Abstract
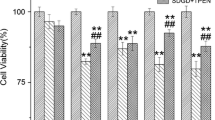
The features of neuronal damage induced by the mitochondrial toxin NaN3 were investigated in rat primary cortical neuron cultures. Cell viability (MTT colorimetric determination) and transmembrane mitochondrial potential (J-C1 fluorescence) were concentration-dependently reduced 24 h after NaN3; neither nuclear fragmentation by DAPI, nor Annexin V positivity by flow cytometry were detected, ruling out the occurrence of apoptosis. The loss in cell viability (to 54 ± 2%) observed 24 h after a 10-min treatment with 3 mM NaN3 was prevented by the NMDA glutamate receptor antagonist MK801 (1 μM), by the antioxidants trolox (100 μM) and acetyl-l-carnitine (1 mM) and by the nitric oxide synthase inhibitor, L-NAME (100 μM), but not by the guanylylcyclase inhibitor ODQ, 10 μM. The mitochondrial dysfunction induced by NaN3 provides a common platform for investigating the mechanisms of both ischemic and degenerative neuronal injury, useful for screening potential protective agents against neuronal death.





Similar content being viewed by others
References
Sas K, Robotka H, Toldi J et al (2007) Mitochondria, metabolic disturbances, oxidative stress and the kynurenine system, with focus on neurodegenerative disorders. J Neurol Sci 257:221–239. doi:10.1016/j.jns.2007.01.033
Mehta SL, Manhas N, Raghubir R (2007) Molecular targets in cerebral ischemia for developing novel therapeutics. Brain Res Brain Res Rev 54:34–66. doi:10.1016/j.brainresrev.2006.11.003
Bano D, Nicotera P (2007) Ca2+ signals and neuronal death in brain ischemia. Stroke 38:674–676. doi:10.1161/01.STR.0000256294.46009.29
Moncada S, Bolaños JP (2006) Nitric oxide, cell bioenergetics and neurodegeneration. J Neurochem 97:1676–1689. doi:10.1111/j.1471-4159.2006.03988.x
Butler TL, Kassed CA, Pennypacker KR (2003) Signal transduction and neurosurvival in experimental models of brain injury. Brain Res Bull 5:339–351. doi:10.1016/S0361-9230(02)00926-7
Duranteau J, Chandel NS, Kulisz A et al (1998) Intracellular signaling by reactive oxygen species during hypoxia in cardiomyocytes. J Biol Chem 273:11619–11624. doi:10.1074/jbc.273.19.11619
Leary SC, Hills BC, Lyons CN et al (2002) Chronic treatment with azide in situ leads to an irreversible loss of cytochrome c oxidase activity via holoenzyme dissociation. J Biol Chem 277:11321–11328. doi:10.1074/jbc.M112303200
Safiulina D, Veksler V, Zharkovsky A et al (2006) Loss of mitochondrial membrane potential is associated with increase in mitochondrial volume: physiological role in neurones. J Cell Physiol 206:347–353. doi:10.1002/jcp.20476
Marino S, Marani L, Nazzaro CP et al (2007) Mechanisms of sodium azide-induced changes in intracellular calcium concentration in rat primary cortical neurons. Neurotoxicology 28:622–629. doi:10.1016/j.neuro.2007.01.005
Hoyagi Y, Yamada T, Nishioka K et al (2000) Selective increase in cellular Aβ42 is related to apoptosis but not necrosis. Neuroreport 11:167–171
Grammatopoulos T, Morris K, Ferguson P et al (2002) Angiotensin protects cortical neurons from hypoxic-induced apoptosis via the angiotensin type 2 receptor. Brain Res Mol Brain Res 99:114–124. doi:10.1016/S0169-328X(02)00101-8
Bertrand R, Solary E, O’Connor P et al (1994) Induction of a common pathway of apoptosis by staurosporine. Exp Cell Res 211:314–321. doi:10.1006/excr.1994.1093
Vanlangenakker N, Vanden Berghe T, Krysko D et al (2008) Molecular mechanisms and pathophysiology of necrotic cell death. Curr Mol Med 8:207–220. doi:10.2174/156652408784221306
Lobner D (2000) Comparison of the LDH and MTT assays for quantifying cell death: validity for neuronal apoptosis? J Neurosci Methods 96:147–152. doi:10.1016/S0165-0270(99)00193-4
Zhang F, Yin W, Chen J (2004) Apoptosis in cerebral ischemia: executional and regulatory signaling mechanisms. Neurol Res 26:835–845. doi:10.1179/016164104X3824
Previati M, Lanzoni I, Astolfi L et al (2007) Cisplatin cytotoxicity in organ of corti-derived immortalized cells. J Cell Biochem 101:1185–1197. doi:10.1002/jcb.21239
Cavallini S, Marti M, Marino S et al (2005) Effects of chemical ischemia in cerebral cortex slices. Focus on nitric oxide. Neurochem Int 47:482–490. doi:10.1016/j.neuint.2005.06.003
Varming T, Drejer J, Frandsen A et al (1996) Characterization of a chemical anoxia model in cerebellar granule neurons using sodium azide: protection by nifedipine and MK-801. J Neurosci Res 44:40–46. doi :10.1002/(SICI)1097-4547(19960401)44:1<40::AID-JNR5>3.0.CO;2-I
Kume T, Nishikawa H, Taguchi R et al (2002) Antagonism of NMDA receptors by σ receptor ligands attenuates chemica ischemia-induced neuronal death in vitro. Eur J Pharmacol 455:91–100. doi:10.1016/S0014-2999(02)02582-7
Shahidullah M, Duncan A, Strachan PD et al (2002) Role of catalase in the smooth muscle relaxant actions of sodium azide and cyanamide. Eur J Pharmacol 435:93–101. doi:10.1016/S0014-2999(01)01540-0
Ahern GP, Klyachko VA, Jackson MB (2002) cGMP and S-nitrosylation: two routes for modulation of neuronal excitability by NO. Trends Neurosci 25:510–517. doi:10.1016/S0166-2236(02)02254-3
Fiscus RR (2002) Involvement of cyclic GMP and protein kinase G in the regulation of apoptosis and survival in neural cells. Neurosignals 11:175–190. doi:10.1159/000065431
Koesling D, Russwurm M, Mergia E et al (2004) Nitric oxide-sensitive guanylyl cyclase: structure and regulation. Neurochem Int 45:813–819. doi:10.1016/j.neuint.2004.03.011
Berry EV, Toms NJ (2006) Pyruvate and oxaloacetate limit zinc-induced oxidative HT-22 neuronal cell injury. Neurotoxicology 27:1043–1051. doi:10.1016/j.neuro.2006.05.011
Farooqui AA, Ong W-Y, Horrock LA (2006) Inhibitors of brain phospholipase A2 activity: their neuropharmacological effects and therapeutic importance for the treatment of neurologic disorders. Pharmacol Rev 58:591–620. doi:10.1124/pr.58.3.7
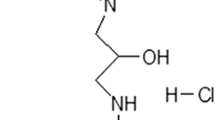
Abdul HM, Calabrese V, Calvani M et al (2006) Acetyl-l-carnitine-induced up regulation of heat shock proteins protects cortical neurons against amyloid-beta peptide 1–42-mediated oxidative stress and neurotoxicity: implications for Alzheimer’s disease. J Neurosci Res 84:398–408. doi:10.1002/jnr.20877
Bigini P, Larini S, Pasquali C et al (2002) Acetyl-l-carnitine shows neuroprotective and neurotrophic activity in primary culture of rat embryo motoneurons. Neurosci Lett 329:334–338. doi:10.1016/S0304-3940(02)00667-5
Zanelli S, Solenski NJ, Rosenthal RE et al (2005) Mechanisms of ischemic neuroprotection by acetyl-l-carnitine. Ann N Y Acad Sci 1053:153–161. doi:10.1196/annals.1344.013
Festjens N, Vanden Berghe T, Vandenabeele P (2006) Necrosis, a well-orchestrated form of cell demise: signalling cascades, important mediators and concomitant immune response. Biochim Biophys Acta 1757:1371–1387. doi:10.1016/j.bbabio.2006.06.014
Selvatici R, Falzarano S, Franceschetti L et al (2006) Differential activation of protein kinase C isoforms following chemical ischemia in rat cerebral cortex slices. Neurochem Int 49:729–736. doi:10.1016/j.neuint.2006.06.003
Siniscalchi A, Cavallini S, Marino S et al (2006) Effects of chemical ischemia in cerebral cortex slices. Focus on mitogen activated protein kinase cascade. Ann N Y Acad Sci 1090:445–454. doi:10.1196/annals.1378.047
Gao J, Zhu ZR, Ding HQ et al (2007) Vulnerability of neurons with mitochondrial dysfunction to oxidative stress is associated with down-regulation of thioredoxin. Neurochem Int 50:379–385. doi:10.1016/j.neuint.2006.09.007
Acknowledgments
This work was supported by Grants from University of Ferrara, Italy, from Associazione Emma e Ernesto Rulfo per la Genetica Medica, Parma, Italy and from Fondazione Cassa di Risparmio di Ferrara. We are grateful to Dr. Amanda Neville for the English revision of the text.
Author information
Authors and Affiliations
Corresponding author
Additional information
Rita Selvatici and Maurizio Previati equally contributed to the work.
Rights and permissions
About this article
Cite this article
Selvatici, R., Previati, M., Marino, S. et al. Sodium Azide Induced Neuronal Damage In Vitro: Evidence for Non-Apoptotic Cell Death. Neurochem Res 34, 909–916 (2009). https://doi.org/10.1007/s11064-008-9852-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11064-008-9852-0