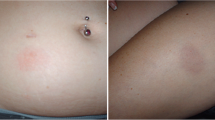
The safety of interferons was assessed by analyzing 499 spontaneous reports in the Russian database from 2015 to 2017. The results led to the conclusion that the safety profile of systemic interferon preparations that are widely used in medical practice to treat multiple sclerosis is defined by data in spontaneous reports for the development of adverse reactions to them or the ineffectiveness of using them.

Similar content being viewed by others
References
O. V. Bykova, A. N. Platonova, N. V. Gol’tsova, et al., Vopr. Sovrem. Pediatr., 8(6), 139 – 145 (2009).
F. Barkhof, C. Polman, E. Radue, et al., 21 st Congress of the European Committee for Treatment and Research in Multiple Sclerosis, Greece (2005), Abstr. 583.
H. Panitch, D. Goodin, G. Francis, et al., Neurology, 59, 1496 – 1506 (2002).
D. S. Kasatkin and N. N. Spirin, Vestn. Novosib. Gos. Univ. Ser.: Biol. Klin. Med., 13(1), 30 – 37 (2015).
I. I. Snegireva, E. Yu. Pasternak, K. E. Zatolochina, et al., Bezop. Risk Farmakoter., 5(4), 161 – 169 (2017).
V. Yu. Lizhdvoi, T. P. Ospel’nikova, and S. V. Kotov, Al’manakh Klin. Med., 44(3), 318 – 323 (2016).
Author information
Authors and Affiliations
Corresponding author
Additional information
Translated from Khimiko-Farmatsevticheskii Zhurnal, Vol. 52, No. 10, pp. 48 – 50, October, 2018.
Rights and permissions
About this article
Cite this article
Snegireva, I.I., Darmostukova, M.A., Kazakov, A.S. et al. Safe Use of Systemic Interferons for Multiple Sclerosis Treatment. Pharm Chem J 52, 863–864 (2019). https://doi.org/10.1007/s11094-019-1916-8
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11094-019-1916-8