Abstract
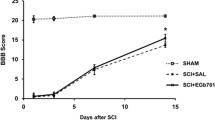
This study aimed to investigate the therapeutic effects of epigallocatechin-3-gallate (EGCG) administered by subarachnoid injection following spinal cord injury (SCI) in rats and to explore the underlying mechanism. Sprague–Dawley rats were randomly divided into four groups of 12 as follows: a sham group (laminectomy only); a control group; a 10 mg/kg EGCG-treated group; and a 20 mg/kg EGCG-treated group. SCI was induced in the rats using the modified weight-drop method (10 g × 4 cm) at the T10 (10th thoracic vertebral) level. EGCG (10 or 20 mg/kg) or vehicle as control was administered by subarachnoid injection at lumbar level 4 immediately after SCI. Locomotor functional recovery was assessed during the four weeks post-operation using open-field locomotor tests and inclined-plane tests. At the end of the study, the segments of spinal cord encompassing the injury site were removed for histopathological analysis. Immunohistochemical and Western blot analyses were performed to observe the expression of: the B cell CLL/lymphoma-2 (Bcl-2), Bcl-2-associated X protein (Bax), brain-derived neurotrophic factor (BDNF) and glial cell line-derived neurotrophic factor (GDNF). The results showed that the EGCG-treated animals had significantly better recovery of locomotor function, less myelin loss, greater Bcl-2 expression and attenuated Bax expression. In addition, the EGCG treatment significantly increased the expression of BDNF and GDNF after SCI. These findings suggest that EGCG treatment can significantly improve locomotor recovery, and this neuroprotective effect may be related to the up-regulation of BDNF and GDNF, and the inhibition of apoptosis-related proteins. Therefore, EGCG may be a promising therapeutic agent for SCI.




Similar content being viewed by others
Change history
22 April 2020
Since the publication of our article [1] it has come to our attention that there was an error in Figure 4 in which the bottom left immunochemistry panel Control/Bax was a duplication of the bottom right immunohistochemistry panel EGCG/GDNF in Figure 3.
References
Pereira JE, Costa LM, Cabrita AM et al (2009) Methylprednisolone fails to improve functional and histological outcome following spinal cord injury in rats. Exp Neurol 220(1):71–81
Yu J, Jia Y, Guo Y et al (2010) Epigallocatechin-3-gallate protects motor neurons and regulates glutamate level. FEBS Lett 584(13):2921–2925
Crespy V, Williamson G (2004) A review of the health effects of green tea catechins in in vivo animal models. J Nutr 134(12):3431S–3440S
Mandel SA, Avramovich-Tirosh Y, Reznichenko L et al (2005) Multifunctional activities of green tea catechins in neuroprotection. Modulation of cell survival genes, iron-dependent oxidative stress and PKC signaling pathway. Neurosignals 14(1–2):46–60
Cho HS, Kim S, Lee SY et al (2008) Protective effect of the green tea component, L-theanine on environmental toxins-induced neuronal cell death. Neurotoxicology 29(4):656–662
Nath S, Bachani M, Harshavardhana D et al (2012) Catechins protect neurons against mitochondrial toxins and HIV proteins via activation of the BDNF pathway. J Neurovirol 18(6):445–455
Kimura M, Umegaki K, Kasuya Y et al (2002) The relation between single/double or repeated tea catechin ingestions and plasma antioxidant activity in humans. Eur J Clin Nutr 56(12):1186–1193
Choi YB, Kim YI, Lee KS et al (1019) (2004) Protective effect of epigallocatechin gallate on brain damage after transient middle cerebral artery occlusion in rats. Brain Res 1–2:47–54
Park JW, Hong JS, Lee KS et al (2010) Green tea polyphenol (-)-epigallocatechin gallate reduces matrix metalloproteinase-9 activity following transient focal cerebral ischemia. J Nutr Biochem 21(11):1038–1044
Itoh T, Imano M, Nishida S et al (2011) (−)-Epigallocatechin-3-gallate protects against neuronal cell death and improves cerebral function after traumatic brain injury in rats. Neuromolecular Med 13(4):300–309
Khalatbary AR, Tiraihi T, Boroujeni MB et al (2010) Effects of epigallocatechin gallate on tissue protection and functional recovery after contusive spinal cord injury in rats. Brain Res 1306:168–175
Khalatbary AR, Ahmadvand H (2011) Anti-inflammatory effect of the epigallocatechin gallate following spinal cord trauma in rat. Iran Biomed J 15(1–2):31–37
Hellal F, Hurtado A, Ruschel J et al (2011) Microtubule stabilization reduces scarring and causes axon regeneration after spinal cord injury. Science 331(6019):928–931
Han X, Yang N, Cui Y et al (2012) Simvastatin mobilizes bone marrow stromal cells migrating to injured areas and promotes functional recovery after spinal cord injury in the rat. Neurosci Lett 521(2):136–141
Choi JI, Kim WM, Lee HG et al (2012) Role of neuronal nitric oxide synthase in the antiallodynic effects of intrathecal EGCG in a neuropathic pain rat model. Neurosci Lett 510(1):53–57
Kuang X, Huang Y, Gu HF et al (2012) Effects of intrathecal epigallocatechin gallate, an inhibitor of Toll-like receptor 4, on chronic neuropathic pain in rats. Eur J Pharmacol 676(1–3):51–56
Han X, Yang N, Xu Y et al (2011) Simvastatin treatment improves functional recovery after experimental spinal cord injury by upregulating the expression of BDNF and GDNF. Neurosci Lett 487(3):255–259
Basso DM, Beattie MS, Bresnahan JC (1995) A sensitive and reliable locomotor rating scale for open field testing in rats. J Neurotrauma 12(1):1–21
Rong W, Wang J, Liu X et al (2012) 17beta-estradiol attenuates neural cell apoptosis through inhibition of JNK phosphorylation in SCI rats and excitotoxicity induced by glutamate in vitro. Int J Neurosci 122(7):381–387
Rong W, Wang J, Liu X et al (2012) Naringin treatment improves functional recovery by increasing BDNF and VEGF expression, inhibiting neuronal apoptosis after spinal cord injury. Neurochem Res 37(8):1615–1623
Wang J, Rong W, Hu X et al (2012) Hyaluronan tetrasaccharide in the cerebrospinal fluid is associated with self-repair of rats after chronic spinal cord compression. Neuroscience 210:467–480
Mandel S, Amit T, Reznichenko L et al (2006) Green tea catechins as brain-permeable, natural iron chelators-antioxidants for the treatment of neurodegenerative disorders. Mol Nutr Food Res 50(2):229–234
Weinreb O, Amit T, Mandel S et al (2009) Neuroprotective molecular mechanisms of (-)-epigallocatechin-3-gallate: a reflective outcome of its antioxidant, iron chelating and neuritogenic properties. Genes Nutr 4(4):283–296
Wang J, Ren Z, Xu Y et al (2012) Epigallocatechin-3-gallate ameliorates experimental autoimmune encephalomyelitis by altering balance among CD4 + T-cell subsets. Am J Pathol 180(1):221–234
Ramer MS (2012) Endogenous neurotrophins and plasticity following spinal deafferentation. Exp Neurol 235(1):70–77
Yaguchi M, Ohta S, Toyama Y et al (2008) Functional recovery after spinal cord injury in mice through activation of microglia and dendritic cells after IL-12 administration. J Neurosci Res 86(9):1972–1980
Zhang L, Ma Z, Smith GM et al (2009) GDNF-enhanced axonal regeneration and myelination following spinal cord injury is mediated by primary effects on neurons. Glia 57(11):1178–1191
Yakovlev AG, Faden AI (2001) Caspase-dependent apoptotic pathways in CNS injury. Mol Neurobiol 24(1–3):131–144
Nakajima H, Uchida K, Yayama T et al (2010) Targeted retrograde gene delivery of brain-derived neurotrophic factor suppresses apoptosis of neurons and oligodendroglia after spinal cord injury in rats. Spine (Phila Pa 1976) 35(5):497–504
Zha H, Aime-Sempe C, Sato T et al (1996) Proapoptotic protein Bax heterodimerizes with Bcl-2 and homodimerizes with Bax via a novel domain (BH3) distinct from BH1 and BH2. J Biol Chem 271(13):7440–7444
Conflict of interest
None.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Tian, W., Han, XG., Liu, YJ. et al. Intrathecal Epigallocatechin Gallate Treatment Improves Functional Recovery After Spinal Cord Injury by Upregulating the Expression of BDNF and GDNF. Neurochem Res 38, 772–779 (2013). https://doi.org/10.1007/s11064-013-0976-5
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-013-0976-5