Abstract
Aim
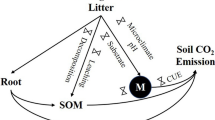
This study was aimed to assess the role that leaf litter play in nutrient cycling, nutrient soil availability and ecosystem processes in an oligotrophic tropical savanna.
Methods
A four year experiment was performed in a Neotropical savanna from the Brazilian plateau (cerrado), in which litter levels were modified, and the resulting changes in biophysical and chemical soil properties were studied. Changes in organic matter decomposition, soil respiration and stem growth of the six most common tree species were also monitored.
Results
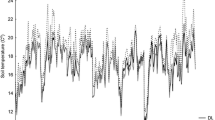
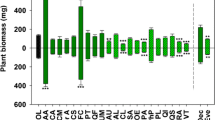
Compared to litter removal plots, double litter plots had lower maximum soil temperature and higher soil water content, and litter decomposition rates in one of three species studied, consistent with higher soil respiration rates observed in this treatment. With the exception of Ca, there were no significant differences in nutrients between the removal, natural and double litter plots, even though most nutrients tended to increase in the double litter plots by the end of the experimental period, while in the control plots nutrient levels remained relatively constant. Of the six tree species used for growth analysis, only one, Sclerolobium paniculatum, a fast growing species with shallow roots, had a significant increase in stem growth due to litter addition.
Conclusion
Preliminary results over four years indicate that litter removal and addition resulted in some significant changes and tendencies that indicate that litter is effectively altering ecosystem processes. The information obtained also suggest that nutrient cycling in plots with natural litter levels (control plots) was in a closed loop; most nutrients released by litter decomposition and mineralization were absorbed and reutilized immediately by the plants, thus minimizing nutrient leakage outside the system.








Similar content being viewed by others
References
Allen SE (1989) Chemical analysis of ecological materials, 2nd edn. Blackwell Scientific, Oxford
Anderson JPE, Domsch KH (1978) A physiological method for quantitative measurement of microbial biomass in soils. Soil Biol Biochem 29:1133–1142
Askew GP, Moffatt DJ, Montgome RF, Searl PL (1970) Interrelationships of soils and vegetation in savanna-forest boundary zone of north-eastern Mato Grosso. Geogr J 136:370–376
Benkobi L, Trlica MJ, Smith JL (1993) Soil loss as affected by different combinations of surface litter and rock. J Environ Qual 22(4):657–661
Bowden RD, Nadelhoffer KJ, Boone RD, Melillo JM, Garrison JB (1993) Contributions of aboveground litter, belowground litter, and root respiration to total soil respiration in a temperate mixed hardwood forest. Can J For Res 23:1402–1407
Bucci SJ, Scholz FG, Goldstein G, Meinzer FC, Franco AC, Campanello PI, Villalobos-Vega R, Bustamante M, Miralles-Wilhelm F (2006) Nutrient availability constrains the hydraulic architecture and water relations of savanna trees. Plant Cell Environ 29(12):2153–2167
Bucci SJ, Scholz FG, Goldstein G, Hoffmann WA, Meinzer FC, Franco AC, Giambelluca T, Miralles-Wilhelm F (2008) Controls on stand transpiration and soil water utilization along a tree density gradient in a Neotropical savanna. Agric For Meteorol 148(6–7):839–849
Cattelino PJ, Becker CA, Fuller LG (1986) Construction and installation of nomemade dendrometer bands. North J Appl For 3:73–75
Felfili JM, Junior MCD, Sevilha AC, Fagg CW, Walter BMT, Nogueira PE, Rezende AV (2004) Diversity, floristic and structural patterns of cerrado vegetation in central Brazil. Plant Ecol 175(1):37–46
Franco AA, Campello EFC, Dias LE, De Faria SM (1996) Uso de leguminosas associadas a microrganismos na revegetação de áreas de mineração de bauxita em Porto Trombetas-Pa. Serie Documentos(27), EMBRAPA-CNPAB, Itaguaí. 71pp
Franco AC, Bustamante M, Caldas LS, Goldstein G, Meinzer FC, Kozovits AR, Rundel P, Coradin VTR (2005) Leaf functional traits of Neotropical savanna trees in relation to seasonal water deficit. Trees Struct Funct 19(3):326–335
Furley PA (1999) The nature and diversity of Neotropical savanna vegetation with particular reference to the Brazilian cerrados. Glob Ecol Biogeogr 8:223–241
Furley PA, Ratter JA (1988) Soil resources and plant communities of the central Brazilian cerrado and their development. J Biogeogr 15:97–108
Garofalo CR (2001) Efeitos do aumento da disponibilidade de nutrients na dinámica de nutrientes en plantas herbáceas e solo em uma área de cerrado stricto sensu. Brazil. University of Brasilia. M.S. thesis
Gibbs PE, Leitao HD, Shepherd G (1983) Floristic composition and community structure in an area of cerrado in SE Brazil. Flora 173:433–449
Gill RW (1969) Soil microarthropod abundance following old-field litter manipulation. Ecology 50:805–816
Ginter DL, Mcleod KW, Sherrod C (1979) Water stress in longleaf pine induced by litter removal. For Ecol Manag 2:13–20
Goodland R, Pollard R (1973) Brazilian cerrado vegetation-fertility gradient. J Ecol 61(1):219–224
Haridasan M (1992) Observations on soils, foliar nutrient concentrations, and floristic composition of cerrado and cerradão communities in central Brazil. In: Proctor J, Ratter JA, Furley PA (eds) The nature and dynamics of forest-savanna boundaries. Chapman and Hall, London, pp 171–184
Haridasan M (2000) Nutrição mineral de plantas nativas do cerrado. Braz J Plant Physiol 12:54–64
Haridasan M (2001) Nutrient cycling as a function of landscape and biotic characteristics in the cerrado of central Brazil. In: McClain ME, Victoria RL, Richey JE (eds) Biogeochemistry of the Amazon basin and its role in a changing world. Oxford University Press, New York, pp 68–83
Hoffmann WA, Silva E, Machado G, Bucci SJ, Scholz FG, Goldstein G, Meinzer FC (2005) Seasonal leaf dynamics across a tree density gradient in a Brazilian savanna. Oecologia 145:307–316
IBGE (1995) Zoneamento ambiental do Córrego Taquara-DF. Volumes I a IV. IBGE, Goiânia
Jackson PC, Meinzer FC, Bustamante M, Goldstein G, Franco A, Rundel PW, Caldas L, Igler E, Causin F (1999) Partitioning of soil water among tree species in a Brazilian cerrado ecosystem. Tree Physiol 19(11):717–724
Kozovits AR, Bustamante MMC, Garofalo CR, Bucci SJ, Franco AC, Goldstein G, Meinzer FC (2007) Nutrient resorption and patterns of litter production and decomposition in a Neotropical savanna. Funct Ecol 21(6):1034–1043
Lenza E, Klink CA (2006) Comportamento fenologico de especies lenhosas em um cerrado sentido restrito de Brasilia, DF. Revista Brasileira de Botanica 29:627–638
Li Y, Xu M, Sun OJ, Cui W (2004) Effects of root and litter exclusion on soil CO2 efflux and microbial biomass in wet tropical forests. Soil Biol Biochem 36:2111–2114
Lunt HA (1951) Liming and twenty years of litter raking and burning under red (and white) pine. Soil Sci Soc Am Proc 15:381–390
Nadelhoffer KJ, Colman BP, Currie WS, Magill A, Aber JD (2004) Decadal-scale fates of N-15 tracers added to oak and pine stands under ambient and elevated N inputs at the Harvard Forest (USA). For Ecol Manag 196(1):89–107
Nilsson MC, Wardle DA, Dahlberg A (1999) Effects of plant litter species composition and diversity on the boreal forest plant-soil system. Oikos 86:16–26
Peng SL, Ren H, Wu JG, Lu HF (2003) Effects of litter removal on plant species diversity: a case study in tropical eucalyptus forest ecosystems in South China. J Environ Sci 15:367–371
Pinheiro JC, Bates DM (2000) Mixed effects models in S and S-Plus. Springer, New York, 528pp
Pires IP, Marcati CR (2005) Anatomia e uso da madeira de duas variedades de Sclerolobium paniculatum Vog. do sul do Maranhão, Brasil. Acta Botanica Brasilica 19(4):669–678
Ponge JF, Arpin P, Vannier G (1993) Collembolan response to experimental perturbations of litter supply in a temperate forest ecosystem. Eur J Soil Biol 29:141–153
Poser T (1990) The influence of litter manipulation on the centipedes of a beech wood. In: Minelli A (ed) Proceedings of the seventh international congress on Myriapods. Brill, Leiden, pp 235–245
Quintanilha de Albuquerque E, Dias LE (1999) Decomposição de folhas de Acacia mangium e Sclerobium paniculatum (tachi-branco) em colunas de solo. Revista Árvore 23:15–22
R Development Core Team (2008) R: A language and environment for statistical computing. R Foundation for Statistical Computing, Vienna, Austria. ISBN 3-900051-07-0, URL http://www.R-project.org
Ribeiro JF (1983) Comparação da concentração de nutrientes da vegetação arbórea e nos solos de um cerrado e um cerradão no Distrito Federal, Brasil. MSc Thesis, University of Brasília, Brazil
Ryan MG, Law BE (2005) Interpreting, measuring, and modeling soil respiration. Biogeochemistry 73(1):3–27
Sayer EJ (2006) Using experimental manipulation to assess the roles of leaf litter in the functioning of forest ecosystems. Biol Rev 81(1):1–31
Sayer EJ, Tanner EVJ, Cheesman AW (2006) Increased litterfall changes fine root distribution in a moist tropical forest. Plant Soil 281(1–2):5–13
Scholz FG, Bucci SJ, Goldstein G, Meinzer FC, Franco AC (2002) Hydraulic redistribution of soil water by Neotropical savanna trees. Tree Physiol 22:603–612
Valenti MW, Cianciaruso MV, Batalha MA (2008) Seasonality of litterfall and leaf decomposition in a cerrado site. Braz J Biol 68(3):459–465
Wang WJ, Zu YG, Wang HM, Hirano T, Takagi K, Sasa K, Koike T (2005) Effect of collar insertion on soil respiration in a larch forest measured with a LI-6400 soil CO2 flux system. J For Res 10(1):57–60
Acknowledgments
We thank the Reserva Ecologica do IBGE for logistic support. We are very grateful to Catarina S. Cartaxo, Ana Salazar Parra, Sybil G. Gotsch, Cristiane Ferreira and Lucas Silva for invaluable assistance with fieldwork. Heloisa S. Miranda and Vania R. Pivello provided excellent comments on the manuscript. This research was supported by NSF Biocomplexity Grant EAR 0322051 and CNPQ, Brazil.
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Harry Olde Venterink.
Rights and permissions
About this article
Cite this article
Villalobos-Vega, R., Goldstein, G., Haridasan, M. et al. Leaf litter manipulations alter soil physicochemical properties and tree growth in a Neotropical savanna. Plant Soil 346, 385–397 (2011). https://doi.org/10.1007/s11104-011-0860-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-011-0860-5