Abstract
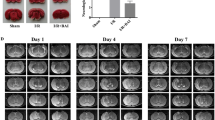
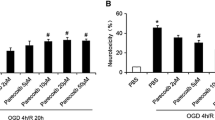
Parecoxib is a recently described novel COX-2 inhibitor whose functional significance and neuroprotective mechanisms remain elusive. Therefore, in this study, we aimed to investigate whether delayed administration of parecoxib inhibited mitochondria-mediated neuronal apoptosis induced by ischemic reperfusion injury via phosphorylating Akt and its downstream target protein, glycogen synthase kinase 3β (GSK-3β). Adult male Sprague–Dawley rats were administered parecoxib (10 or 30 mg kg−1, IP) or isotonic saline twice a day starting 24 h after middle cerebral artery occlusion (MCAO) for three consecutive days. Cerebral infarct volume, apoptotic neuron, caspase-3 immunoreactivity and the protein expression of p-Akt, p-GSK-3β and Cytochrome C in cerebral ischemic cortex were evaluated at 96 h after reperfusion. Parecoxib significantly diminished infarct volume and attenuated neuron apoptosis in a dose-independent manner, compared with MCAO group alone. Increased p-Akt and p-GSK-3β was observed in the ischemic penumbra of parecoxib group after stroke. Moreover, parecoxib also reduced the release of Cytochrome C from mitochondrial into cytosol and attenuated the caspase-3 immunoreactivity in the penumbra. Taken together, these results suggested that parecoxib ameliorated postischemic mitochondria-mediated neuronal apoptosis induced by focal cerebral ischemia in rats and this neuroprotective potential is involved in phosphorylation of Akt and GSK-3β.





Similar content being viewed by others
Abbreviations
- COX-2:
-
Cyclooxygenase-2
- GSK-3β:
-
Glycogen synthase kinase 3β
- MCAO:
-
Middle cerebral artery occlusion
- PI3 K:
-
Phosphatidylinositol-3 kinase
- NSAIDS:
-
Non-steroidal anti-inflammatory drugs
- TTC:
-
2, 3, 5-triphenyl-tetrazolium chloride
- BBB:
-
Blood brain barrier
- ADC:
-
Apparent diffusion coefficient
- ANT:
-
Adenine nucleotide translocase
- MPTP:
-
Mitochondrial permeability transition pore
References
Wang Q, Tang XN, Yenari MA (2007) The inflammatory response in stroke. J Neuroimmunol 184:53–68
Zheng Z, Yenari MA (2004) Post-ischemic inflammation: molecular mechanisms and therapeutic implications. Neurol Res 26:884–892
Phillis JW, Horrocks LA, Farooqui AA (2006) Cyclooxygenases, lipoxygenases, and epoxygenases in CNS: their role and involvement in neurological disorders. Brain Res Rev 52:201–243
Li J, Liang X, Wang Q, Breyer RM, McCullough L, Andreasson K (2008) Misoprostol, an anti-ulcer agent and PGE2 receptor agonist, protects against cerebral ischemia. Neurosci Lett 438:210–215
Candelario-Jalil E (2008) Nimesulide as a promising neuroprotectant in brain ischemia: new experimental evidences. Pharmacol Res 57:266–273
Sugimoto K, Iadecola C (2003) Delayed effect of administration of COX-2 inhibitor in mice with acute cerebral ischemia. Brain Res 960:273–276
Candelario-Jalil E, Fiebich BL (2008) Cyclooxygenase inhibition in ischemic brain injury. Curr Pharm Des 14:1401–1418
Iadecola C, Niwa K, Nogawa S, Zhao X, Nagayama M, Araki E, Morham S, Ross ME (2001) Reduced susceptibility to ischemic brain injury and N-methyl-D-aspartate mediated neurotoxicity in cyclooxygenase-2-deficient mice. Proc Natl Acad Sci USA 98:1294–1299
Doré S, Otsuka T, Mito T, Sugo N, Hand T, Wu L, Hurn PD, Traystman RJ, Andreasson K (2003) Neuronal overexpression of cyclooxygenase-2 increases cerebral infarction. Ann Neurol 54:155–162
Abbate A, Salloum FN, Ockaili RA, Fowler AA III, Biondi-Zoccai GG, Straino S, Lipinski MJ, Baldi A, Crea F, Biasucci LM, Vetrovec GW, Kukreja RC (2007) Improvement of cardiac function with parecoxib, a cyclooxygenase-2 inhibitor, in a rat model of ischemic heart failure. J Cardiovasc Pharmacol 49:416–418
Salloum FN, Hoke NN, Seropian IM, Varma A, Ownby ED, Houser JE, Van Tassell BW, Abbate A (2009) Parecoxib inhibits apoptosis in acute myocardial infarction due to permanent coronary ligation but not due to ischemia-reperfusion. J Cardiovasc Pharmacol 53:495–498
Nørregaard R, Jensen BL, Topcu SO, Wang G, Schweer H, Nielsen S, Frøkiaer J (2010) Urinary tract obstruction induces transient accumulation of COX-2 derived prostanoids in kidney tissue. Am J Physiol Regul Integr Comp Physiol 298:R1017–R1025
Patel NS, Cuzzocrea S, Collino M, Chaterjee PK, Mazzon E, Britti D, Yaqoob MM, Thiemermann C (2007) The role of cycloxygenase-2 in the rodent kidney following ischaemia/reperfusion injury in vivo. Eur J Pharmacol 562:148–154
Kelsen J, Kjaer K, Chen G, Pedersen M, Røhl L, Frøkiaer J, Nielsen S, Nyengaard JR, Rønn LC (2006) Parecoxib is neuroprotective in spontaneously hypertensive rats after transient middle cerebral artery occlusion: a divided treatment response? J Neuroinflammation 3:31
Polascheck N, Bankstahl M, Löscher W (2010) The COX-2 inhibitor parecoxib is neuroprotective but not antiepileptogenic in the pilocarpine model of temporal lobe epilepsy. Exp Neurol 224:219–233
Graham SH, Chen J (2001) Programmed cell death in cerebral ischemia. J Cereb Blood Flow Metab 21:99–109
Reed JC (2000) Mechanisms of apoptosis. Am J Pathol 15:1415–1430
Noshita N, Lewén A, Sugawara T, Chan PH (2001) Evidence of phosphorylation of Akt and neuronal survival after transient focal cerebral ischemia in mice. J Cereb Blood Flow Metab 21:1442–1450
Hanada M, Feng J, Hemmings BA (2004) Structure, regulation and function of PKB/AKT—a major therapeutic target. Biochim Biophys Acta 1697:3–16
Clodfelder-Miller B, De Sarno P, Zmijewska AA, Song L, Jope RS (2005) Physiological and pathological changes in glucose regulate brain Akt and glycogen synthase kinase-3β. J Biol Chem 280:39723–39731
Leclerc S, Garnier M, Hoessel R, Marko D, Bibb JA, Snyder GL, Greengard P, Biernat J, Wu YZ, Mandelkow EM, Eisenbrand G, Meijer L (2001) Indirubins inhibit glycogen synthase-3β and CDK5/P25, two protein kinases involved in abnormal tau phosphorylation in Alzheimer’s disease. J Biol Chem 276:251–260
Cimarosti H, Zamin LL, Frozza R, Nassif M, Horn AP, Tavares A, Netto CA, Salbego C (2005) Estradiol protects against oxygen and glucose deprivation in rat hippocampal organotypic cultures and activates Akt and inactivates GSK-3beta. Neurochem Res 30:191–199
Nishimoto T, Kihara T, Akaike A, Niidome T, Sugimoto H (2008) Alpha-amino-3-hydroxy-5-methyl-4-isoxazole propionate attenuates glutamate-induced caspase-3 cleavage via regulation of glycogen synthase kinase 3beta. J Neurosci Res 86:1096–1105
Brywe KG, Mallard C, Gustavsson M, Hedtjärn M, Leverin AL, Wang X, Blomgren K, Isgaard J, Hagberg H (2005) IGF-I neuroprotection in the immature brain after hypoxia-ischemia, involvement of Akt and GSK3beta? Eur J Neurosci 21:1489–1502
Song JQ, Teng X, Cai Y, Tang CS, Qi YF (2009) Activation of Akt/GSK-3beta signaling pathway is involved in intermedin(1–53) protection against myocardial apoptosis induced by ischemia/reperfusion. Apoptosis 14:1299–1307
Chu K, Jeong SW, Jung KH, Han SY, Lee ST, Kim M, Roh JK (2004) Celecoxib induces functional recovery after intracerebral hemorrhage with reduction of brain edema and perihe-matomal cell death. J Cereb Blood Flow Metab 24:926–933
Gopez JJ, Yue H, Vasudevan R, Malik AS, Fogelsanger LN, Lewis S, Panikashvili D, Shohami E, Jansen SA, Narayan RK, Strauss KI (2005) Cyclooxygenase-2-specific inhibitor improves functional outcomes, provides neuroprotection, and reduces inflammation in a rat model of traumatic brain injury. Neurosurgery 56:590–604
Candelario-Jalil E, González-Falcón A, García-Cabrera M, León OS, Fiebich BL (2004) Wide therapeutic time window for nimesulide neuroprotection in a model of transient focal cerebral ischemia in the rat. Brain Res 1007:98–108
Belayev L, Alonso OF, Busto R, Zhao W, Ginsberg MD (1996) Middle cerebral artery occlusion in the rat by intraluminal suture. Neurological and pathological evaluation of an improved model. Stroke 27:1616–1622
Wang HY, Wang GL, Yu YH, Wang Y (2009) The role of phosphoinositide-3-kinase/Akt pathway in propofol-induced postconditioning against focal cerebral ischemia-reperfusion injury in rats. Brain Res 1297:177–184
Li L, Peng L, Zuo Z (2008) Isoflurane preconditioning increases B-cell lymphoma-2 expression and reduces Cytochrome C release from the mitochondria in the ischemic penumbra of rat brain. Eur J Pharmacol 586:106–113
Hirai K, Hayashi T, Chan PH, Zeng J, Yang GY, Basus VJ, James TL, Litt L (2004) PI3 K inhibition in neonatal rat brain slices during and after hypoxia reduces phospho-Akt and increases cytosolic Cytochrome C and apoptosis. Brain Res Mol Brain Res 124:51–61
Dirnagl U (2004) Inflammation in stroke: the good, the bad, and the unknown. Ernst Schering Res Found Workshop 47:87–99
Kawano T, Anrather J, Zhou P, Park L, Wang G, Frys KA (2006) Prostaglandin E2 EP1 receptors: downstream effectors of COX-2 neurotoxicity. Nat Med 12:225–229
Xiang Z, Thomas S, Pasinetti G (2007) Increased neuronal injury in transgenic mice with neuronal overexpression of human cyclooxygenase-2 is reversed by hypothermia and rofecoxib treatment. Curr Neurovasc Res 4:274–279
Candelario-Jalil E, Gonzalez-Falcon A, Garcia-Cabrera M, Alvarez D, Al-Dalain S, Martinez G (2003) Assessment of the relative contribution of COX-1 and COX-2 isoforms to ischemia-induced oxidative damage and neurodegeneration following transient global cerebral ischemia. J Neurochem 86:545–555
Nogawa S, Zhang F, Ross ME, Iadecola C (1997) Cyclooxygenase-2 gene expression in neurons contributes to ischemic brain damage. J Neurosci 17:2746–2755
Ohtsuki T, Kitagawa K, Yamagata K, Mandai K, Mabuchi T, Matsushita K (1996) Induction of cyclooxygenase-2 mRNA in gerbil hippocampal neurons after transient forebrain ischemia. Brain Res 736:353–356
Hazell AS (2007) Excitotoxic mechanisms in stroke: an update of concepts and treatment strategies. Neurochem Int 50:941–953
Wu T, Wu H, Wang J, Wang J (2011) Expression and cellular localization of cyclooxygenases and prostaglandin E synthases in the hemorrhagic brain. J Neuroinflammation 8:22
Kelso ML, Scheff SW, Pauly JR, Loftin CD (2009) Effects of genetic deficiency of cyclooxygenase-1 or cyclooxygenase-2 on functional and histological outcomes following traumatic brain injury in mice. BMC Neurosci 10:108
Hakan T, Toklu HZ, Biber N, Ozevren H, Solakoglu S, Demirturk P, Aker FV (2010) Effect of COX-2 inhibitor meloxicam against traumatic brain injury-induced biochemical, histopathological changes and blood-brain barrier permeability. Neurol Res 32:629–635
Dembo G, Park SB, Kharasch ED (2005) Central nervous system concentrations of cyclooxygenase-2 inhibitors in humans. Anesthesiology 102:409–415
Jiang X, Wang X (2000) Cytochrome C promotes caspase-9 activation by inducing nucleotide binding to Apaf-1. J Biol Chem 275:31199–31203
Boatright KM, Renatus M, Scott FL, Sperandio S, Shin H, Pedersen IM (2003) A unified model for apical caspase activation. Mol Cell 11:529–541
Forde JE, Dale TC (2007) Glycogen synthase kinase 3: a key regulator of cellular fate. Cell Mol Life Sci 64:1930–1944
Raphael J, Abedat S, Rivo J, Meir K, Beeri R, Pugatsch T (2006) Volatile anesthetic preconditioning attenuates myocardial apoptosis in rabbits after regional ischemia and reperfusion via Akt signaling and modulation of Bcl-2 family proteins. J Pharmacol Exp Ther 318:186–194
Zhao H, Shimohata T, Wang JQ, Sun G, Schaal DW, Sapolsky RM (2005) Akt contributes to neuroprotection by hypothermia against cerebral ischemia in rats. J Neurosci 25:9794–9806
Juhaszova M, Zorov DB, Kim SH, Pepe S, Fu Q, Fishbein KW, Ziman BD, Wang S, Ytrehus K, Antos CL, Olson EN, Sollott SJ (2004) Glycogen synthase kinase-3beta mediates convergence of protection signaling to inhibit the mitochondrial permeability transition pore. J Clin Invest 113:1535–1549
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ye, Z., Wang, N., Xia, P. et al. Delayed Administration of Parecoxib, a Specific COX-2 Inhibitor, Attenuated Postischemic Neuronal Apoptosis by Phosphorylation Akt and GSK-3β. Neurochem Res 37, 321–329 (2012). https://doi.org/10.1007/s11064-011-0615-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-011-0615-y