Abstract
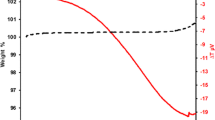
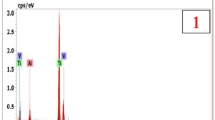
The present study investigates oxidation at 600 °C of alloy Sanicro 25 (42Fe22Cr25NiWCuNbN) in dry and wet O2 environments. The exposure time was 1–168 h. The oxidized samples were analyzed by grazing incidence X-ray diffraction, glow discharge optical emission spectroscopy, scanning electron microscopy, transmission electron microscopy and energy dispersive X-ray spectroscopy. Alloy Sanicro 25 showed protective oxidation behaviour under the present conditions. Initially, a thin and smooth corundum-type single layer base oxide formed, featuring a Cr-rich bottom part and a Fe-rich top. With time, double-layered oxide nodules form consisting of inward- and outward-growing parts. Below the oxide scale a 100–200 nm thick oxidation-affected zone formed in the alloy, which was depleted in Cr and enriched in Ni. In this region the chromium carbides and copper-rich particles present in the bulk alloy were dissolved. In O2 + H2O environment, chromium volatilized from the surface, causing the chromium content of the oxide to be lower than after oxidation in dry O2. However, under present experimental conditions, the Cr depletion of the scale was not enough to trigger accelerated corrosion of the alloy.




















Similar content being viewed by others
References
P. Kofstad, High Temperature Corrosion (Elsevier Applied Science Publishers Ltd, London and New York, 1988).
M. Thiele, H. Teichmann, W. Schwartz, and W. J. Quaddakers, VGB Kraftwerkstechnik 77, 129 (1997).
S. R. J. Saunders and N. L. MecCartney, Materials Science Forum 522–523, 119 (2006).
I. G. Wright and B. A. Pint, in Proceedings, NACE Corrosion 2002, Denver, CO, USA, April 8–11 (2002).
J. Ehlers, D. J. Young, E. J. Smaardijk, A. K. Tyagi, H. J. Penkalla, L. Singheiser, and W. J. Quadakkers, Corrosion Science 48, 3428 (2006).
T. Norby, Journal de Physique 4, 99 (1993).
B. Tveten, G. Hultquist, and T. Norby, Oxidation of Metals 51, 221 (1999).
A. Holt and P. Kofstad, Solid State Ionics 69, 137 (1994).
S. Jianian, Z. Longjiang, and L. Tiefan, Oxidation of Metals 48, 347 (1997).
C. A. Sterns, F. J. Kohl, and G. C. Frynerg, Journal of the Electrochemical Society 121, 945 (1974).
J. R. C. S. Tedmon, Journal of The Electrochemical Society 113, 766 (1966).
H. Asteman, J. E. Svensson, M. Norell, and L. G. Johansson, Oxidation of Metals 54, 11 (2000).
T. Jonsson, S. Canovic, F. Liu, H. Asteman, J. E. Svensson, L. G. Johansson, and M. Halvarsson, Oxidation of Metals 66, 295 (2006).
I. Panas, J. E. Svensson, H. Asteman, T. J. R. Johnson, and L. G. Johansson, Chemical Physics Letters 383, 549 (2004).
B. B. Ebbinghaus, Combustion and Flame 93, 119 (1993).
E. J. Opila, D. L. Myers, N. S. Jacobson, I. M. B. Nielsen, D. F. Johnson, J. K. Olminsky, and M. D. Allendorf, The Journal of Physical Chemistry A 111, 1971 (2007).
G. R. Holcomb, in Chromia Evaporation in Advanced Ultra-Supercritical Steam Boilers and Turbines, Thermodynamics—Kinetics of Dynamic Systems, ed. J. C. M. Piraján. InTech (2011). ISBN: 978-953-307-627-0. http://www.intechopen.com/articles/show/title/chromia-evaporation-in-advanced-ultra-supercritical-steam-boilers-and-turbines.
G. R. Holcomb, Oxidation of Metals 69, 163 (2008).
G. R. Holcomb, Journal of Electrochemical Society 156, C292 (2009).
J. E. Tang, F. Liu, H. Asteman, J. E. Svensson, L. G. Johansson, and M. Halvarsson, Materials at High Temperature 24, 27 (2007).
B. Pujilaksono, T. Jonsson, M. Halvarsson, I. Panas, J. E. Svensson, and L. G. Johansson, Oxidation of Metals 70, 163 (2008).
T. Jonsson, B. Pujilaksono, S. Hallström, J. Ågren, J. E. Svensson, L. G. Johansson, and M. Halvarsson, Corrosion Science 51, 1914 (2009).
N. Ramasubramanian, P. B. Sewell, and M. Cohen, Journal of the Electrochemical Society 115, 12 (1968).
T. Jonsson, S. Canovic, F. Liu, H. Asteman, J. E. Svensson, L. G. Johansson, and M. Halvarsson, Materials at High Temperature 22, 231 (2005).
F. Liu, J. E. Tang, T. Jonsson, S. Canovic, K. Segerdahl, J. E. Svensson, and M. Halvarsson, Oxidation of Metals 66, 295 (2006).
B. Pujilaksono, T. Jonsson, H. Heidari, M. Halvarsson, J. E. Svensson, and L. G. Johansson, Oxidation of Metals 75, 183 (2011).
H. Asteman, J. E. Svensson, and L. G. Johansson, Oxidation of Metals 57, 193 (2002).
C. Pettersson, T. Jonsson, C. Proff, M. Halvarsson, J. E. Svensson, and L. G. Johansson, Oxidation of Metals 74, 93 (2010).
H. Asteman, K. Segerdahl, J. E. Svensson, L. G. Johansson, M. Halvarsson, and J. E. Tang, Materials Science Forum 461–464, 775 (2004).
D. J. Young and B. Gleeson, Corrosion Science 44, 345 (2002).
S. N. Basu and G. J. Yurek, Oxidation of Metals 36, 281 (1991).
F. H. Stott, G. C. Wood, and J. Stringer, Oxidation of Metals 44, 113 (1995).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Intiso, L., Johansson, LG., Canovic, S. et al. Oxidation Behaviour of Sanicro 25 (42Fe22Cr25NiWCuNbN) in O2/H2O Mixture at 600 °C. Oxid Met 77, 209–235 (2012). https://doi.org/10.1007/s11085-011-9281-3
Received:
Revised:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11085-011-9281-3