Abstract
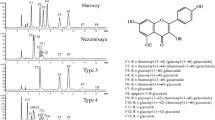
There are substantial genotypic differences in the levels of flavonol glycosides (FGs) in soybean leaves. The first objective of this study was to identify and locate genes responsible for FG biosynthesis in the soybean genome. The second objective was to clone and verify the function of these candidate genes. Recombinant inbred lines (RILs) were developed by crossing the Kitakomachi and Koganejiro cultivars. The FGs were separated by high performance liquid chromatography (HPLC) and identified. The FGs of Koganejiro had rhamnose at the 6″-position of the glucose or galactose bound to the 3-position of kaempferol, whereas FGs of Kitakomachi were devoid of rhamnose. Among the 94 RILs, 53 RILs had HPLC peaks classified as Koganejiro type, and 41 RILs had peaks classified as Kitakomachi type. The segregation fitted a 1:1 ratio, suggesting that a single gene controls FG composition. SSR analysis, linkage mapping and genome database survey revealed a candidate gene in the molecular linkage group O (chromosome 10). The coding region of the gene from Koganejiro, designated as GmF3G6″Rt-a, is 1,392 bp long and encodes 464 amino acids, whereas the gene of Kitakomachi, GmF3G6″Rt-b, has a two-base deletion resulting in a truncated polypeptide consisting of 314 amino acids. The recombinant GmF3G6″Rt-a protein converted kaempferol 3-O-glucoside to kaempferol 3-O-rutinoside and utilized 3-O-glucosylated/galactosylated flavonols and UDP-rhamnose as substrates. GmF3G6″Rt-b protein had no activity. These results indicate that GmF3G6″Rt encodes a flavonol 3-O-glucoside (1 → 6) rhamnosyltransferase and it probably corresponds to the Fg2 gene. GmF3G6″Rt was designated as UGT79A6 by the UGT Nomenclature Committee.







Similar content being viewed by others
References
Altschul SF, Madden TL, Schaffer AA, Zhang J, Zhang Z, Miller W, Lipman DJ (1997) Gapped BLAST and PSI-BLAST: a new generation of protein database search programs. Nucleic Acids Res 25:3389–3402
Buttery BR, Buzzell RI (1973) Varietal differences in leaf flavonoids of soybeans. Crop Sci 13:103–106
Buttery BR, Buzzell RI (1975) Soybean flavonol glycosides: identification and biochemical genetics. Can J Bot 53:219–224
Buttery BR, Buzzell RI (1976) Flavonol glycoside genes and photosynthesis in soybean. Crop Sci 16:547–550
Buzzell RI (1974) Soybean linkage tests. Soybean Genet Newsl 1:11–14
Buzzell RI, Buttery BR (1974) Flavonol glycoside genes in soybeans. Can J Genet Cytol 16:897–899
Buzzell RI, Buttery BR (1977) Inheritance and linkage of a magenta flower gene in soybeans. Can J Genet Cytol 19:749–751
Buzzell RI, Buttery BR (1992) Inheritance of an anomalous flavonol glycoside gene in soybean. Genome 35:636–638
Buzzell RI, Buttery BR (1998) Genetics of leaf waviness in soybean. Soybean Genet Newsl 25:23–24
Chang YF, Imam JS, Wilkinson MF (2007) The nonsense-mediated decay RNA surveillance pathway. Ann Rev Biochem 76:51–74
Cregan PB, Jarvic T, Bush AL, Shoemaker RC, Lark KG, Kahler AL, Kaya N, VanToai TT, Lohnes DG, Chung J, Specht JE (1999) An integrated genetic map of the soybean genome. Crop Sci 39:1464–1490
Fehr WR, Caviness CE, Burmood DT, Pennington JS (1971) Stage of development descriptions for soybeans, Glucine max (L.) Merrill. Crop Sci 11:929–931
Frydman A, Liberman R, Huhman DV, Carnmeli-Weissberg M, Sapir-Mir M, Ophir R, Sumner LW, Eyal Y (2013) The molecular and enzymatic basis of bitter/non-biter flavor of citrus fruit: evolution of branch-forming rhamnosyltransferases under domestication. Plant J 73:166–178
Githiri SM, Yang D, Khan NA, Xu D, Komatsuda T, Takahashi R (2007) QTL analysis of low temperature-induced browning in soybean seed coats. J Hered 98:360–366
Hisano H, Sato S, Isobe S, Sasamoto S, Wada T, Matsuno A, Fujishiro T, Yamada M, Nakayama S, Nakamura Y, Watanabe S, Harada K, Tabata S (2007) Characterization of the soybean genome using EST-derived microsatellite markers. DNA Res 14:271–281
Iwashina T, Benitez ER, Takahashi R (2006a) Analysis of flavonoids in pubescence of soybean near-isogenic lines for pubescence color loci. J Hered 97:438–443
Iwashina T, Kitajima J, Matsumoto S (2006b) Flavonoids in the species of Cyrtomium (Dryopteridaceae) and related genera. Biochem Syst Ecol 34:14–24
Iwashina T, Githiri SM, Benitez ER, Takemura T, Kitajima J, Takahashi R (2007) Analysis of flavonoids in flower petals of soybean near-isogenic lines for flower and pubescence color genes. J Hered 98:250–257
Iwashina T, Oyoo ME, Khan NA, Matsumura H, Takahashi R (2008) Analysis of flavonoids in flower petals of soybean flower color variants. Crop Sci 48:1918–1924
Iwashina T, Yamaguchi M, Nakayama M, Onozaki T, Yoshida H, Kawanobu S, Ono H, Okamura M (2010) Kaempferol glycosides in the flowers of carnation and their contribution to the creamy white flower color. Nat Prod Commun 5:1903–1906
Iwata H, Ninomiya S (2006) AntMap: constructing genetic linkage maps using an ant colony optimization algorithm. Breed Sci 56:371–377
Kroon J, Souer E, de Graaff A, Xue Y, Mol J, Koes R (1994) Cloning and structural analysis of the anthocyanin pigmentation locus Rt of Petunia hybrida: characterization of the insertion sequences in two mutant alleles. Plant J 5:69–80
Lairson LL, Henrissat B, Davies GJ, Withers SG (2008) Glycosyltransferases: structures, functions, and mechanisms. Annu Rev Biochem 77:521–555
Mackenzie PI, Owens IS, Burchell B, Bock KW, Bairoch A, Belanger A, Fournel-Gigleux S, Green M, Hum DW, Iyanagi T, Lancet D, Louisot P, Magdalou J, Chowdhury JR, Ritter JK, Schachter H, Tephly TR, Tipton KF, Nebert DW (1997) The UDP glycosyltransferase gene superfamily: recommended nomenclature update based on evolutionary divergence. Pharmacogenetics 7:255–269
Masada S, Terasawa K, Mizukami H (2007) A single amino acid in the PSPG-box plays an important role in the catalytic function of CaUGT2 (Curcumin glucosyltransferase), a Group D Family 1 glucosyltransferase from Catharanthus roseus. FEBS Lett 581:2605–2610
Murai Y, Takahashi R, Rojas Rodas F, Kitajima J, Iwashina T (2013) New flavonol triglycosides from the leaves of soybean cultivars. Nat Prod Commun 8:453–456
Murray MG, Thompson WF (1980) Rapid isolation of high molecular weight plant DNA. Nucleic Acids Res 8:4321–4325
Page RDM (1996) TreeView: an application to display phylogenetic trees on personal computers. Comput Appl Biosci 12:357–358
Paquette S, Møller BL, Bak S (2003) On the origin of family 1 plant glycosyltransferases. Phytochemistry 62:399–413
Rice-Evans CA, Miller NJ, Bolwell PG, Bramley PM, Pridham JB (1995) The relative antioxidant activities of plant-derived polyphenolic flavonoids. Free Radic Res 22:375–383
Rode MW, Bernard RL (1975) Inheritance of wavy leaf. Soybean Genet Newsl 2:42–44
Severin AJ, Woody JL, Bolon YT, Joseph B, Diers BW, Farmer AD, Muehlbauer GJ, Nelson RT, Grant D, Specht JE, Graham MA, Cannon SB, May GD, Vance CP, Shoemaker RC (2010) RNA-Seq Atlas of Glycine max: a guide to the soybean transcriptome. BMC Plant Biol 10:160
Shah DM, Hightower RC, Meagher RB (1983) Genes encoding actin in higher plants: intron positions are highly conserved but the coding sequences are not. J Mol Appl Genet 2:111–126
Song QJ, Marek LF, Shoemaker RC, Lark KG, Concibido VC, Delannay X, Specht JE, Cregan PB (2004) A new integrated genetic linkage map of the soybean. Theor Appl Genet 109:122–128
Sunada K, Ito T (1982) Soybean grain quality as affected by low temperature treatments in plants (color of hilum, seed coat cracking) (in Japanese). Rep Hokkaido Branch, Crop Sci Soc Jpn and Hokkaido Branch. Jpn Soc Breed 22:34
Takahashi R (1997) Association of soybean genes I and T with low-temperature induced seed coat deterioration. Crop Sci 37:1755–1759
Takahashi R, Asanuma S (1996) Association of T gene with chilling tolerance in soybean. Crop Sci 36:559–562
Takahashi R, Benitez ER, Funatsuki H, Ohnishi S (2005) Soybean maturity and pubescence color genes improve chilling tolerance. Crop Sci 45:1387–1393
Takahashi R, Githiri SM, Hatayama K, Dubouzet EG, Shimada N, Aoki T, Ayabe S, Toda K, Matsumura H (2007) A single-base deletion in soybean flavonol synthase gene is associated with magenta flower color. Plant Mol Biol 63:125–135
Tamura K, Peterson D, Peterson N, Stecher G, Nei M, Kumar S (2011) MEGA5: molecular evolutionary genetics analysis using maximum likelihood, evolutionary distance, and maximum parsimony method. Mol Biol Evol 28:2731–2739
Toda K, Yang D, Yamanaka N, Watanabe S, Harada K, Takahashi R (2002) A single-base deletion in soybean flavonoid 3′-hydroxylase gene is associated with gray pubescence color. Plant Mol Biol 50:187–196
Toda K, Takahashi R, Iwashina T, Hajika M (2011) Difference in chilling-induced flavonoid profiles, antioxidant activity and chilling tolerance between soybean near-isogenic lines for pubescence color gene. J Plant Res 124:173–182
Toda K, Kuroiwa H, Senthil K, Shimada N, Aoki T, Ayabe S, Shimada S, Sakuta M, Miyazaki Y, Takahashi R (2012) The soybean F3′H protein is localized to the tonoplast in the seed coat hilum. Planta 236:79–89
Vinson JA, Dabbagh YA, Serry MM, Jang JH (1995) Plant flavonoids, especially tea flavones, are powerful antioxidants using an in vitro oxidation model for heart disease. J Agric Food Chem 43:2800–2802
Yonekura-Sakakibara K, Hanada K (2011) An evolutionary view of functional diversity in family 1 glycosyltransferases. Plant J 66:182–193
Yonekura-Sakakibara K, Tohge T, Niida R, Saito K (2007) Identification of a flavonol 7-O-rhamnosyltransferase gene determining flavonoid pattern in Arabidopsis by transcriptome coexpression analysis and reverse genetics. J Biol Chem 282:14932–14941
Acknowledgments
The authors are grateful to Dr. Joseph G. Dubouzet (Scion Research, New Zealand) for critical reading of the manuscript, and to Mr. Tetsuya Mori (RIKEN CSRS, Japan) for his technical support. This study was partially supported by the Japanese Government (MEXT) Scholarship (to F.R.R. and T.O.R) and the JSPS KAKENHI Grant Number 25440148 (to K.Y.-S.).
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
11103_2013_133_MOESM1_ESM.eps
Multiple alignment of flavonoid glycoside glycosyltransferases (GGTs). The PSPG motif is within the box. Amino acid residues conserved in flavonoid GGTs are highlighted in black. Amino acid residues conserved in flavonoid G6″GTs and G2″GTs are highlighted in red and blue, respectively. (EPS 1059 kb)
Rights and permissions
About this article
Cite this article
Rojas Rodas, F., Rodriguez, T.O., Murai, Y. et al. Linkage mapping, molecular cloning and functional analysis of soybean gene Fg2 encoding flavonol 3-O-glucoside (1 → 6) rhamnosyltransferase. Plant Mol Biol 84, 287–300 (2014). https://doi.org/10.1007/s11103-013-0133-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11103-013-0133-1