Abstract
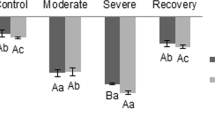
Despite the evidence for a critical role of Mn in malate decarboxylation and CO2 release for carbon fixation reactions in C-4 plants, there is a lack of information on their Mn requirement. The objective of this study was to establish Mn levels needed for optimum growth and photosynthesis of four agriculturally important C-4 species, NAD-ME C-4 pearl millet and purple amaranth, and NADP-ME C-4 corn and sorghum, as compared to two C-3 species, wheat and squash. Plants were grown hydroponically in a complete nutrient solution with Mn concentrations ranging from 0 to 100 μM. We report that under these conditions, C-3 and NADP-ME C-4 plants reached their maximum biomass production with 2–5 μM Mn, the concentration commonly used in plant nutrient media. In contrast, Mn concentrations supporting maximum performance of NAD-ME C-4 plants were up to 20-fold higher and ranged between 50 and 100 μM. Although leaf tissue Mn concentrations increased in parallel with Mn nutrition in all plants, the higher leaf Mn had no effect on NADP-ME C-4 or C-3 plants, but it caused a large, up to 100%, increase in net photosynthetic rate in NAD-ME C-4 species. The highest photosynthetic rates across the spectrum of photon flux density were recorded for C-3 and NADP-ME C-4 plants receiving 2–5 μM Mn, and for NAD-ME C-4 species millet and amaranth supplied with 50 or 100 μM Mn, respectively. Squash (C-3) plants were the most sensitive to Mn and their photosynthetic rate was severely depressed with more than 10 μM Mn. The increase in photosynthetic rates of NAD-ME C-4 species occurred without an increase in stomatal conductance, eliminating CO2 uptake as the main cause. We propose that the higher photosynthetic rates in NAD-ME C-4 species supplied with higher Mn were a result of increased activation of the Mn-dependent NAD-ME in bundle sheath cells, producing greater CO2 supply for Calvin cycle reactions. This is, to our knowledge, the first report on a significantly higher Mn requirement for optimum photosynthesis and biomass production of NAD-ME C-4 species.




Similar content being viewed by others
References
Anderson I, Evans HJ (1956) Effect of manganese and certain metal cations on isocitric dehydrogenase and malic enzyme activities in Phaseolus vulgaris. Plant Physiol 31:22–28
Burnell JN (1986) Purification and properties of phosphoenolpyruvate carboxy kinase from C4 plants. Aust J Plant Physiol 13:577–587
Cheng L, Fuchigami LH, Breen PJ (2001) The relationship between photosystem II efficiency and quantum yield for CO2 assimilation is not affected by nitrogen content in apple leaves. J Exp Bot 52:1865–1872 doi:10.1093/jexbot/52.362.1865
Dengler N, Donnelly PM, Dengler RE (1996) Differentiation of bundle sheath, mesophyll and distinctive cells in the C4 grass Arundinella hirta (Poacea). Am J Bot 83:1391–1405 doi:10.2307/2446094
Dever LV, Pearson M, Ireland RJ, Leegood RC, Lea PJ (1998) The isolation and characterisation of a mutant of the C4 plant, Amaranthus edulis deficient in NAD-malic enzyme activity. Planta 206:649–656 doi:10.1007/s004250050443
Ehleringer JR, Monson RK (1993) Evolutionary and ecological aspects of photosynthetic pathway variation. Annu Rev Ecol Syst 24:411–439 doi:10.1146/annurev.es.24.110193.002211
Epstein E, Bloom AJ (eds) (2005) In: Mineral nutrition of plants. Principles and perspectives. Sinauer, Sunderland, p 31
Hatch MD, Kagawa T (1974) Activity, location and role of NAD malic enzyme in leaves with C4-pathway photosynthesis. Aust J Plant Physiol 1:357–369
Hatch MD, Carnal NW (1992) The role of mitochondria in C4 photosynthesis. In: Lambers H, van der Plas LHW (eds) Molecular, biochemical and physiological aspects of plant respiration. SPB Academic, Hague, pp 135–148
Hatch MD, Kagawa T, Graig S (1975) Subdivision of C4-pathway species based on differing C4 acid decarboxylating systems and ultrastructural features. Aust J Plant Physiol 2:111–128
Long SP (1999) Environmental response. In: Sage RF, Monson RK (eds) C4 biology. Academic, San Diego, pp 215–249
Marshner H (ed) (1995) In: Mineral nutrition of higher plants. Academic, New York, pp 324–333
Maynard LS, Cotzias AC (1955) The partition of manganese among organs and intracellular organelles of the rat. J Biol Chem 214:489–495
Reinbott TM, Blevins DG (1999) Phosphorus nutritional effects on root hydraulic conductance, xylem water flow and flux of magnesium and calcium in squash. Plant Soil 209:263–273 doi:10.1023/A:1004646732225
Rustin P, Lance C (1989) The divalent cation pump and its role in the regulation of malic enzyme activity in purified mitochondria from potato tuber. Plant Cell Physiol 30:505–512
SAS Institute (2004) The SAS system, version 9.1. SAS Institute Inc., Cary
Underwood EJ (1977) Trace elements in human and animal nutrition. Academic, New York, pp 170–195
Zhi-Hui C, Walker RP, Acheson RM, Leegood RC (2002) Phosphoenolpyruvate carboxy kinase assayed at physiological concentrations of metal ions has a high affinity for CO2. Plant Physiol 128:160–164 doi:10.1104/pp.128.1.160
Author information
Authors and Affiliations
Corresponding author
Additional information
Responsible Editor: Richard W. Bell.
Rights and permissions
About this article
Cite this article
Kering, M.K., Lukaszewska, K. & Blevins, D.G. Manganese requirement for optimum photosynthesis and growth in NAD-malic enzyme C-4 species. Plant Soil 316, 217–226 (2009). https://doi.org/10.1007/s11104-008-9772-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11104-008-9772-4