Abstract
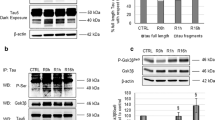
Altered proteostasis induced by amyloid peptide aggregation and hyperphosphorylation of tau protein, is a prominent feature of Alzheimer’s disease, which highlights the occurrence of endoplasmic reticulum stress and triggers the activation of the unfolded protein response (UPR), a signaling pathway that enforces adaptive programs to sustain proteostasis. In this study, we investigated the role of geniposide in the activation of UPR induced by high glucose in primary cortical neurons. We found that high glucose induced a significant activation of UPR, and geniposide enhanced the effect of high glucose on the phosphorylation of IRE1α, the most conserved UPR signaling branch. We observed that geniposide induced the expression of HRD1, an ubiquitin-ligase E3 in a time dependent manner, and amplified the expression of HRD1 induced by high glucose in primary cortical neurons. Suppression of IRE1α activity with STF-083010, an inhibitor of IRE1 phosphorylation, prevented the roles of geniposide on the expression of HRD1 and APP degradation in high glucose-treated cortical neurons. In addition, the results from RNA interfere on HRD1 revealed that HRD1 was involved in geniposide regulating APP degradation in cortical neurons. These data suggest that geniposide might be benefit to re-establish proteostasis by enhancing the UPR to decrease the load of APP in neurons challenged by high glucose.






Similar content being viewed by others
Abbreviations
- AD:
-
Alzheimer’s disease
- CHX:
-
Cyclohemide
- ER:
-
Endoplasmic reticulum
- ERAD:
-
Endoplasmic reticulum-associated degradation
- HRD1:
-
Hmg-CoA reductase degradation ligase
- UPR:
-
Unfolded protein response
References
Papassotiropoulos A, Streffer JR, Tsolaki M, Schmid S, Thal D, Nicosia F, Iakovidou V, Maddalena A, Lutjohann D, Ghebremedhin E, Hegi T, Pasch T, Traxler M, Bruhl A, Benussi L, Binetti G, Braak H, Nitsch RM, Hock C (2003) Increased brain beta-amyloid load, phosphorylated tau, and risk of Alzheimer disease associated with an intronic CYP46 polymorphism. Arch Neurol 60:29–35
Smith MA, Drew KL, Nunomura A, Takeda A, Hirai K, Zhu X, Atwood CS, Raina AK, Rottkamp CA, Sayre LM, Friedland RP, Perry G (2002) Amyloid-beta, tau alterations and mitochondrial dysfunction in Alzheimer disease: the chickens or the eggs? Neurochem Int 40:527–531
Taylor JP, Hardy J, Fischbeck KH (2002) Toxic proteins in neurodegenerative disease. Science 296:1991–1995
Delic M, Rebnegger C, Wanka F, Puxbaum V, Haberhauer-Troyer C, Hann S, Kollensperger G, Mattanovich D, Gasser B (2012) Oxidative protein folding and unfolded protein response elicit differing redox regulation in endoplasmic reticulum and cytosol of yeast. Free Radical Biol Med 52:2000–2012
Hoozemans JJ, Veerhuis R, Van Haastert ES, Rozemuller JM, Baas F, Eikelenboom P, Scheper W (2005) The unfolded protein response is activated in Alzheimer’s disease. Acta Neuropathol 110:165–172
Hoozemans JJ, van Haastert ES, Nijholt DA, Rozemuller AJ, Scheper W (2012) Activation of the unfolded protein response is an early event in Alzheimer’s and Parkinson’s disease. Neuro-Degenerative Dis 10:212–215
Scheper W, Nijholt DA, Hoozemans JJ (2011) The unfolded protein response and proteostasis in Alzheimer disease: preferential activation of autophagy by endoplasmic reticulum stress. Autophagy 7:910–911
Cunnane S, Nugent S, Roy M, Courchesne-Loyer A, Croteau E, Tremblay S, Castellano A, Pifferi F, Bocti C, Paquet N, Begdouri H, Bentourkia M, Turcotte E, Allard M, Barberger-Gateau P, Fulop T, Rapoport SI (2011) Brain fuel metabolism, aging, and Alzheimer’s disease. Nutrition 27:3–20
Dukart J, Kherif F, Mueller K, Adaszewski S, Schroeter ML, Frackowiak RS, Draganski B, Alzheimer’s Disease Neuroimaging I (2013) Generative FDG-PET and MRI model of aging and disease progression in Alzheimer’s disease. PLoS Comput Biol 9:e1002987
Yin F, Sancheti H, Patil I, Cadenas E (2016) Energy metabolism and inflammation in brain aging and Alzheimer’s disease. Free Radical Biol Med 100:108–122
Liu J, Yin F, Zheng X, Jing J, Hu Y (2007) Geniposide, a novel agonist for GLP-1 receptor, prevents PC12 cells from oxidative damage via MAP kinase pathway. Neurochem Int 51:361–369
Liu J, Zheng X, Yin F, Hu Y, Guo L, Deng X, Chen G, Jiajia J, Zhang H (2006) Neurotrophic property of geniposide for inducing the neuronal differentiation of PC12 cells. Int J Dev Neurosci 24:419–424
Zhang Y, Yin F, Liu J, Liu Z (2016) Geniposide attenuates the phosphorylation of Tau protein in cellular and insulin-deficient APP/PS1 transgenic mouse model of Alzheimer’s disease. Chem Biol Drug Des 87:409–418
Liu J, Zhang Y, Deng X, Yin F (2013) Geniposide decreases the level of Abeta1-42 in the hippocampus of streptozotocin-induced diabetic rats. Acta Biochim et Biophys Sin 45:787–791
Zhang Y, Yin F, Liu J, Liu Z, Guo L, Xia Z, Zidichouski J (2015) Geniposide attenuates insulin-deficiency-induced acceleration of beta-amyloidosis in an APP/PS1 transgenic model of Alzheimer’s disease. Neurochem Int 89:7–16
Butterfield DA, Di Domenico F, Barone E (2014) Elevated risk of type 2 diabetes for development of Alzheimer disease: a key role for oxidative stress in brain. Biochim et Biophys Acta 1842:1693–1706
Razay G, Vreugdenhil A, Wilcock G (2007) The metabolic syndrome and Alzheimer disease. Arch Neurol 64:93–96
Sato N, Morishita R (2015) The roles of lipid and glucose metabolism in modulation of beta-amyloid, tau, and neurodegeneration in the pathogenesis of Alzheimer disease. Front Aging Neurosci 7:199
Negishi T, Ishii Y, Kyuwa S, Kuroda Y, Yoshikawa Y (2003) Inhibition of staurosporine-induced neuronal cell death by bisphenol A and nonylphenol in primary cultured rat hippocampal and cortical neurons. Neurosci Lett 353:99–102
Yin F, Zhang Y, Guo L, Kong S, Liu J (2012) Geniposide regulates insulin-degrading enzyme expression to inhibit the cytotoxicity of Abeta(1)(-)(4)(2) in cortical neurons. CNS Neurol Disord Drug Targets 11:1045–1051
Schafer FQ, Buettner GR (2001) Redox environment of the cell as viewed through the redox state of the glutathione disulfide/glutathione couple. Free Radical Biol Med 30:1191–1212
Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin phenol reagent. J Biol Chem 193:265–275
Liu C, Hao Y, Yin F, Zhang Y, Liu J (2017) Geniposide protects pancreatic beta cells from high glucose-mediated injury by activation of AMP-activated protein kinase. Cell Biol Int 41:544–554
Higa A, Chevet E (2012) Redox signaling loops in the unfolded protein response. Cell Signal 24:1548–1555
Luis A, Martins JD, Silva A, Ferreira I, Cruz MT, Neves BM (2014) Oxidative stress-dependent activation of the eIF2alpha-ATF4 unfolded protein response branch by skin sensitizer 1-fluoro-2,4-dinitrobenzene modulates dendritic-like cell maturation and inflammatory status in a biphasic manner [corrected]. Free Radical Biol Med 77:217–229
Milisav I, Suput D, Ribaric S (2015) Unfolded protein response and macroautophagy in Alzheimer’s, Parkinson’s and Prion diseases. Molecules 20:22718–22756
Dattilo S, Mancuso C, Koverech G, Di Mauro P, Ontario ML, Petralia CC, Petralia A, Maiolino L, Serra A, Calabrese EJ, Calabrese V (2015) Heat shock proteins and hormesis in the diagnosis and treatment of neurodegenerative diseases. Immun Ageing 12:20
Calabrese V, Cornelius C, Mancuso C, Pennisi G, Calafato S, Bellia F, Bates TE, Giuffrida Stella AM, Schapira T, Dinkova Kostova AT, Rizzarelli E (2008) Cellular stress response: a novel target for chemoprevention and nutritional neuroprotection in aging, neurodegenerative disorders and longevity. Neurochem Res 33:2444–2471
Boyce M, Bryant KF, Jousse C, Long K, Harding HP, Scheuner D, Kaufman RJ, Ma D, Coen DM, Ron D, Yuan J (2005) A selective inhibitor of eIF2alpha dephosphorylation protects cells from ER stress. Science 307:935–939
Wiseman RL, Balch WE (2005) A new pharmacology–drugging stressed folding pathways. Trends Mol Med 11:347–350
Imaizumi K, Miyoshi K, Katayama T, Yoneda T, Taniguchi M, Kudo T, Tohyama M (2001) The unfolded protein response and Alzheimer’s disease. Biochim et Biophys Acta 1536:85–96
Matus S, Lisbona F, Torres M, Leon C, Thielen P, Hetz C (2008) The stress rheostat: an interplay between the unfolded protein response (UPR) and autophagy in neurodegeneration. Curr Mol Med 8:157–172
Santos CX, Tanaka LY, Wosniak J, Laurindo FR (2009) Mechanisms and implications of reactive oxygen species generation during the unfolded protein response: roles of endoplasmic reticulum oxidoreductases, mitochondrial electron transport, and NADPH oxidase. Antioxid Redox Signal 11:2409–2427
Jung ES, Hong H, Kim C, Mook-Jung I (2015) Acute ER stress regulates amyloid precursor protein processing through ubiquitin-dependent degradation. Sci Rep 5:8805
Bernasconi R, Galli C, Kokame K, Molinari M (2013) Autoadaptive ER-associated degradation defines a preemptive unfolded protein response pathway. Mol Cell 52:783–793
Placido AI, Pereira CM, Duarte AI, Candeias E, Correia SC, Santos RX, Carvalho C, Cardoso S, Oliveira CR, Moreira PI (2014) The role of endoplasmic reticulum in amyloid precursor protein processing and trafficking: implications for Alzheimer’s disease. Biochim et Biophys Acta 1842:1444–1453
Kaneko M, Koike H, Saito R, Kitamura Y, Okuma Y, Nomura Y (2010) Loss of HRD1-mediated protein degradation causes amyloid precursor protein accumulation and amyloid-beta generation. J Neurosci 30:3924–3932
Umeda T, Tomiyama T, Sakama N, Tanaka S, Lambert MP, Klein WL, Mori H (2011) Intraneuronal amyloid beta oligomers cause cell death via endoplasmic reticulum stress, endosomal/lysosomal leakage, and mitochondrial dysfunction in vivo. J Neurosci Res 89:1031–1042
Saito R, Kaneko M, Okuma Y, Nomura Y (2010) Correlation between decrease in protein levels of ubiquitin ligase HRD1 and amyloid-beta production. J Pharmacol Sci 113:285–288
Acknowledgements
This work was supported by grants from Chongqing Science and Technology Committee (CSTC, 2015jcyjbx0064), Chongqing Science Found for Distinguished Young Scholars (2014jcyjjq10003), the Innovation of Science and Technology Leading Talent in Chongqing (2014kjcxljrc0018), the Innovative Research Team Development Program at the University of Chongqing (CXTDX201601031), and Chongqing University of Technology (2015XH23, 2015ZD26).
Author information
Authors and Affiliations
Corresponding authors
Rights and permissions
About this article
Cite this article
Cui, H., Deng, M., Zhang, Y. et al. Geniposide Increases Unfolded Protein Response-Mediating HRD1 Expression to Accelerate APP Degradation in Primary Cortical Neurons. Neurochem Res 43, 669–680 (2018). https://doi.org/10.1007/s11064-018-2469-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-018-2469-z