ABSTRACT
Purpose
To identify a LNCaP-specific peptide using a phage display library and evaluate its potential applications in targeted drug delivery.
Methods
Binding abilities of selected phages were evaluated by cell phage ELISA. The KYL peptide encoded by the most specific phage clone was synthesized, labeled with fluorescein, and assayed in various cell lines. A fusion peptide composed of the KYL peptide and a proapoptotic peptide D (KLAKLAK)2 was synthesized, and the cell death effect was evaluated on different cells. Moreover, the KYL peptide was conjugated to a cationic protein, protamine, to explore its potential application in siRNA delivery.
Results
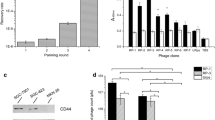
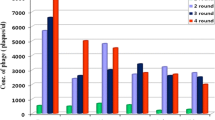
One phage clone with a high binding affinity to LNCaP cells was identified. Cell phage ELISA and immunostaining demonstrated high specificity of this phage to LNCaP cells. The fluorescein-labeled KYL peptide exhibited higher binding to LNCaP cells in comparison to other cells. The fusion peptide composed of the KYL peptide and the proapoptotic peptide induced cell death in LNCaP cells, but not in PC-3 cells. The KYL peptide-protamine conjugate also efficiently delivered a fluorescein-labeled siRNA into LNCaP cells.
Conclusion
We identified a LNCaP-specific peptide and demonstrated its potential applications in targeted drug delivery to LNCaP cells.









Similar content being viewed by others
REFERENCES
Jemal A, Siegel R, Ward E, Murray T, Xu J, Thun MJ. Cancer statistics, 2007. CA Cancer J Clin. 2007;57:43–66.
Mabjeesh NJ, Zhong H, Simons JW. Gene therapy of prostate cancer: current and future directions. Endocr-Relat Cancer. 2002;9:115–39.
Tai W, Mahato R, Cheng K. The role of HER2 in cancer therapy and targeted drug delivery. J Control Release 146:264–75.
Chang DK, Lin CT, Wu CH, Wu HC. A novel peptide enhances therapeutic efficacy of liposomal anti-cancer drugs in mice models of human lung cancer. PLoS ONE. 2009;4:e4171.
Yoneda Y, Steiniger SC, Capkova K, Mee JM, Liu Y, Kaufmann GF, et al. A cell-penetrating peptidic GRP78 ligand for tumor cell-specific prodrug therapy. Bioorg Med Chem Lett. 2008;18:1632–6.
Cesarone G, Edupuganti OP, Chen CP, Wickstrom E. Insulin receptor substrate 1 knockdown in human MCF7 ER+ breast cancer cells by nuclease-resistant IRS1 siRNA conjugated to a disulfide-bridged D-peptide analogue of insulin-like growth factor 1. Bioconjug Chem. 2007;18:1831–40.
Deutscher SL, Figueroa SD, Kumar SR. In-labeled KCCYSL peptide as an imaging probe for ErbB-2-expressing ovarian carcinomas. J Labelled Comp Radiopharm. 2009;52:583–90.
Lee S, Xie J, Chen X. Peptide-based probes for targeted molecular imaging. Biochemistry 49:1364–76.
Aggarwal S, Singh P, Topaloglu O, Isaacs JT, Denmeade SR. A dimeric peptide that binds selectively to prostate-specific membrane antigen and inhibits its enzymatic activity. Cancer Res. 2006;66:9171–7.
Burg MA, Pasqualini R, Arap W, Ruoslahti E, Stallcup WB. NG2 proteoglycan-binding peptides target tumor neovasculature. Cancer Res. 1999;59:2869–74.
Yang W, Luo D, Wang S, Wang R, Chen R, Liu Y, et al. TMTP1, a novel tumor-homing peptide specifically targeting metastasis. Clin Cancer Res. 2008;14:5494–502.
Wu C, Lo SL, Boulaire J, Hong ML, Beh HM, Leung DS, et al. A peptide-based carrier for intracellular delivery of proteins into malignant glial cells in vitro. J Control Release. 2008;130:140–5.
Kim Y, Lillo AM, Steiniger SC, Liu Y, Ballatore C, Anichini A, et al. Targeting heat shock proteins on cancer cells: selection, characterization, and cell-penetrating properties of a peptidic GRP78 ligand. Biochemistry. 2006;45:9434–44.
Jager S, Jahnke A, Wilmes T, Adebahr S, Vogtle FN, Delima-Hahn E, et al. Leukemia-targeting ligands isolated from phage-display peptide libraries. Leukemia. 2007;21:411–20.
Nishimura S, Takahashi S, Kamikatahira H, Kuroki Y, Jaalouk DE, O’Brien S, et al. Combinatorial targeting of the macropinocytotic pathway in leukemia and lymphoma cells. J Biol Chem. 2008;283:11752–62.
Joyce JA, Laakkonen P, Bernasconi M, Bergers G, Ruoslahti E, Hanahan D. Stage-specific vascular markers revealed by phage display in a mouse model of pancreatic islet tumorigenesis. Cancer Cell. 2003;4:393–403.
Newton JR, Kelly KA, Mahmood U, Weissleder R, Deutscher SL. In vivo selection of phage for the optical imaging of PC-3 human prostate carcinoma in mice. Neoplasia. 2006;8:772–80.
Shuklaand GS, Krag DN. Phage display selection for cell-specific ligands: development of a screening procedure suitable for small tumor specimens. J Drug Target. 2005;13:7–18.
Smithand GP, Petrenko VA. Phage display. Chem Rev. 1997;97:391–410.
Pero SC, Shukla GS, Armstrong AL, Peterson D, Fuller SP, Godin K, et al. Identification of a small peptide that inhibits the phosphorylation of ErbB2 and proliferation of ErbB2 overexpressing breast cancer cells. Int J Cancer. 2004;111:951–60.
Karasseva NG, Glinsky VV, Chen NX, Komatireddy R, Quinn TP. Identification and characterization of peptides that bind human ErbB-2 selected from a bacteriophage display library. J Protein Chem. 2002;21:287–96.
Sobeland RE, Sadar MD. Cell lines used in prostate cancer research: a compendium of old and new lines–part 2. J Urol. 2005;173:360–72.
Sobeland RE, Sadar MD. Cell lines used in prostate cancer research: a compendium of old and new lines–part 1. J Urol. 2005;173:342–59.
Israeli RS, Powell CT, Corr JG, Fair WR, Heston WD. Expression of the prostate-specific membrane antigen. Cancer Res. 1994;54:1807–11.
Giordano RJ, Cardo-Vila M, Lahdenranta J, Pasqualini R, Arap W. Biopanning and rapid analysis of selective interactive ligands. Nat Med. 2001;7:1249–53.
Liang S, Lin T, Ding J, Pan Y, Dang D, Guo C, et al. Screening and identification of vascular-endothelial-cell-specific binding peptide in gastric cancer. J Mol Med. 2006;84:764–73.
Bruccoleri RE, Haber E, Novotny J. Structure of antibody hypervariable loops reproduced by a conformational search algorithm. Nature. 1988;335:564–8.
Mai JC, Mi Z, Kim SH, Ng B, Robbins PD. A proapoptotic peptide for the treatment of solid tumors. Cancer Res. 2001;61:7709–12.
Arap MA, Lahdenranta J, Mintz PJ, Hajitou A, Sarkis AS, Arap W, et al. Cell surface expression of the stress response chaperone GRP78 enables tumor targeting by circulating ligands. Cancer Cell. 2004;6:275–84.
Bruckheimer E, Harvie P, Orthel J, Dutzar B, Furstoss K, Mebel E, et al. In vivo efficacy of folate-targeted lipid-protamine-DNA (LPD-PEG-Folate) complexes in an immunocompetent syngeneic model for breast adenocarcinoma. Cancer Gene Ther. 2004;11:128–34.
Zhang JS, Li S, Huang L. Cationic liposome-protamine-DNA complexes for gene delivery. Meth Enzymol. 2003;373:332–42.
Song E, Zhu P, Lee SK, Chowdhury D, Kussman S, Dykxhoorn DM, et al. Antibody mediated in vivo delivery of small interfering RNAs via cell-surface receptors. Nat Biotechnol. 2005;23:709–17.
Dinauer N, Lochmann D, Demirhan I, Bouazzaoui A, Zimmer A, Chandra A, et al. Intracellular tracking of protamine/antisense oligonucleotide nanoparticles and their inhibitory effect on HIV-1 transactivation. J Control Release. 2004;96:497–507.
Mannervik B, Axelsson K, Sundewall AC, Holmgren A. Relative contributions of thioltransferase-and thioredoxin-dependent systems in reduction of low-molecular-mass and protein disulphides. Biochem J. 1983;213:519–23.
Silver DA, Pellicer I, Fair WR, Heston WD, Cordon-Cardo C. Prostate-specific membrane antigen expression in normal and malignant human tissues. Clin Cancer Res. 1997;3:81–5.
McNamara 2nd JO, Andrechek ER, Wang Y, Viles KD, Rempel RE, Gilboa E, et al. Cell type-specific delivery of siRNAs with aptamer-siRNA chimeras. Nat Biotechnol. 2006;24:1005–15.
Dozmorov MG, Hurst RE, Culkin DJ, Kropp BP, Frank MB, Osban J, et al. Unique patterns of molecular profiling between human prostate cancer LNCaP and PC-3 cells. Prostate. 2009;69:1077–90.
Ellerby HM, Arap W, Ellerby LM, Kain R, Andrusiak R, Rio GD, et al. Anti-cancer activity of targeted pro-apoptotic peptides. Nat Med. 1999;5:1032–8.
Sithanandam G, Fornwald LW, Fields J, Anderson LM. Inactivation of ErbB3 by siRNA promotes apoptosis and attenuates growth and invasiveness of human lung adenocarcinoma cell line A549. Oncogene. 2005;24:1847–59.
Liu TG, Yin JQ, Shang BY, Min Z, He HW, Jiang JM, et al. Silencing of hdm2 oncogene by siRNA inhibits p53-dependent human breast cancer. Cancer Gene Ther. 2004;11:748–56.
ACKNOWLEDGMENTS
This work was supported by awards from National Cancer Institute (NCI), NIH (1R21CA143683-01) and National Institute of Alcohol Abuse and Alcoholism (NIAAA), NIH (1R21AA017960-01A1). We also would like to express thanks for the financial support from a start-up package at the University of Missouri-Kansas City.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Qin, B., Tai, W., Shukla, R.S. et al. Identification of a LNCaP-Specific Binding Peptide Using Phage Display. Pharm Res 28, 2422–2434 (2011). https://doi.org/10.1007/s11095-011-0469-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11095-011-0469-7