Abstract
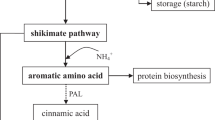
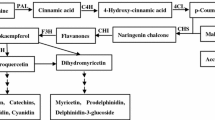
K limitation could decrease the flavonoids content in many Chinese traditional herbs. These results may be caused by the influence of K deficiency on the synthesis pathway of flavonoids. In this paper, we aim to study the influence of K deficiency on secondary metabolites and activities of phenylalanine ammonia lyase (PAL), 4-coumarate coenzyme A ligase (4CL) and cinnamate 4-hydroxylase (C4H) enzymes in the process of flavonoid synthesis in Chrysanthemum morifolium Ramat. The results show that K deficiency decreased the flavonoid and chlorogenic acid contents slightly in flower of C. morifolium while the same effect was obtained on C4H and PAL. Total flavonoids content increased with the content of cinnamic acid and p-coumaric acid in the plant under K deficiency, respectively. The regression equations between content of flavonoids and cinnamic acid/ p-coumaric acid should be expressed in term of linear effects (R2 = 0.9809, P < 0.001 for cinnamic acid and R2 = 0.9929, P < 0.0001 for p-coumaric acid, respectively). But the effect of phenylalanine on flavonoids should be expressed in term of quadratic pattern (R2 = 0.9375, P < 0.05). There were two kinds principal components from chorismate to coumaryl CoA synthesis process, principal 1 was the substrate (phenylalanine, cinnamic acid, p-coumaric acid and PAL) and principal 2 was the enzyme (4CL, C4H), the principal 1 was the domination principal in both K deficient (88.36% and 10.57%) and sufficient treatments (88.17% and 9.64%), however, the factor loading (correlation coefficient of measured targets and the corresponding principal component) in principal 1 was opposite under different K application.





Similar content being viewed by others
References
Amtmann A, Blatt MR (2009) Regulation of macronutrient transport. New Phytol 181:35–52
Andrist-Rangel Y, Edwards AC, Hillier S, Oborn I (2007) Long-term K dynamics in organic and conventional mixed cropping systems as related to management and soil properties. Agric Ecosyst Environ 122:413–426
Armengaud P, Sulpice R, Miller AJ, Stitt M, Amtmann A, Gibon Y (2009) Multilevel analysis of primary metabolism provides new insights into the role of Potassium nutrition for Glycolysis and Nitrogen assimilation in Arabidopsis roots. Plant Physiol 150:772–785
Aviram M, Fuhrman B (1998) Polyphenolic flavonoids inhibit macrophage-mediated oxidation of LDL and attenuate atherogenesis. Atherosclerosis 137:45–50
Awad MA, Jager A (2002) Relationship between fruit nutrients and concentrations of flavonoids and chlorogenic acid in ‘Elstar’ apple skin. Sci Hortic 92:265–276
Bednarz CW, Oosterhuis DM (1999) Physiological changes associated with potassium deficiency in cotton. J Plant Nutr 22:303–313
Bi HH, Zeng RS, Su LM, An M, Luo SM (2007) Rice allelopathy induced by methyl jasmonate and methyl salicylate. J Chem Ecol 33:1089–1103
Cai GM, Fu QW, Qin BS (2002) The analysis of the influences of chemical fertilizer on the growth of young Ginkgo tree and its leaf flavone (in Chinese). Journal of Guangxi Agriculture (Supplement): 83–87
Cakmak I, Hengeler C, Marschner H (1994a) Prtitioning of shoot and root dry matter and carbohydrates in bean plants suffering from phosphorus, potassium and magnesium deficiency. J Exp Bot 45:1245–1250
Cakmak I, Hengeler C, Marschner H (1994b) Changes in phloem export of sucrose in leaves in response to phosphorus, potassium and magnesium deficiency in bean plants. J Exp Bot 45:1251–1257
Duarte J, VizcaÌno FP, Utrilla P, Jiménez J, Tamargo J, Zarzuelo A (1993) Vasodilatatory effects of flavonoids in rat aortic smooth muscle. Structure-activity relationship. Gen Pharmacol 24:857–862
Duh PD (1999) Antioxiodant activity of water extract of four Harng Jyur (Chrysanthemum morifolium Ramat.) varieties in soybean oil emulsion. Food Chem 66:471–476
Duh PD, Tu YY, Yen GC (1999) Antioxidant activity of water extract of four Harng Jyur (Chrysanthemum morifolium Ramat). Lebensm-Wissu-Technol 32:269–277
Estiarte M, Filella I, Serra J, Penuelas J (1994) Effects of nutrient and water stress on leaf phenolic content of peppers and susceptibility to generalist herbivore Helicove armigra (Hubner). Oecologia 99:387–391
Farley RF, Draycott PA (1975) Growth and yield of sugar beet in relation to potassium and sodium supply. J Sci Food Agric 26:385–392
Fitzpatrick DF, Hirschfield L, Coffey RG (1993) Endothelium-dependent vasorelaxing activity of wine and other grape products. Am J Physiol 265:774–778
Gremigni P, Hamblin J, Harris D (1997) Alkaloid level in lupin seed is affected by nutritional stresses. Proceedings of the International Food Legume Research Conference III, September 1997. Adelaide, SA. p 143
Gremigni P, Gazey C, Hamblin J, Harris D (1998) Soil nutritional status affects alkaloid levels in Australian sweet lupin. Proceeding of the 3rd European Conference on Grain Legumes, November. AEP, Paris, France. p 179
Gremigni P, Hamblin J, Harris D (1999) Genotype × environment interactions and lupin alkaloids. (eds) Van Santen E, Wink M, Weissman S, Romer P. pp 362–365
Helal H, Mengel K (1979) Nitrogen metabolism of young barley plants as affected by NaCl-salinity and potassium. Plant Soil 51:457–462
Herrera MD, Zarzuelo A, Jimenez J, Marhuenda E, Duarte J (1996) Effects of flavonoids on rat aortic smooth muscle contractility: structure-activity relationships. Gen Pharmacol 27:273–277
Hertog MG, Feskens EJ, Hollman PC, Katan MB, Kromhout D (1993a) Dietary antioxidant flavonoids and risk of coronary heart disease: the Zutphen Elderly Study. Lancet 342:1007–1011
Hertog MGL, Holman PCH, Katan MB, Kromhout D (1993b) Intake of potentially anticarcinogenic flavonoids and their determinants in adults in The Netherlands. Nutr Cancer 20:21–29
Huber SC (1984) Biochemical basis for effects of K-deficiency on assimilate export rate and accumulation of soluble sugars in soybean leaves. Plant Physiol 76:424–430
Kangasjarvi J, Talvinen J, Utriainen M, Karjalainen R (1994) Plant defence systems induced by ozone. Plant Cell Environ 17:783–794
Kayser M, Isselstein J (2005) Potassium cycling and losses in grassland systems: a review. Grass Forage Sci 60:213–224
Keinänen M, Julkunen-Tiitto R (1998) High-performance liquid chromatographic determination of flavonoids in Betula pendula and Betula pubescens leaves. J Chromatogr A 793:370–377
Kneckt P, Jarvinen R, Reunanen A, Maatela J (1996) Flavonoid intake and coronary mortality in Finland: a cohort study. Br Med J 312:478–481
Knoblock KH, Hahlbrock K (1975) Isoenzymes of p-coumarate: CoA ligase from cell suspension cultures of Glycine max. Eur J Biochem 52:311–320
Koch K, Mengel K (1974) The influence of the level of potassium supply to young tobacco plants (Nicotiana tabacum L.) on short-term uptake and utilisation of nitrate nitrogen (15N). J Sci Food Agric 25:465–471
Koukol J, Conn EE (1961) The metabolism of aromatic compounds in higher plants. Purification and properties of the L-phenylalanine deaminase of Hordeum vulagare. J Biol Chem 236:2692–2698
Krause J, Reznik H (1976) Investigation of flavonol accumulation in Fagopyrum esculentum Moench as influenced by P-and N deficiency. Z Pflanzenphysiol 70:392–400
Lamb C, Rubery PH (1975) A spectrophotometric assay for trans-cinnamic acid 4-hydroxylase activity. Anal Biochem 68:554–561
Lancaster JE (1992) Regulation of skin color in apples. Crit Rev Plant Sci 10:487–502
Lawanson AO, Akindole BB, Fasalojo PB, Akpe BL (1972) Time course of anthocyanin formation during deficiencys of nitrogen, phosphorus and potassium in seedling of Zea mays Linn. Var.E.S. l. Z Pflanzenphysiol 66:251–253
Lazarowych NJ, Pekos P (1998) Use of fingerprinting and marker compounds for identification and standardization of botanical drugs: strategies for applying pharmaceutical HPLC analysis to herbal products. Drug Inf J 32:497–512
Li R, Volenec JJ, Joern BC, Cunningham SM (1997) Potassium and nitrogen effects on carbohydrate and protein metabolism in alfalfa roots. J Plant Nutr 20:511–529
Li Z, Chen ZM, Liao LS, Lin SS (1999) Determination of chlorogenic acid in flower of Chrysanthemum morifolium Ramat. China Journal of Chinese Materia Media 27:329–330
Li QY, Si MZ, Rao GX, Li YZ (2009) Correlation between total flavonoid content and IR spectrum of Erigenon Breviscapus produced in different planting locations of Yunnan. The Journal of Light Scattering 21:73–76
Lindhauer MG (1989) The role of K+ in cell extension, growth and storge of assimilates. In: Methods of K-research in plants (ed) By International Potash Institute, Bern, Switzerland, 21st Colloquium of the Int. Potash Inst. Louvainla-Neuve, Belgium, pp 161–187
Liu JQ, Shen QQ, Liu JS, Wu DL, Wang JT (2001) Studies on the chemical constituents from Chrysanthemum morifolium Ramat. China Journal of Chinese Materia Media 26:547–548
Liu DH, Zhu DW, Zhou WB, Liu W, Chen KL (2006) Effect of nitrogen, phosphorus, and potassium on yield and quality of Futian’s Chrysanthemum morifolium. Chinese Traditional and Herbal Drugs 37:125–129
Liu DH, Yang TW, Zhu DW, Zhou WB, Geng MJ, Liu W (2007) Effect of potassium application at various rates on yield and quality of flower in Chrysanthemum morifolium from Futianhe region. Chinese Traditional and Herbal Drugs 38:120–124
Liu DH, Guo LP, Zhu DW, Liu W, Jin H (2009a) Characteristics of accumulation and distribution of nitrogen, phosphorus, potassium, calcium and magnesium in Chrysanthemum morifolium. China Journal of Chinese Materia Medica 34:2444–2448
Liu DH, Guo LP, Zhu DW, Huang LQ (2009b) Effect of fertilization and plastic film mulching on yield and quality of Chrysanthemum morifolium flower. Chinese Traditional and Herbal Drugs 40:788–792
Mayer AM, Harel E (1979) Polyphenoloxidase in plants. Phytochemistry 18:193–215
Moody PW, Bell MJ (2006) Availability of soil potassium and diagnostic soil tests. Aust J Soil Res 44:265–275
Pettigrew WT (1999) Potassium deficiency increases specific leaf weights and leaf glucose levels in field-grown cotton. Agron J 91:962–968
Rice-Evans AC, Miller NJ, Paganga G (1997) Antioxidant properties of phenolic compounds. New Trend Plant Sci Rev 2:152–159
Sorger GJ, Ford RE, Evans HJ (1965) Effects of univalent cations on the immunoelectrophoretic behavior of pyruvic kinase. Proc Nctl Acad Sci USA 54:1614–1621
Tan SC (1980) Phenylalanine ammonia-lyase and the phenylalanine ammonia-lyase inactivating system: effects of light, temperature and mineral deficiencies. Aust J Plant Physiol 7:159–167
Tony VYNJ, Yin XH, Tom WB, Chung-Ja CJ, Istvan R, Sylvie MB (2002) Potassium fertilization effects on isoflavone concentrations in soybean. J Agric Food Chem 50:3501–3506
Ward GM (1960) Potassium in plant metabolism. III. Some carbohydrate changes in the wheat seedling associated with varying rates of potassium supply. Can J Plant Sci 40:729–735
Yang J, Jiang HD, Ge Z, Ye ZJ (2003) Variation of the contents of chlorogenic acid and other components in chrysanthemum morifolium Ramat. collected at different time. Chin Pharm J 38:833–836
Zhang AL, Ma Q, Gao JM, Zhang KJ, Wang L (2001) Studies on bioactivities of chlorogenic acid and its analogues. Chinese Traditional and Herbal Drugs 32:173–176
Zhao L, Xu R, Wan DR (2009) Identification of Herba Sedi collected in the different habitats and periods by FT-IR. J Tradit Chin Med 24:361–364
Acknowledgements
The authors would like to thank all the researchers in Laboratory of Plant Nutrition and Ecological Environment Research. Many people have made invaluable contributions, both directly and indirectly to our research. The work was supported by the Ministry of Science and Technology of Hubei Province, China (Project 2001AA304A).
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: F.J.M. Maathuis.
Rights and permissions
About this article
Cite this article
Liu, W., Zhu, D., Liu, D. et al. Comparative metabolic activity related to flavonoid synthesis in leaves and flowers of Chrysanthemum morifolium in response to K deficiency. Plant Soil 335, 325–337 (2010). https://doi.org/10.1007/s11104-010-0421-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-010-0421-3