (2002) Rhizobium tropici genes involved in free-living salt tolerance are required for the establishment of efficient nitrogen-fixing symbiosis with Phaseolus vulgaris. Mol Plant–Microbe Interact 15:225–232
Oren A (1999) Bioenergetic aspects of halophilism. Microbiol Mol Biol Rev 63:334–348
Oren A, Heldal M, Norland S, Galinski EA (2002) Intracellular ion and organic solute concentrations of the extremely halophilic bacterium Salinibacter ruber. Extremophiles 6:491–498
Ramos LMG, Boddey RM (1987) Yield and nodulation of Phaseolus vulgaris and the competitiveness of an introduced Rhizobium strain: effects of lime, mulch and repeated cropping. Soil Biol Chem 19:171–177
Rao DL, Giller N, Yeo KE, Flowers TJ (2002) The effects of salinity and sodicity upon nodulation and nitrogen Fixation in Chickpea (Cicer arietinum). Ann Bot 89:563–570
Rasband WS (1997) ImageJ. National Institutes of Health. Bethesda, Maryland, USA. http://rsbinfonihgov/ij/.
Reis VM, Estrada-de los Santos P, Tenorio-Salgado S, Vogel J, Stoffels M, Guyon S, Mavingui P, Baldani VLD, Schmid M, Baldani JI, Balandreau J, Hartmann A, Caballero-Mellado J (2004) Burkholderia tropica sp. nov., a novel nitrogen-fixing, plant-associated bacterium. Int J Syst Evol Microbiol 54:2155–2162
Riccillo PM, Collavino MM, Grasso DH, England R, de Bruijn FJ, Aguilar OM (2000) A guaB mutant of Rhizobium tropici CIAT899 pleitropically defective in thermal tolerance and symbiosis. Mol Plant-Microbe Interact 13:1228–1236
Rigaud J, Puppo A (1975) Indol-3-acetic catabolism by soybean bacteroids. J Gen Microbiol 88:223–228
Rivas R, Velázquez E, Willems A, Vizcaíno N, Subba-Rao NS, Mateos PF, Gillis M, Dazzo FB, Martínez-Molina E (2002) A new species of Devosia that forms a unique nitrogen-fixing root nodule symbosis with the aquatic legume Neptunia natans (L.F.) Druce. Appl Environ Microbiol 68:5217–5222
Rivas R, García-Fraile P, Velázquez E (2009) Taxonomy of bacteria nodulating legumes. Rev Microbiol Insight 2:251–269
Sambrook J, Russell DW (2004) Molecular Cloning: a Laboratory Manual, 3dth edn. Harbor Laboratory Press, Cold Spring Harbor, New York, Cold Spring
Shamseldin A, Werner D (2005) High salt and high pH tolerance of new isolated Rhizobium etli strains from Egyptian soils. Curr Microbiol 50:11–16
Sheu SY, Chou JH, Bontemps C, Elliott GN, Gross E, James EK, Sprent JI, Young JP, Chen WM (2012a) Burkholderia symbiotica sp. nov., isolated from root nodules of Mimosa spp. native to North East Brazil. Int J Syst Evol Microbiol. doi:10.1099/ijs.0.037408-0
Sheu S-Y, Chou J-H, Bontemps C, Elliott GN, Gross E, dos Reis Junior FB, Melkonian R, Moulin L, James EK, Sprent JI, Young JPW, Chen W-M (2012b) Burkholderia diazotrophica sp. nov., isolated from root nodules of Mimosa spp. Int J Syst Evol Microbiol. doi:10.1099/ijs.0.039859-0
Smith LT, Smith GM, Madkour MA (1990) Osmoregulation in Agrobacterium tumefaciens: accumulation of a novel disaccharide is controlled by osmotic strength and glycine betaine. J Bacteriol 172:6849–6855
Spriggs AC, Dakora FD (2009) Assessing the suitability of antibiotic resistance markers and the indirect ELISA technique for studying the competitive ability of selected Cyclopia Vent. rhizobia under glasshouse and field conditions in South Africa. BMC Microbiol 9:1–11
Strøm AR, Kaasen I (1993) Trehalose metabolism in Escherichia coli: stress protection and stress regulation of gene expression. Mol Microbiol 8:205–210
Stuurman N, Pacios Bras C, Schlaman HRM, Wijfjes AHM, Bloemberg G, Spaink HP (2000) Use of green fluorescent protein color variants expressed on stable broad-range vectors to visualize rhizobia interacting with plants. Mol Plant-Microbe Interact 13:1163–1169
Sugawara M, Cytryn EJ, Sadowsky MJ (2010) Functional role of Bradyrhizobium japonicum trehalose biosynthesis and metabolism genes during physiological stress and nodulation. Appl Environ Microbiol 76:1071–1081
Sy A, Giraud E, Jourand P, García N, Willems A, de Lajudie P, Prin Y, Neyra M, Gillis M, Boivin-Masson C, Dreyfus B (2001) Methylotrophic Methylobacterium bacteria nodulate and fix nitrogen in symbiosis with legumes. J Bacteriol 183:214–220
Talbi C, Delgado MJ, Girard L, Ramírez-Trujillo A, Caballero-Mellado J, Bedmar EJ (2010) Burkholderia phymatum strains capable of nodulating Phaseolus vulgaris are present in Moroccan soils. Appl Environ Microbiol 76:4587–91
Tejera NA, Campos R, Sanjuan J, Lluch C (2004) Nitrogenase and antioxidant enzyme activities in Phaseolus vulgaris nodules formed by Rhizobium tropici isogenic strains with varying tolerance to salt stress. J Plant Physiol 161:329–338
Trujillo MJ, Willems A, Abril A, Planchuelo AM, Rivas R, Ludena R, Mateos PF, Martínez-Molina E (2005) Nodulation of Lupinus albus by strains of Ochrobactrum lupine sp. nov. Appl Environ Microbiol 71:1318–1327
Tu JC (1981) Effect of salinity on Rhizobium-root hair interaction, nodulation and growth of soybean. Can J Plant Sci 6:231–239
Tuteja N (2007) Mechanisms of high salinity tolerance in plants. Methods Enzymol 428:419–438
Valverde A, Velázquez E, Fernández-Santos F, Vizcaíno N, Rivas R, Mateos PF, Martínez-Molina E, Igual MJ, Willems A (2005) Phyllobacterium trifolii spp. nov. nodulating Trifolium lupini and Lupinus in Spanish soils. Int J Syst Evol Microbiol 55:1985–1989
Vandamme P, Coenye T (2004) Taxonomy of the genus Cupriavidus: a tale of lost and found. Int J Syst Evol Microbiol 54:2285–2289
Vandamme P, Goris J, Chen WM, de Vos P, Willems A (2002) Burkholderia tuberum sp. nov. and Burkholderia phymatum sp. nov. nodulate the roots of tropical legumes. Syst Appl Microbiol 25:507–512
Velázquez E, García-Fraile P, Ramírez-Bahena MH, Peix A, Rivas R (2010) Proteobacteria forming nitrogen fixing symbiosis with higher plants. In: Sezenna ML (ed) Proteobacteria: Phylogeny, metabolic diversity and ecological effects. Nova Science Publishers Inc. New York, USA, pp 37–56
Vincent JM (1970) A manual for the practical study of root-nodule bacteria IBP Handbook 15. Blackwell Scientific Publications, Oxford
Vinuesa P, Neumann-Silkow F, Pacios-Bras C, Spaink HP, Martínez-Romero E, Werner D (2003) Genetic analysis of a pH-regulated operon from Rhizobium tropici CIAT899 involved in acid tolerance and nodulation competitiveness. Mol Plant-Microbe Interact 16:159–168
Zahran HH (1999) Rhizobium-legume symbiosis and nitrogen fixation under severe conditions and in arid climates. Microbiol Mol Biol Rev 63:968–989
Zahran HH, Sprent JI (1986) Effects of sodium chloride and polyethyleneglycol on root hair infection and nodulation of Vicia faba L plants by Rhizobium leguminosarum. Planta 167:303–309
Zurayk R, Adian M, Baalbaki R, Saxena MC (1998) Interactive effects of salinity and biological nitrogen fixation on Chickpea (Cicer arietinum L) growth. J Agron Crop Sci 180:249–258
Acknowledgments
This work was supported by a European Regional Development Fund (ERDF)-cofinanced grant CVI-3177 from Junta de Andalucıa (Spain) and grant AGL2010-18607 from Ministerio de Economia y Competitividad (Spain). Grant 107PICO312 from Programa Iberoamericano de Ciencia y Tecnologıa para el Desarrollo (CYTED) and support from Junta de Andalucía to Group BIO-275 is also acknowledged.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Euan K. James.
Rights and permissions
About this article
Cite this article
Talbi, C., Argandoña, M., Salvador, M. et al. Burkholderia phymatum improves salt tolerance of symbiotic nitrogen fixation in Phaseolus vulgaris . Plant Soil 367, 673–685 (2013). https://doi.org/10.1007/s11104-012-1499-6
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-012-1499-6